Exothermic and Endothermic Reactions Explained
•
3 recomendaciones•3,212 vistas
interpret energy level diagram for exothermic & endothermic reaction
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
Thermodynamic Principal #chemical engineering microproject 

Thermodynamic Principal #chemical engineering microproject
Similar a Exothermic and Endothermic Reactions Explained
Similar a Exothermic and Endothermic Reactions Explained (20)
Más de sweemoi khor
Más de sweemoi khor (20)
Question Analysis for Paper 3 Trial Chemistry 2012 from Different States

Question Analysis for Paper 3 Trial Chemistry 2012 from Different States
Operational Definition of Reactivity of Alkali Metals with oxygen

Operational Definition of Reactivity of Alkali Metals with oxygen
Question Analysis for Paper 3 Trial Chemistry 2011 from Different States

Question Analysis for Paper 3 Trial Chemistry 2011 from Different States
Exothermic and Endothermic Reactions Explained
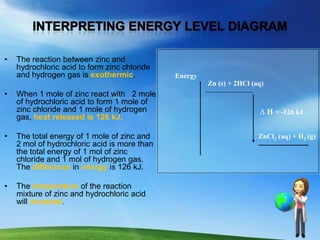
- 1. • The reaction between zinc and hydrochloric acid to form zinc chloride and hydrogen gas is exothermic. Energy Zn (s) + 2HCl (aq) • When 1 mole of zinc react with 2 mole of hydrochloric acid to form 1 mole of zinc chloride and 1 mole of hydrogen ∆ H = -126 kJ gas, heat released is 126 kJ. • The total energy of 1 mole of zinc and ZnCl2 (aq) + H2 (g) 2 mol of hydrochloric acid is more than the total energy of 1 mol of zinc chloride and 1 mol of hydrogen gas. The difference in energy is 126 kJ. • The temperature of the reaction mixture of zinc and hydrochloric acid will increase.
- 2. • The reaction between nitrogen and oxygen to form nitrogen dioxide is endothermic. Energy 2NO2 (g) • When 1 mole of nitrogen react with 2 mole of oxygen to form 2 mole of nitrogen dioxide, heat ∆ H = +66 kJ absorbed is 66 kJ. • The total energy of 2 mole of N2 (g) + 2O2 (g) nitrogen dioxide is more than the total energy of 1 mol of nitrogen chloride and 2 mol of oxygen. The difference in energy is 66kJ. • The temperature of the reaction mixture of zinc and hydrochloric acid will decrease.