Colon specific drug delivery
- 1. Colon – Specific Drug Delivery System
- 2. CONTENTS •Introduction •Anatomy & Physiology of Colon •Factors governing the colon drug delivery •Colon absorption •The major functions •Advantages •Pharmaceutical Approaches for Targeting Drugs to Colon •Platform Technologies for CTDDS •Evaluation •Conclusion
- 3. Introduction: Colon as a site for drug delivery. Colon was considered as BLACK-BOX as most of the drugs are absorbed from upper part of the GI tract. The site specific delivery of drugs to lower parts of the GIT is advantage for localized treatment of several colonic diseases(IDB). The CDDS drug release and absorption should not occur in the stomach as well as small intestine, but only released and absorbed once the system reaches to the colon.
- 4. Why is CDDS needed? Ensure direct treatment at the disease site. Lower dosing and less side effects. Beneficial in the treatment of colon diseases. Suitable absorption site for protein and peptide drugs. Used to prolong the drug therapy. B R Nahata College of Pharmacy Mandsaur (M.P.)
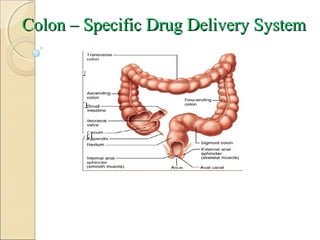
- 5. ANATOmy & PhySiOlOgy Of COlON GIT: •Stomach •Small intestine •Large intestine COLON: Colon Rectum Anal canal Colon consists: • Cecum • Ascending colon • Transverse colon • Descending colon • Sigmoid colon B R Nahata College of Pharmacy Mandsaur (M.P.)
- 6. lAyErS : •Serosa •Muscularis externa •Submucosa •mucosa B R Nahata College of Pharmacy Mandsaur (M.P.)
- 7. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 8. Factors governing the colon drug delivery Gastrointestinal transit Small intestinal transit Colonic transit Gastric emptying Stomach and intestinal pH Colonic micro flora and enzymes B R Nahata College of Pharmacy Mandsaur (M.P.)
- 9. pH of GIT: LOCATION Ph 1.STOMACH: 1.5-2.0 Fasted 3.0-5.0 Fed 5.0-6.5 2.SMALL INTESTINE: 6.0-7.5 Jejunum 6.4 Ileum 6.7-7.3 3.LARGE INTESTINE: 6.5-7.0 Right colon 6.6-7.0 Mid colon 6.6 Left colon 7.0 B R Nahata College of Pharmacy Mandsaur (M.P.)
- 10. Drug absorption in the colon Drug molecules pass from the apical to basolateral surface of the epithelial cell by 1. Transcellular - Passing through colonocytes. 2. paracellular - Passing between adjacent colonocytes. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 11. ThE mAjOr fuNCTiONS The absorptive capacity is very high; each day about 2000 ml of fluid enters the colon through the ileocecal valve from which more than 90% of the fluid is absorbed. Creation of a suitable environment for the growth of colonic microorganisms, such as Bacteroides, Eubacterium, and Enterobacteriaceae. Expulsion of the contents of the colon at a suitable time. Absorption of water and Na+ from the lumen, concentrating the fecal content, and secretion of K+ ions B R Nahata College of Pharmacy Mandsaur (M.P.)
- 12. AdvANTAgES Drugs are directly available at the target site. Comparatively lesser amount of required dose. Decreased side effects. Improved drug utilization. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 13. Pharmaceutical Approaches for Targeting Drugs to Colon pH sensitive systems Microbially triggered system ◦ Prodrugs ◦ Polysaccharide based systems Timed release systems Osmotically controlled drug delivery systems Pressure dependent release systems B R Nahata College of Pharmacy Mandsaur (M.P.)
- 14. pH sensitive systems Solid formulations for colonic delivery that are based on pH-dependent drug release mechanism are similar to conventional enteric-coated formulations. Utilize enteric polymers that have relatively higher threshold pH for dissolution. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 15. Polymers B R Nahata College of Pharmacy Mandsaur (M.P.)
- 16. mEChANiSm Of ACTiON B R Nahata College of Pharmacy Mandsaur (M.P.)
- 17. miCrObiAlly TriggErEd SySTEmS Bacterial count in the colon is much higher around 1011-1012 CFU/ml. 400 species Fundamentally anaerobic in nature. Predominant species: Bacteroides, Bifidobacterium and Eubacterium. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 18. Major metabolic processes occurring in the colon are hydrolysis and reduction ENzymES iN COlON Reducing enzymes Hydrolytic enzymes Nitroreductase Esterases Azoreductase Amidases N-oxide reductase Glycosidases Sulphoxide reductase Glucuronidase Hydrogenase Sulfatase Azoreductases, which reduces azo-bonds selectively and Polysaccharidases which degrades the polysaccharides . B R Nahata College of Pharmacy Mandsaur (M.P.)
- 19. AzObONd PrOdrugS Hydrolysis of sulphasalazine (i) into 5-aminosalicylic acid (ii) and sulfapyridine (iii). B R Nahata College of Pharmacy Mandsaur (M.P.)
- 20. glyCOSidE CONjugATES B R Nahata College of Pharmacy Mandsaur (M.P.)
- 21. NATurAl POlySACChAridES AS POlymEr fOr COlON drug dElivEry Chitoson Pectin Guar gum Chondroitin sulphate Dextran Almond gum Locust bean gum Cyclodextrins Inulin Boswellia gum Khaya gum B R Nahata College of Pharmacy Mandsaur (M.P.)
- 22. Enteric-coated polysaccharide matrix B R Nahata College of Pharmacy Mandsaur (M.P.)
- 23. Compression Coated Tablets B R Nahata College of Pharmacy Mandsaur (M.P.)
- 24. Mixed Film Coated Tablets B R Nahata College of Pharmacy Mandsaur (M.P.)
- 25. TimEd rElEASE SySTEmS Releases the drug after a predetermined lag time The lag time usually starts after gastric emptying because most of the time- controlled formulations are enteric coated Drug release from these systems is not pH dependent B R Nahata College of Pharmacy Mandsaur (M.P.)
- 26. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 27. OSmOTiCAlly CONTrOllEd drug dElivEry SySTEmS Delivery port Rigid semi permeable membrane Osmotic agent layer Fluid to be pumped Depend up on the osmotic pressure exerted by Depend osmogen on drug compartment with which though drug get released slowly though the orifice B R Nahata College of Pharmacy Mandsaur (M.P.)
- 28. Pressure DePenDent release systems Relies on the relatively strong peristaltic waves in the colon that lead to an increased luminal pressure, in response to raised pressure of the colon the dosage form get ruptured and release the drug at desired site B R Nahata College of Pharmacy Mandsaur (M.P.)
- 29. Platform Technologies for CTDDS • PULSINCAP • OROS-CT • CODESTM • CHRONOTROPIC® SYSTEM B R Nahata College of Pharmacy Mandsaur (M.P.)
- 30. PULSINCAP B R Nahata College of Pharmacy Mandsaur (M.P.)
- 31. OrOS-CT B R Nahata College of Pharmacy Mandsaur (M.P.)
- 32. COdES Tm B R Nahata College of Pharmacy Mandsaur (M.P.)
- 33. ChrONOTrOPiC® SySTEm Enteric coat Drug containing core HPMC Coat B R Nahata College of Pharmacy Mandsaur (M.P.)
- 34. EvAluATiON Invitro models Invitro test for intactness of coating and carriers in simulated conditions of stomach and intestine step1 Drug release study in 0.1N HCL for 2 hours (mean gastric emptying) step 2 Drug release study in phosphate buffer for 3 hours (mean small intestine transit time) B R Nahata College of Pharmacy Mandsaur (M.P.)
- 35. In vitro enzymatic degradation test Method 1: Drug release in buffer medium containing enzymes(e.g.pectinase, dextranase) or rat or guinea pig or rabbit decal contents Amount of drug release in particular time directly proportional to the rate of degradation of polymer carrier. Method 2: Incubating carrier drug system in fermenter Suitable medium containing colonic bacteria (streptococcus faecium or B.ovatus) Amount of drug released at different time intervels determined . B R Nahata College of Pharmacy Mandsaur (M.P.)
- 36. Clinical evaluation of colon-specific drug delivery system • Gamma Scintigraphy • High-frequency capsule B R Nahata College of Pharmacy Mandsaur (M.P.)
- 37. Gamma Scintigraphy showing the spread of the tracer all along the ascending, transverse, descending and sigmoid colon B R Nahata College of Pharmacy Mandsaur (M.P.)
- 38. High-frequency capsule method: Therelative BA of CDDS can be evaluated by high frequency capsules smoth plastic capsule containing small latex bollon, drug and radiotracer taken orally Triggering system (high frequency generator) Release of drug and radiotracer Triggered by an impules, the release is monitered In different parts of GIT by Radiological localization B R Nahata College of Pharmacy Mandsaur (M.P.)
- 39. CONCluSiON The colonic region of the GIT has become an increasingly important site for drug delivery and absorption. CDDS offers therapeutic benefits to patients in both local and systemic treatment. Systems utilize natural materials that are degraded by colonic bacterial enzymes. Colon provides favorable factors and conditions for designing of delivery systems. High commercial viability. Increasing number of international patents and research work in this particular mode of drug delivery itself shows its potential for pharmaceutical market. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 40. S.P. Vyas, R.K. Khar ; controlled Drug Delivery N.K.JAIN; Progress in controlled and novel drug delivery systems. Vincent H.L. Lee and Suman k. Mukherjee ; Encyclopedia of pharmaceutical Technology. Edi 2007 Van den Mooter G. V., Kinget R, (1995) Oral colon- specific drug delivery: a review DrugDeliv, 2: 81-93. Sarasija S, Hota A. (2002) Colon-specific drug delivery systems. Ind J Pharm Sci. 62(1):1-8. Colon targeted drug delivery system: A Review on primary and novel approaches.Oman Medical J,volume 25, issue 2, april2010. B R Nahata College of Pharmacy Mandsaur (M.P.)
- 41. Colon-specific drug delivery system: IJPS,2000,62,1-8. Oral colon targeted delivery systems for treatment of IBD: synthesis,in vitro and invivo assessment-IJPharm 358(2008)248-255. Novel Pharmaceutical Approaches for Colon Drug Delivery: An overview- Journal of Pharmacy Research, july-sep 2008 B R Nahata College of Pharmacy Mandsaur (M.P.)
- 42. ThANK yOu B R Nahata College of Pharmacy Mandsaur (M.P.)
