Organic chemistry
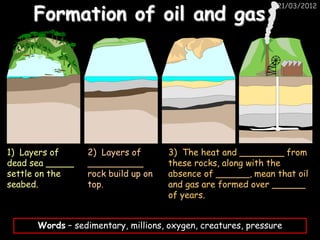
- 1. Formation of oil and gas 21/03/2012 1) Layers of 2) Layers of 3) The heat and ________ from dead sea _____ __________ these rocks, along with the settle on the rock build up on absence of ______, mean that oil seabed. top. and gas are formed over ______ of years. Words – sedimentary, millions, oxygen, creatures, pressure
- 2. Hydrocarbons and crude oil 21/03/2012 Crude oil is a mixture of HYDROCARBONS (compounds made up of carbon and hydrogen). Some examples: H H Longer chains mean… Increasing length H C C H H H 1. Less ability to flow Ethane 2. Less flammable H H H H H C C C C H 3. Less volatile H H H H 4. Higher boiling point Butane
- 3. Fractional distillation 21/03/2012 Crude oil can be separated by fractional distillation. The oil is evaporated and the hydrocarbon chains of different lengths condense at different temperatures: Fractions with low boiling points condense at the top Fractions with high boiling points condense at the bottom
- 4. Cracking 21/03/2012 Shorter chain hydrocarbons are in greater demand because they burn easier. They can be made from long chain hydrocarbons by “cracking”: Butane Ethane For example, this bond can be “cracked” to give two of these: Ethane
- 5. Cracking 21/03/2012 This is a THERMAL DECOMPOSITION reaction, with clay used as a catalyst Cracking is used to produce plastics such as polymers and polyethanes. The waste products from this reaction include carbon dioxide, sulphur dioxide and water vapour. There are three main environmental problems here: 1) Carbon dioxide causes the _________ effect 2) Sulphur dioxide causes _____ _____ 3) Plastics are not _____________
- 6. Alkanes 21/03/2012 Alkanes are SATURATED HYDROCARBONS. What does this mean? HYDROCARBONS are molecules that are made up of hydrogen and carbon atoms SATURATED means that all of these atoms are held together by single bonds, for example: Ethane Butane Alkanes are fairly unreactive (but they do burn well).
- 7. Alkenes 21/03/2012 Alkenes are different to alkanes; they contain DOUBLE COVALENT bonds. For example: ALKANES ALKENES Ethane Ethene Butane Butene This double bond means that alkenes have the potential to join with other molecules – this make them REACTIVE. We can test for alkenes because they turn bromine water colourless.
- 8. Monomers and Polymers 21/03/2012 Here’s ethene again. Ethene is called a MONOMER because it is just one small molecule. We can use ethene to make plastics… Ethene Step 1: Break the double bond Step 2: Add the molecules together: This molecule is called POLYETHENE, and the process that made it is called POLYMERISATION
- 9. Another way of drawing it… 21/03/2012 Instead of circles, let’s use letters… H H H H H H H H C C C C C C C C H H H H H H H H Ethene Ethene Poly(e)thene General formula for addition polymerisation: n C C C C n H CH3 H CH3 e.g. n C C C C H H H H n