MPGN Pam
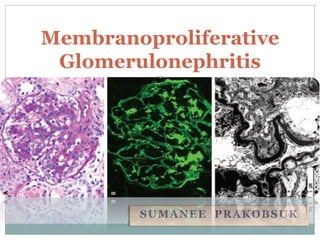
- 1. Membranoproliferative Glomerulonephritis SUMANEE PRAKOBSUK
- 2. Scopes Classification Pathophysiology Pathology Clinical features Treatment
- 3. Overview Also termed mesangiocapillary glomerulonephritis. MPGN accounts for approximately 7 to 10% of all cases of biopsy-confirmed glomerulonephritis. Brenner&Rector’s: The Kidney 9th Edition
- 4. Classification Idiopathic Secondary Type I Infection Hepatitis C and B Type II Visceral abscess Infectious endocarditis Type III Shunt nephritis Mycoplasma infection Rheumatologic disease SLE Scleroderna Sjogren syndrome Mixed essential cryoglobulinemia with or without hepatitis C infection Malignancy Carcinoma,Lymphoma,Leukemia Brenner&Rectoer’s 9th Edition Complement deficiency Hereditary C2,C4 deficiency
- 5. Classification Traditionally been classified into three subtypes MPGN types I MPGN types II MPGN types III The primary basis for this classification is histologic, the appearance of the capillary wall by electron microscopy and the location of electron-dense deposits. Brenner&Rector’s: The Kidney 9th Edition
- 6. Type I : Subendothelial deposits Type II : Dense deposits in Lamina densa of GBM Type III : Subendothelial &Subepithelial deposits
- 7. Membranoproliferative Glomerulonephritis Type I Epidemiology The majority of patients with MPGN are children between the ages of 8 and 16 years. The proportions of males and females with the disorder are nearly equal. Brenner&Rector’s: The Kidney 9th Edition
- 8. IMMUNE-COMPLEX-MEDIATED MPGN Pathogenesis MPGN typeI Results from the deposition of immune complexs in the glomeruli owing to persistent antigenemia. Type I MGPN often is secondary to recognizable causes. Brenner&Rector’s: The Kidney 9th Edition
- 9. Secondary MPGN Brenner&Rector’s: The Kidney 9th Edition
- 10. Pathogenesis of MPGN type I
- 12. Pathology of MPGN Type I LM Mesagial proliferation Endocapillary proliferation Diffuse global capillary wall thickening “Lobular glomerulonephritis” Brenner&Rector’s: The Kidney 9th Edition
- 13. Pathology of MPGN Type I LM Mesangial interposition Doubing or replication of GBM Brenner&Rector’s: The Kidney 9th Edition
- 14. Pathology of MPGN Type I IF Granular staining for complement, especially C3, and usually immunoglobulins (IgG or IGM) GBM,Mesangial Few at TBM Brenner&Rector’s: The Kidney 9th Edition
- 15. Pathology of MPGN Type I EM The ultrastructural hallmark of type I Subendothelial Electron- dense -deposits Mesangial interposition Brenner&Rector’s: The Kidney 9th Edition
- 16. Clinical Features of MPGN typeI Nephrotic syndrome 40-70% Acute nephritic syndrome 25 % Asymptomatic proteinuria and hematuria 25 % HT (not severe) Renal insufficiency 50% Pediatr Nephrol.2010;25:1409-18.
- 17. Membranoproliferative Membranoproliferative Glomerulonephritis Type III Type III MPGN occurs in a very small number of children and young adults. Clinical features of disease quite similar to those of type I MPGN. Regardless of the pathologic distinctions of MPGN type III ,few distinguishing clinical characteristics are noted in these patients. EM: subendothelial and subepithelial EDD Brenner&Rector’s: The Kidney 9th Edition
- 18. Membranoproliferative Glomerulonephritis Type II Dense Deposit Disease Epidemiology About 25% of MPGN in children but is much less common in adults. The large majority of patients are children 8 - 16 years. 2-3person/million. Female:male = 3:2 J Am Soc Nephrol 16: 1392–1404, 2005 Brenner&Rector’s: The Kidney 9th Edition
- 19. Pathogenesis of MPGN type II (DDD) DDD is characterized by deposits of dense material within the basement membranes of glomeruli, Bowman’s capsule, and tubules. A porcine model of MPGN Massive deposition of C3 and the terminal C5b9 complement complex (the membrane attack complex). No immune complex deposits were detected in renal tissue. Dysregulation of alternative pathway. J Am Soc Nephrol 16: 1392–1404, 2005 Brenner&Rector’s: The Kidney 9th Edition
- 20. Peter F. Zipfel*‡ and Christine Skerka* NATuRe RevIeWs Immunology vOLuMe 9 | OCTObeR 2009
- 21. Factor H Factor H
- 22. Pathogenesis of MPGN type II (DDD) Three distinct mechanisms result in uncontrolled activation of C3 convertase. 1 .The development of an autoantibody, the C3 nephritic factor (C3NeF). - Protects C3 convertase form factor H. 2. The absence of circulating factor H . 3. The presence of a circulating inhibitor of factorH. Brenner&Rector’s: The Kidney 9th Edition
- 23. Dysregulation of the alternative pathway
- 24. To investigate causes for AP dysregulation 32 patients with DDD (renal Bx C3+ and intramembranous electron-dense deposits by EM) C3Nef detection assays Factor H antoantibodies(FHAA) Factor B autoantibodies (FBAA) Factor H Mutation screening.
- 25. Results C3Nef detection assays: 25 patiests (78%) Factor H antoantibodies(FHAA) 1 patients (3%) Factor B autoantibodies (FBAA) 3 patiests (9%) Factor H Mutation screening. 26 patiests(81%) carried at least one copy of the FH His402 polymorphism.
- 27. Pathology :MPGN II( DDD) LM 5 distinct patterns: (1) Membranoproliferative pattern (2) Mesangioproliferative pattern (3) Crescentic pattern (4) Acute proliferative and exudative pattern (5) Unclassified dense deposit disease. Sibley RK, Kim Y. Dense intramembranous deposit disease: new pathologic features. Kidney Int. 1984;25:660-670.
- 28. Pathology :MPGN II( DDD) •Intense capillary wall linear to bandlike staining for C3 •Little or no staining for Ig
- 29. Pathology :MPGN II( DDD) A bandlike intramembranous dense deposit
- 30. Clinical Features :MPGN type II (DDD) ¾ of patients have all of the components of nephrotic syndrome on presentation. ¼ of patients have acute nephritic syndrome. HT is typically mild, but may be severe in some cases. Renal dysfunction occurs in at least half of cases and is more common in adults han in children. Brenner&Rector’s: The Kidney 9th Edition
- 31. Clinical Features :MPGN type II (DDD) May have deposits in the retina There is no correlation between the severity of kidney and ocular involvement. J Am Soc Nephrol 16: 1392–1404, 2005
- 32. Clinical Features :MPGN type II (DDD) DDD may be associated with the syndrome of acquired partial lipodystrophy. About 80% of patients with this syndrome have low C3 levels and C3NeF. About 20% of patients develop MPGN
- 33. Prognosis:MPGN II (DDD) The prognosis for type II is worse than that for type I, worse in adults than in children. Clinical remissions are rare,occurring in fewer than 5% of children. Patients generally reach ESRD in 8 to 12 years from the onset of disease. Brenner&Rector’s: The Kidney 9th Edition
- 34. Proposal for A new classification of MPGN Proposal of a New classification Immune complex mediated MPGN. Complement mediated MPGN. Sanjeev Sethi, Fernando C. Semin Nephrol 31:341-348 © 2011
- 35. Semin Nephrol 31:341-348 © 2011
- 36. Treatment Based on the heterogeneity of cause and pattern of histologic injury of MPGN. All patients with MPGN must be thoroughly evaluated for underlying diseases before classifying as idiopathic MPGN, and before any specific treatment decisions can be made. When there is a secondary MPGN, treatment should be directed against that cause
- 37. Treatment Idiopathic MPGN is now an uncommon condition. The few RCTs of treatment of idiopathic MPGN in children and adults have given inconsistent and largely inconclusive results . Many of the reported trials have weak experimental design or are underpowered, and the evidence base underlying the recommendations for treatment of “idiopathic” MPGN is very weak.
- 38. Treatment Immunosuppressive agent Prednisolone Cytotoxic agent Cyclophosphamide Mycophenolate Antiplatelet Non-specific Tx
- 39. Double-blinded RCT. Compare altermate day prednisolone VS placebo Between February 1970 and October 1980 Pediatr Nephrol(1992)6:123-130
- 40. Treatment of mesangiocapillary glomerulonephritis with alternate-day prednisone Inclusion criteria Inclusion criteria 1. Biopsy-prove MCGN 2. GFR by crcl ≥ 70 ml/min per 1.73 m 2 3. Heavy proteinuria ≥ 40 mg/h per m 2 4. No evidence of SLE, HSP, nephritis accompanying bacteremia (such as IE), or malaria, 5. No treatment with corticosteroids during the year prior to entry into the trial or with immunosuppressive agents at any time. Pediatr Nephrol(1992)6:123-130
- 41. Treatment of mesangiocapillary glomerulonephritis with alternate-day prednisone • Prednisolone 40 mg/m2 alternate days Treatment • Maximum dose 60 mg • 5 years • Treatment failure: increase from baseline in serum creatinine of 30% or more, or more than 0.4 mg/dl. Outcome • Renal failure : cr ≥ 4 mg/dl • Stable: no change or increase in S.Cr <0.4 mg/dl Pediatr Nephrol(1992)6:123-130
- 43. 61% P=0.07 12% Mean Tx 41 months Alternate- day prednisone therapy Treatment failure improves the prognosis of Treatment 40% patients with MCGN, when used at doses control 54% of 40 mg/m2.
- 44. Prednisolone for Idiopathic MPGN There has been no systematic evaluation of glucocorticoid therapy for idiopathic MPGN in adults. Retrospective studies showed no clear benefit from glucocorticoid therapy, but treatment was not as prolonged in adults as it was in children. Kidney International,Vol55(1999)pps41-s46
- 45. Cytotoxic Agent Cyclophosphamide Mycophenolate
- 46. RCT Compare Combination Cyclophosphamide 1.5-2.0 mg/dl Coumadin keep PT 2-2.5 times Dipyridamole start 25 mg qid full dose 100 mg qid No specific therapy
- 47. Cyclo Inclusion MPGN type I &II ( no crescent) Proteinuria > 2 g/day Ccr < 80 mI/mm. Ruled out Secondary causes of MPGN Exclusion Active infectin Previous tuberculosis, Hx PU Uncontrolled HT Outcome Change of crcl from baseline at 18 months
- 48. Baseline MPGN Type I Control(N =25) Treatmemt (N=22) Age 33 (6-70) 38 (12-77) Crcl (ml/min/1.73 m2) 64 (18-135) 65 (16-113) Cr (mg/dl) 1.8 (0.7-7.0) 1.7 (0.7-5.9) Proteinuria ( g/day) 5.2 (0.85-16.4) 3.6(0.4-8.3) MPGN Type II Control(N =7) Treatmemt (N=5) Age 19(7-58) 17.5 (6-26) Crcl (ml/min/1.73 m2) 87 (51-131) 63 (37-115) Cr (mg/dl) 0.9 (0.3-1.2) 1.2 (0.7-2.1) Proteinuria ( g/day) 4.3 (0.1-11.7) 6.8 (1.7-12.8)
- 49. Change in Crcl /18 months MPGN Type I MPGN Type II P=0.4 P=0.5 -1 0 -14 -8
- 50. Proteinuria ,g/day No significant benefit could be observed over a period of 18 months.
- 51. Retrospective analysis Treatment group MMF started 500 mg/daymaximum 2 gm /day Oral Prednisolone 60 mg/day , tapering to 20mg within 2 months and withdrawn by 1 year. Control group Did not receive immunosuppressive therapy
- 53. Change in proteinuria over time in control and MMF treated patients P=0.03 control MMF
- 54. Change in creatinine clearance over time in control and MMF treated patients. MMF P=0.06 control
- 55. Antiplatelete Rationale Demonstrations of platelet activation and deposition of platele antigen are invole in initiating or contributing to glomerular injury . Glomerular prostaglandin and thromboxane systesis in rat nephrotoxic serum nephritis.J Clin Invest 1983;72:1439-48
- 56. Randomized,double blind placebo-controlled trial . Renal biopsy prove MPGN TypeI 1975-1981 Only 2 were receiveing therapy with prednisolone,with discontinued at the time of entry None had been treated with cytotoxic drugs. Excluded SLE,essential mixed cryoglobulinemia,PIGN,dialysis James V.Donadio,JR.Nejm1984;vol310(22)
- 57. Treatment Dipyridamole 75 mg Aspirin 325 mg 12 months Outcome Treatment failure: decline of 25% Iotalamate clearance from pre treatment. James V.Donadio,JR.Nejm1984;vol310(22)
- 58. Pretreatment Clinical manifestations Treatment Placebo N=21 N=19
- 59. Treatment failures(loss of renal function) P<0.05 James V.Donadio,JR.Nejm1984;vol310(22)
- 60. Change in Iothalamate clearance -1.3 ml/min -19.6 ml/min P<0.02 No differences between the groups Proteinuria,Amount of RBC cast, C3,C4,CH50 James V.Donadio,JR.Nejm1984;vol310(22)
- 62. Follow up 7 years ESRD - 47 % in Placebo (33 months) - 14 % in Treatment (62 months) GFR and was better maintained, and progression to ESRD occurred less often and over a longer period,in the group treated with platelet-inhibitor drugs than in the group given placebo James V.Donadio,JR.Nejm1984;vol310(22)
- 63. Plasmapheresis Case report , improve renal function MPGN type II Rucurrent MPGN typeII post trasplant
- 64. Immunosuppressive drugs Level of Author Design N Rx Duration Results/Comments Evidence [Rx : C] 1 Tarshish RCT 80 [47/33] Pred 40 mg/m2 130 mo Children only, predomly MPGN I, stable renal fn AD vs pcb Pred 61% [Rx] vs 12% [C] No difference found, 1 Cattran RCT 59 CY + coumadin 18 mo [27/32] + dipyridamole mixed MPGN I > II CY vs No Rx 1985 3 Strife UCT 17 Pred 2 mk AD 2y MPGN III only, nephrotic range [16/1] did worse, 3/16 dvled RF 3 Davis CT 27 Pred+IS [NS] - No effect [19/8] 3 Orlowski UCT 50 P/AZA/CP/chlorambucil 79 mo 10 y F/U, ↓ Uprot c triple drug in combi Rx 3 Ford UCT 19 PO/IV pred 2 mk + 8-10 wk, Children; 6.5 y F/U, early Rx, ACEI then x2-3 shorter course, y tapered ↓ glom prolif dose 3 Faedda UCT 19 IV/PO CY+P 10 mo 15/19 remission, 7 y F/U 1994 [different combinations] Adeera Levin., Mx of MPGN; Evidence based recommendations; KI 1999; 55: S41-S46
- 65. Antiplatelet Level Author Design Rx Durati Results/Comments of evi N [Rx : C] on 1 Donadio 1984 RCT Dipyridamole 75mg/d + ASA 325 12 mo -Tx : significantly delay rate of GFR ↓ Prospective mg/d vs Pcb [Sig difference at 7 y] 40 [21/19] -No change in Uprot, hematuria, Complement -Mild bleeding complication required discontinuation 15% 1 Zimmer man RCT Warfarin [INR 1.5-2] + dipyridamole [75-100 12 mo [2 y Prot restriction & HTN control standardized. Crossover mg qid] vs Pcb study] Sig reduction in proturia 18 [8/10] NS diff in renal fn Significantly ↓ Uprot 1 Zauner 1994 RCT ASA 500 mg/d + dipyridamole 75 36 mo [1 g Tx vs 8 g control] [Sig] 18 [9/9] mg/d PR : Tx 7/10 vs Control [I 15 + III [both : prot restriction 2/8 3] & HTN control] -Comment : short F/U SCr 1.8, Uprot 7 g/d Adeera Levin., Mx of MPGN; Evidence based recommendations; KI 1999; 55: S41-S46
- 66. Evidence based recommendations : KI 1999 Idiopathic MPGN 24 h Uprot & CCr <3 g >3 g Normal renal function Abn renal function Normal renal function Abn renal fn Grade C; Grade A; Grade B; Child : trial of steroid x 3 Child : trial of Adult : trial of ASA mo [AD; IV or PO 1 mk] steroids 40 mg/m2 325 mg/d & Or AD x 6-12 mo Dipyridamole 75-100 Adult : no Rx, observe mg tid x 6-12 mo F/U q 3 mo; BP, Lipid monitoring -No change : continued F/U, ↓ frequency - Increase cr or proteinuria Rx Adeera Levin., Mx of MPGN; Evidence based recommendations; KI 1999; 55: S41-S46
- 67. Pediatr Nephrol (2010) 25:1409–1418
- 70. Recurrence in KT 67-100% in MPGN II , graft loss 34-66% 20-33% in MPGN I Related to severity of previous disease > type of MPGN.
- 71. The Mayo Clinic Transplant database. On examination of the records of 1321 patients following kidney transplant over an 11-year period 29 patients of MPGN Excluded MPGN type II, secondary MPGN Follow up Protocal Bx 0,4,12,24,60 months Kidney International (2010) 77, 721–728
- 72. 52 months of follow up rMPGN 12 /29 patients (41.4%) Recurrence occurred during first 14 months( Median 3.3 months) Kidney International (2010) 77, 721–728
