Targeted Therapy for Melanoma - Dr. Michael Davies
•
7 likes•10,882 views
Dr. Michael Davies presents the latest information on targeted melanoma therapies at the MRF's Patient Symposium at MD Anderson Cancer Center on January 31, 2015.
Report
Share
Report
Share
Download to read offline
Recommended
Overview about Total Neoadjuvant therapy in locally advanced carcinoma RectumTotal Neoadjuvant therapy in locally advanced carcinoma Rectum

Total Neoadjuvant therapy in locally advanced carcinoma RectumCancer surgery By Royapettah Oncology Group
Landmark trials in Breast Cancer surgery trials about RM,BCS,MRM in Invasive Breast Cancer as Landmark trials in breast Cancer surgery - NSABP 04,06,MILAN,EORTC 10853, ECO...

Landmark trials in breast Cancer surgery - NSABP 04,06,MILAN,EORTC 10853, ECO...Dr.Bhavin Vadodariya
Recommended
Overview about Total Neoadjuvant therapy in locally advanced carcinoma RectumTotal Neoadjuvant therapy in locally advanced carcinoma Rectum

Total Neoadjuvant therapy in locally advanced carcinoma RectumCancer surgery By Royapettah Oncology Group
Landmark trials in Breast Cancer surgery trials about RM,BCS,MRM in Invasive Breast Cancer as Landmark trials in breast Cancer surgery - NSABP 04,06,MILAN,EORTC 10853, ECO...

Landmark trials in breast Cancer surgery - NSABP 04,06,MILAN,EORTC 10853, ECO...Dr.Bhavin Vadodariya
More Related Content
What's hot
What's hot (20)
Adjuvant treatment in early and localy advanced breast cancer

Adjuvant treatment in early and localy advanced breast cancer
MANAGEMENT OF EARLY OPERABLE HER2+ BREAST CANCER.pptx

MANAGEMENT OF EARLY OPERABLE HER2+ BREAST CANCER.pptx
Adjuvant Endocrine Therapy For Postmenopausal Breast Cancer

Adjuvant Endocrine Therapy For Postmenopausal Breast Cancer
ROLE OF NEOADJUVANT CHEMORADIATION IN LOCALLY ADVANCED BREAST CANCER

ROLE OF NEOADJUVANT CHEMORADIATION IN LOCALLY ADVANCED BREAST CANCER
Dr arun Triple Negative Breast cancer Presentation

Dr arun Triple Negative Breast cancer Presentation
Viewers also liked
Community Oncology Clinical Debates: Advanced Melanoma
Downloadable slide decks are a great tool for self study and teaching purposes. They are non-certified resources available to enhance your knowledge.
Review a downloadable slide deck by Antoni Ribas, MD, PhD, covering the most clinically relevant new data reported from Community Oncology Clinical Debates: Advanced Melanoma.
Target Audience
This educational activity has been designed to meet the unique learning needs of oncologists involved in the treatment of patients with advanced melanoma.
Disclaimer
This slide deck in its original and unaltered format is for educational purposes and is current as of June 2012. All materials contained herein reflect the views of the faculty, and not those of IMER, the CME provider, or the commercial supporter. These materials may discuss therapeutic products that have not been approved by the US Food and Drug Administration and off-label uses of approved products. Readers should not rely on this information as a substitute for professional medical advice, diagnosis, or treatment. The use of any information provided is solely at your own risk,
and readers should verify the prescribing information and all data before treating patients or employing any therapeutic products described in this educational activity.
Community Oncology Clinical Debates: Advanced Melanoma

Community Oncology Clinical Debates: Advanced MelanomaInstitute For Medical Education and Research (IMER)
This slidedeck presents an up-to-date disease overview of BCC, reviews current treatment options in BCC, explains the hedgehog signaling pathway and its role in BCC, review recent data of the first-in-class hedgehog inhibitor, vismodegib, and other novel agents in clinical trials. Faculty will also review recently approved novel agents in melanoma, to include treatment planning and managing adverse events. Case studies will demonstrate the practical application of current and emerging clinical evidence for the treatment of BCC and melanoma. During the panel discussion, faculty will discuss the importance of cross-communication in the treatment planning process and strategies to optimize the continuum of care for patients with BCC. Targeted Therapy for the Treatment of Basal Cell Carcinoma and Melanoma

Targeted Therapy for the Treatment of Basal Cell Carcinoma and MelanomaInstitute For Medical Education and Research (IMER)
Gene Profiling in Clinical Oncology - Slide 5 - R. Labianca - What do we do t...

Gene Profiling in Clinical Oncology - Slide 5 - R. Labianca - What do we do t...European School of Oncology
Viewers also liked (20)
Community Oncology Clinical Debates: Advanced Melanoma

Community Oncology Clinical Debates: Advanced Melanoma
Targeted Therapy for the Treatment of Basal Cell Carcinoma and Melanoma

Targeted Therapy for the Treatment of Basal Cell Carcinoma and Melanoma
Gene Profiling in Clinical Oncology - Slide 5 - R. Labianca - What do we do t...

Gene Profiling in Clinical Oncology - Slide 5 - R. Labianca - What do we do t...
Potential cancer cells and therapy to treat melanoma

Potential cancer cells and therapy to treat melanoma
Melanoma sign symptom management surgery prevention ملانوم 

Melanoma sign symptom management surgery prevention ملانوم
Similar to Targeted Therapy for Melanoma - Dr. Michael Davies
Dr. Cathy Eng delivers updates on current trials and studies impacting the treatment of colon and rectal cancer. Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...

Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...Fight Colorectal Cancer
NY Prostate Cancer Conference - M.H. Hussain - Session 7: Role of predictive ...

NY Prostate Cancer Conference - M.H. Hussain - Session 7: Role of predictive ...European School of Oncology
Similar to Targeted Therapy for Melanoma - Dr. Michael Davies (20)
Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...

Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...
Report Back from San Antonio Breast Cancer Symposium (SABCS) 2023: Spotlight ...

Report Back from San Antonio Breast Cancer Symposium (SABCS) 2023: Spotlight ...
Research Biopsies and Translational Research - Dr. Jennifer Wargo

Research Biopsies and Translational Research - Dr. Jennifer Wargo
Question of Quality Conference 2016 - Personalized Cancer Medicine

Question of Quality Conference 2016 - Personalized Cancer Medicine
CCO Rational Options in Breast Cancer : How Molecular Understanding Informs T...

CCO Rational Options in Breast Cancer : How Molecular Understanding Informs T...
Targeted Therapy for Uveal Melanoma - Richard Carvajal, MD

Targeted Therapy for Uveal Melanoma - Richard Carvajal, MD
Terapia quimiohormonal en Cáncer de Próstata Sensible a la Castración (CSPC)

Terapia quimiohormonal en Cáncer de Próstata Sensible a la Castración (CSPC)
Personalized vs. Precision, let’s call it Medicine

Personalized vs. Precision, let’s call it Medicine
Colon Cancer Updates - 2015/2016 - Based on ASCO GI 2016

Colon Cancer Updates - 2015/2016 - Based on ASCO GI 2016
NY Prostate Cancer Conference - M.H. Hussain - Session 7: Role of predictive ...

NY Prostate Cancer Conference - M.H. Hussain - Session 7: Role of predictive ...
More from Melanoma Research Foundation
More from Melanoma Research Foundation (20)
Metastatic Uveal Melanoma Treatment - 2019 CURE OM Symposium

Metastatic Uveal Melanoma Treatment - 2019 CURE OM Symposium
Uveal Melanoma Liver Metastases - 2019 CURE OM Symposium

Uveal Melanoma Liver Metastases - 2019 CURE OM Symposium
CURE OM Patient Registry Update - 2019 CURE OM Symposium

CURE OM Patient Registry Update - 2019 CURE OM Symposium
Targeted Therapy Clinical Trial Update - 2019 CURE OM Symposium

Targeted Therapy Clinical Trial Update - 2019 CURE OM Symposium
Auburn Uveal Melanoma Study - 2019 CURE OM Symposium

Auburn Uveal Melanoma Study - 2019 CURE OM Symposium
The Past, Present and Future of Uveal Melanoma - 2019 CURE OM Symposium

The Past, Present and Future of Uveal Melanoma - 2019 CURE OM Symposium
Clinical Trials Panel Discussion: 2019 CURE OM Symposium

Clinical Trials Panel Discussion: 2019 CURE OM Symposium
A Potential Molecular Target for the Treatment of Uveal Melanoma

A Potential Molecular Target for the Treatment of Uveal Melanoma
Recently uploaded
Recently uploaded (20)
ICT Role in 21st Century Education & its Challenges.pptx

ICT Role in 21st Century Education & its Challenges.pptx
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
Salient Features of India constitution especially power and functions

Salient Features of India constitution especially power and functions
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Targeted Therapy for Melanoma - Dr. Michael Davies
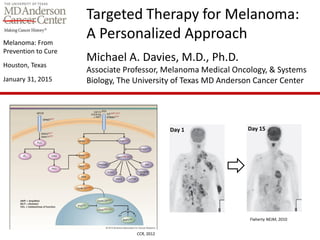
- 1. Melanoma: From Prevention to Cure Houston, Texas January 31, 2015 Targeted Therapy for Melanoma: A Personalized Approach Michael A. Davies, M.D., Ph.D. Associate Professor, Melanoma Medical Oncology, & Systems Biology, The University of Texas MD Anderson Cancer Center CCR, 2012 Day 1 Day 15 Flaherty NEJM, 2010
- 2. Disclosure Information I have the following financial relationships to disclose: Consultant for: GSK, Roche/Genentech, Novartis, Sanofi-Aventis Grant/Research support from: GSK, Roche/Genentech, Astrazeneca, Merck, Myriad, Oncothyreon, Sanofi-Aventis I will discuss off-label or investigational use of: Imatinib, TAK-733
- 3. New Agents for Stage IV Melanoma US Approval Targeted TherapyImmunotherapy Pre-1998 Approvals w/o (+) randomized trials 1998-2011 No approvals 2011-2014 7 new regimens approved 1975 DTIC 1998 HD-IL2 1998 - 2011 2011 Ipilimumab Vemurafenib 2012 2013 Dabrafenib Trametinib 2014 Dabrafenib +Trametinib Pembrolizumab Nivolumab Chemotherapy
- 4. Targeted Therapy: A Personalized Approach • What is targeted therapy? • Why do we use targeted therapy for melanoma? • What have we learned & what are we working on
- 5. A Brief History of Chemotherapy • WW I: Nitrogen mustards gas • → 1946 1st successful systemic treatment of cancer with chemotherapy • 1950s-1980s most standard chemotherapies • 1977 First report of an oncogene • 1980s-2000s Oncogenes in most cancers Dr. Sidney Farber And Patient ‘Targeted Therapy’: Treat cancer by targeting the genes activated in cancer cells
- 6. Targeted Therapy: Why Melanoma? • Overall melanomas have more mutations than any other type of cancer • Targetable mutations can be identified in ~70% of patients with cutaneous melanoma
- 7. PET CT BRAFi 6 Weeks The Promise of Targeted Therapy: BRAF
- 8. Effective for Brain Metastases Long et al, Lancet Oncology, 2013 BRAFi 32 Weeks
- 9. Clinical Activity of BRAF Inhibitors Tumor Grew Tumor Shrank • 50% of patient achieve a “clinical response” • 90% achieve disease control, usually very quickly (<1 month) • KEY: Only work in patients that have a BRAFV600 mutation • If don’t have a mutation they will make the tumors grow faster FDA-Approved BRAF Inhibitors • Vemurafenib, 2011 • Dabrafenib, 2013 Champman et al, NEJM, 2011
- 10. Baseline 2 Weeks 16 Weeks Resistance to BRAF Inhibitors Average response duration ~6 months
- 11. BRAFi: Why Does Resistance Happen MAPK & PI3K Pathway Re-Activated MAPK Pathway Re-Activated Unknown Mechanism of Resistance Shi,… Lo, Cancer Discovery, 2013 All patients still have the BRAFV600 mutation at the time of resistance → Rationale to combine BRAF with other MAPK inhibitors
- 12. Combinations to Overcome Resistance: Targeted Therapy + Targeted Therapy Testing of other combinations underway -100 -80 -60 -40 -20 0 20 40 60 80 100 Tumor Shrank BRAFi + MEKi • ~100% disease control rate • Average response duration 11 months (~2X BRAFi) • Less skin toxicity than BRAFi alone Flaherty et al, NEJM, 2012
- 13. A New Challenge: Heterogeneity Shi,… Lo, Cancer Discovery, 2013 Tumor # Different tumor with different resistance mechanisms in the same patient
- 14. A New Hypothesis: Combining BRAF Inhibitors & Immunotherapy BRAF Inhibitors • High rates of responses • Responses generally < 1 yr Immunotherapies • Lower rates of responses • Responses can → cures Can BRAF Inhibitors + Immunotherapy → High rate of cures?
- 15. Combinations to Overcome Resistance: Targeted Therapy + Immune Therapy Liu, …Hwu, CCR, 2013 Frederick,…Wargo, CCR, 2013 In Mice In Patients Challenges: • More side effects • Which agents to combine • How to combine them
- 16. Targeted Therapy: Beyond BRAFV600 ‘Wild-type’ BRAF >50% cutaneous MM >80% non-cutaneous BRAF V600 Mutant BRAF V600 Wild-Type
- 17. Different Types of Melanomas Have Different Mutations Cutaneous w/o Chronic Sun Damage (C.S.D) Acral Melanoma Mucosal Melanoma Uveal 20-30% mutations in c-KIT 45% BRAF Mutations 20% NRAS Mutations Acral: 20% BRAF 10% NRAS Mucosal: 3% BRAF 5% NRAS Virtually No BRAF/NRAS ~80% mutations in GNAQ/GNA11
- 18. Targeted Therapy for KIT-Mutant Melanoma Hodi et al, JCO, 2008 Imatinib Imatinib • 1% chance of response in unselected melanoma patients • 30-50% chance of response with recurrent KIT mutations
- 19. Melanoma TCGA: Initial Results 100 150 215 NRAS mut (28%) BRAF mut (52%) NF1 mut (14%) MITF amp (4%) BRAF amp (12%) MDM2 amp(12%) NOTCH2 amp (5%) KIT amp (3%) KIT mut (3%) PDGFRα amp (3%) PDGFRα mut (10%) CCND1 amp (4%) TERT amp (5%) PPP6C mut (7%) PTEN del (12%) PTEN mut (8%) CDKN2A del(40%) CDKN2A mut (13%) TP53 mut (7%) 50 “Wild-Type”BRAFV600NRAS Cancer Genome Atlas Research Network, 2012
- 20. Dahlman, et al Cancer Discovery, 2012 MEKi for Non-V600 BRAF Mutations Cutaneous & Unknown Primary melanomas WITHOUT BRAFV600 or NRAS mutations MDACC, n=113 Siroy, et al, J Inv Derm, 2014 Melanoma BRAF L597V TAK-733 (MEKi)
- 21. Targeted Therapy for Melanoma: What Have We Learned & What Are We Working On
- 22. Targeted Therapy: What We Have Learned • Virtually all patients with cutaneous melanomas have >100 mutations • BRAF-Mutant Melanoma (~ 50%) – Inhibiting driver oncogenes can → benefit – Right patient, right drug, right dose – Combinations can be even better! • Have to identify ways to understand, prevent & overcome resistance
- 23. Targeted Therapy: What We Are Working On • Identify mutations that help us to manage patients across the full continuum of melanoma • BRAF-Mutant Melanoma – Benefit of BRAF and MEK inhibitors in earlier stages – New combinations • Testing new strategies for other mutations
- 24. Thank you for your attention and to our patients Michael Davies, M.D., Ph.D. Departments of Melanoma Medical Oncology and Systems Biology University of Texas M. D. Anderson Cancer Center mdavies@mdanderson.org Research Support NIH/NCI Cancer Prevention Research Institute of Texas (CPRIT) Melanoma Research Alliance Melanoma Research Foundation American Society for Clinical Oncology MDACC SPORE in Melanoma MDACC Physician-Scientist Program Dunn Foundation for Chemical Genomics GlaxoSmithKline AstraZeneca Genentech Merck Myriad Oncothyreon Sanofi-Aventis Dr. John E. Davies, 1921 - 1999
