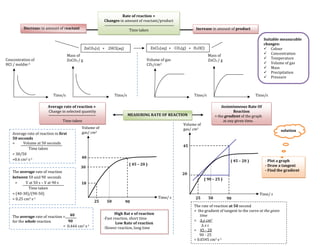
Rate of reaction
- 1. Rate of reaction = Changes in amount of reactant/product Decrease in amount of reactant Time taken Increase in amount of product Suitable measurable changes: ZnCO3(s) + 2HCl(aq) ZnCl2(aq) + CO2(g) + H2O(l) Colour Mass of Mass of Concentration Concentration of ZnCO3 / g Volume of gas ZnCl2 / g Temperature HCl / moldm-3 CO2/cm3 Volume of gas Mass Precipitation Pressure Time/s Time/s Time/s Time/s Average rate of reaction = Instantaneous Rate Of Change in selected quantity Reaction MEASURING RATE OF REACTION = the gradient of the graph Time taken at any given time. Volume of Volume of gas/ cm3 gas/ cm3 solution Average rate of reaction in first 50 seconds = Volume at 50 seconds 45 Time taken = 30/50 40 =0.6 cm3 s-1 ( 45 – 20 ) - Plot a graph ( 45 – 20 ) - Draw a tangent 30 The average rate of reaction - Find the gradient 20 between 50 and 90 seconds ( 90 – 25 ) = V at 50 s – V at 90 s 18 Time taken = (40-30)/(90-50) Time/ s = 0.25 cm3 s-1 Time/ s 25 50 90 25 50 90 The rate of reaction at 50 second = the gradient of tangent to the curve at the given High Rat e of reaction The average rate of reaction = 40 -Fast reaction, short time time for the whole reaction 90 = ∆ y cm3 Low Rate of reaction = 0.444 cm3 s-1 ∆xs -Slower reaction, long time = 45 - 20 90 - 25 = 0.0345 cm3 s-1
- 2. Factors affecting rate of reaction SIZE TEMPERATURE PRESSURE Reaction When pressure When total surface When temperature has stopped increase rate of area larger, rate of increases, rate of reaction increase reaction increase reaction increase CONCENTRATION CATALYST When concentration When positive of reactant increase catalyst are used. Gradient in reaction I steeper > Gradient rate of reaction rate of reaction Gradient at t1 steeper > Gradient at t2 in reaction II increase Rate of reaction t1 >Rate of reaction t2 Rate of reaction I >Rate of reaction II Volume of EFFECT OF THE SIZE OF REACTANT ON carbon dioxide/ cm3 RATE OF REACTION CO2 CO2 gas Gas Experiment I hydrochloric acid Aim : To investigate the effect of the size of reactant (small chip) on the rate of reaction Problem statement : How does the size of calcium carbonate chips affect the rate of its reaction with Experiment II calcium (large chip) dilute hydrochloric acid? carbonate Hypothesis : The rate of reaction between calsium Water carbonate and hydrochloric acid is increases when smaller size calcium carbonate used Time/s Experiment I: 20 cm3 of 0.5 mol dm-3 Manipulated variable : The size of calcium carbonate hydrochloric acid + excess of CaCO3 SMALL Responding variable : The rate of reaction • The rate of reaction in experiment II is higher than CHIPS Fixed variables : Volume and concentration of HCl experiment I because the gradient of the graph II is greater than graph I throughout the reaction. Experiment II: 20 cm3 of 0.5 mol dm-3 Observable Change: Volume of gas CO2 in every 30 s hydrochloric acid + excess of CaCO3 LARGE • The rate of reaction of the small calcium carbonate CHIPS chips is higher compared than large calcium The number of mole of HCl in both experiments: carbonate chip Equation: = MV/1000 2CaCO3 + 2HCl CaCl2 + H2O + CO2 = 22 x 0.5)/1000 • The maximum volume of carbon dioxide gas =0.01 mol collected for both experiments are equal because the no. of mole of hydrochloric acid are the same
- 3. CONCENTRATION Volume of carbon dioxide/ cm3 Eye Experiment I: 50 cm3 of 0.2 mol dm-3 sodium thiosulphate solution + 5 cm3 of 0.5 mol dm-3 Exp I (high concentration) hydrochloric acid Sodium thiosulphate Experiment is repeated four times using 0.2 mol dm-3 solution sodium thiosulphate solution diluted with different Exp II + Hydrochloric acid volume of distilled water (Low concentration) ‘X’ Equation: mark Na2S2O3 + 2HCl 2NaCl + S + SO2 + H2O Observable Change: Yellow precipitate formed. Experiment 1 2 3 4 5 - The rate of reaction in exp I is higher than exp II Aim : To investigate the effect concentration of sodium Volume of 0.2 - Exp I has higher concentration thiosulphate on the rate of reaction moldm-3 Na2S2O3 , 50 40 30 20 10 than Exp II V1 cm3 - Gradient I is steeper than Problem statement : How does concentration of Volume of distilled graph II sodium thiosulphate affect on the rate of reaction 0.0 10 20 30 40 water added/cm3 - The maximum volume of Volume of 1.0 mol carbon dioxide gas collected Hypothesis : When concentration of sodium 5.0 5.0 5.0 5.0 5.0 HCl acid added/cm3 for both experiments are thiosulphate increase, rate of reaction will increase. Concentration of equal 0.2 0.16 0.12 0.08 0.04 Na2S2O3/moldm-3 - no. of mole of hydrochloric Manipulated variable : concentration of sodium Time taken/s 20 23 32 46 95 acid are the same thiosulphate Responding variable : The rate of reaction 1/time , s-1 0.05 0.043 0.031 0.022 0.011 Fixed variables : Volume and concentration of HCl - Concentration of Na2S2O3 (mol dm-3) Concentration is directly Concentration of proportional to 1/time. Experiment 1: Na2S2O3 (mol dm-3) Concentration is inversely 2.0 g Magnesium + 50 cm3 of [ 1/time shows the rate of 2.0 mol dm-3 hydrochloric proportional to time. acid reaction ] When the concentration of When the concentration of Experiment II Na2S2O3 increases, a shorter 2.0 g Magnesium + 50 cm3 of Na2S2O3 increases, the rate of 1.0 mol dm-3 hydrochloric time is needed for marked reaction is increase acid across to disappear. Ionic Equation: 1/time (s-1) S2O3 2- + 2H+ S + SO2 + H2O Time /s
- 4. Equation: Ionic Equation: Experiment 1: Na2S2O3 + 2HCl 2NaCl + S + SO2 + H2O S2O3 2- + 2H+ S + SO2 + H2O 2.0 g Magnesium + 50 cm3 of 1.0 mol dm-3 hydrochloric acid at 25 oC CONCENTRATION TEMPERATURE Eye Experiment II Eye 2.0 g Magnesium + 50 cm3 of Observable changes: 1.0 mol dm-3 hydrochloric acid at 60 oC Time required for mark Sodium thiosulphate ‘X’ disappear from view. Sodium thiosulphate solution solution Volume of H2 + Hydrochloric acid + Hydrochloric acid / cm3 Experiment is repeated four times using 0.2 ‘X’ ‘X’ mol dm-3 sodium thiosulphate solution diluted mark Exp II (60 oC) mark Paper with different volume of distilled water Paper sheet sheet Exp I Concentration of (25 oC) Na2S2O3 (mol dm-3) Experiment 1 2 3 4 Temperature Na2S2O3 (mol dm-3) Temperature/oC 30 40 50 60 Volume of 0.2 50 40 30 20 When temperature When the concentration moldm-3 Na2S2O3 , Time /s increase, Shorter time increase, Shorter time Volume of distilled is needed for mark ‘X’ 0.0 10 20 30 is needed for mark ‘X’ lower gradient water added/cm3 disappear. disappear. :. Lower rate Volume of 1.0 mol 5.0 5.0 5.0 5.0 HCl acid added/cm3 Concentration of Steeper gradient 0.2 0.16 0.12 0.08 :. Higher rate Na2S2O3/moldm-3 Time taken/s 20 23 32 46 Volume of H2 1/time , s-1 0.05 0.043 0.031 0.022 / cm3 Time /s Time /s Exp I Concentration of (high concentration) Na2S2O3 (mol dm-3) Temperature concentration of Na2S2O3 increase Na2S2O3 (mol dm-3) Temperature of Na2S2O3 increase the rate of reaction increase the rate of reaction increase Exp II (low concentration) Time /s Experiment 1: 2.0 g Magnesium + 50 cm3 of 1.0 mol dm-3 hydrochloric acid 1/time (s-1) 1/time (s-1) Experiment II 2.0 g Magnesium + 50 cm3 of 1.0 mol dm-3 Shows the rate of reaction sulphuric acid
- 5. Properties of catalyst Decomposition Need a small amount H2O2 2 H2O + O2 Specific in action Chemically unchanged AMOUNT OF CATALYST Does not affect amount PRESENCE OF CATALYST product Increase rate of Problem statement : How does the amount reaction Problem statement : How does the presence of of catalyst affect the rate of composition of Observable changes: catalyst affect the rate of composition of hydrogen peroxide solution? The presence of oxygen gas, tested with glowing hydrogen peroxide solution? Hypothesis : When amount of catalyst used Observable changes: wooden splinter Hypothesis : Presence of catalyst increase the increase, the rate of decomposition of Volume of gas carbon rate of decomposition of hydrogen peroxide hydrogen peroxide increase dioxide in every 30 s is recorded Experiment 1: Manipulated variable : Mass of catalyst Decomposition of 50 Manipulated variable : Presence of catalyst Responding variable : The rate of reaction Responding variable : The rate of reaction cm3 of 1.0 mol dm-3 Fixed variables : temperature, volume and Hydrogen Peroxide Fixed variables : temperature, volume and concentration of hydrogen peroxide concentration of hydrogen peroxide Experiment 1: Experiment II Decomposition of 50 Decomposition of 50 cm3 of 1.0 mol dm-3 cm3 of 1.0 mol dm-3 Hydrogen Peroxide + Hydrogen Peroxide + 0.5 g manganese (IV) 1.0 g manganese (IV) oxide oxide Experiment II Decomposition of 50 Volume of O2 cm3 of 1.0 mol dm-3 / cm3 Hydrogen Peroxide + 1.0 g manganese (IV) Exp II Volume of O2 oxide (with catalyst) / cm3 Exp II Exp I (1.0 g MnO2) When amount Manganese(IV) (without oxide increase , rate of reaction catalyst) increase Exp I Total volume for both exp I and II (0.5 g MnO2) Time /s same Because the molarity and volume Lower gradient of hydrogen peroxide in both :. Lower rate Manganese(IV) oxide act as catalyst reaction are same Time /s to increase rate of reaction Quantity of catalyst does not affect Total volume for both exp I and II same Lower gradient the total volume of produced Steeper gradient Because the molarity and volume of :. Lower rate :. Higher rate hydrogen peroxide in both reaction are same Steeper gradient :. Higher rate
- 6. The Collision Theory Endothermic reaction Energy Energy Achieved a Ea Right Exothermic Molecule ust minimun Ea collide orientation of amoun of reaction collision energy (Ea) Product ” reactants Ea . Ea ’ products The collisions that lead to a chemical reaction are known as effective collisions Reactant Reactant Progress of reaction Progress of reaction Explanation using Collision Theory Energy Profile Diagram And Activation Energy, Ea’: Ea – The minimum energy the reactant SIZE CONCENTRATION TEMPERATUR TEMPERATURE CATALYST Ea’ – The lower activation energy in the presence of a catalyst. The higher the The higher the Catalyst provides The smaller the size concentration of temperature, the an alternative path Haber Process (NH3) of reactant, the reactants, the higher is the kinetic of reaction which Iron, Fe larger is the total higher is the energy of reacting needs lower surface area number of particles particles. The activation energy exposed to collision in a unit volume. reacting particles (Ea’) Uses of Ostwald process (HNO3) move faster. Catalyst in Platinum, Pt Industrial Contact process (H2SO4) Vanadium (V) oxide, The frequency of collision between particles increases. V2O5 The frequency of effective collision between particles increases Cooking of solid food in smaller size The total surface area on a smaller cut pieces of food is larger The food can absorbed more heat. The rate of reaction increases. The time taken for the food to be cooked is shorter me Cooking in a pressure cooker The high pressure in pressure cooker increases the boiling Storage of food in a refrigerator point of water to a temperature above 100 °C. When the food kept in refrigerator, the food lasts longer t The kinetic energy of the particles in the food is higher higher. The low temperature in the refrigerator slows down the Time taken for the food to be cooked is decrease activity of the bacteria. Thus the food cooked faster at a higher temperature in a The bacteria produce less toxin , pressure cooker. the rate of decomposition of food becomes lower
- 7. FACTOR EXPLANATION DIAGRAM Volume of Size H2/ cm3 Size of zinc in exp. II is smaller than exp I. Total surface area exposed to collision in exp. Exp I: II is larger than exp. I 2 g of Zinc chip + 50 cm3 1.0 The frequency of collision between zinc and mol dm-3 HCl hydrogen ion in exp II is higher Exp II Frequency of effective collision between zinc Exp II : and hydrogen ion in exp II is higher 2 g of Zinc powder + 50 cm3 1.0 Rate of reaction in exp. II is higher Exp I mol dm-3 HCl Time/s Concentration Volume of H2 Concentration of hydrochloric acid in exp. II is / cm3 higher than exp I Exp I: 2 g of Zinc powder + 50 The number particles per unit volume in exp. Exp II cm3 0.5 mol dm-3 HCl II is higher than exp. I Exp II : 2 g of Zinc powder + 50 The frequency of collision between zinc and cm3 1.0 mol dm-3 HCl hydrogen ion in exp II is higher Exp II Frequency of effective collision between zinc and hydrogen ion in exp II is higher Rate of reaction in exp. II is higher Time /s Experiment I and II Exp I use ethanoic acid (weak acid) and exp II use hydrochloric acid (strong acid) The number of hydrogen ions per unit volume Volume of H2 in exp. II is higher than exp. I / cm3 Concentration The frequency of collision between zinc and hydrogen ion in exp II is higher Frequency of effective collision between zinc Exp III Exp I: 2 g of Zinc powder + 50 and hydrogen ion in exp II is higher cm3 1.0 mol dm-3 CH3COOH Rate of reaction in exp. II is higher Exp II Exp II : 2 g of Zinc powder + 50 cm3 1.0 mol dm-3 HCl Exp III : 2 g of Zinc powder + 50 Experiment II and III Exp I cm3 1.0 mol dm-3 H2SO4 Exp III use sulphuric acid (diprotic acid) and exp II use hydrochloric acid (monoprotic acid) The number of hydrogen ions per unit volume Time /s in exp. III is higher than exp. II The frequency of collision between zinc and hydrogen ion in exp II is higher Frequency of effective collision between zinc and hydrogen ion in exp II is higher Rate of reaction in exp. II is higher
- 8. Volume of Temperature carbon dioxide/ cm3 Temperature of exp. II is higher than exp I. Exp I: The kinetic energy of reactant in exp II is higher 2 g of Zinc chip + 50 cm3 1.0 than I mol dm-3 HCl at 25 oC The frequency of collision between zinc and Exp II hydrogen ion in exp II is higher Exp II : Frequency of effective collision between zinc 2 g of Zinc powder + 50 cm3 1.0 and hydrogen ion in exp II is higher mol dm-3 HCl at 40 oC Rate of reaction in exp. II is higher Exp I Time/s Catalyst Volume of H2 Exp II use copper (II) sulphate act as catalyst / cm3 Exp I: Catalyst provides an alternative path of reaction Exp II 2 g of Zinc powder + 50 cm3 0.5 which needs lolower activation energy (Ea’) (with catalyst) mol dm-3 HCl The frequency of collision between zinc and hydrogen ion in exp II is higher Exp II : Frequency of effective collision between zinc Exp I 2 g of Zinc powder + 50 cm3 1.0 and hydrogen ion in exp II is higher mol dm-3 HCl and 2cm3 of Rate of reaction in exp. II is higher copper (II) sulphate Time /s HAK MILIK SLM 2011
