IB Chemistry on Quantum Numbers, Electronic Configuration and De Broglie Wavelength
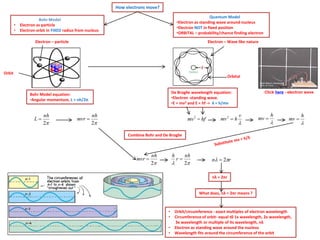
- 1. How electrons move? • • Quantum Model •Electron as standing wave around nucleus •Electron NOT in fixed position •ORBITAL – probability/chance finding electron Bohr Model Electron as particle Electron orbit in FIXED radius from nucleus Electron – particle Electron – Wave like nature Orbit Orbital De Broglie wavelength equation: •Electron -standing wave. •E = mv2 and E = hf -> λ = h/mv Bohr Model equation: •Angular momentum, L = nh/2π L nh 2 mvr nh 2 mv 2 = hf mv 2 h Click here - electron wave v mv h mv Combine Bohr and De Broglie mvr nh 2 h r nh 2 n 2r nλ = 2πr What does, nλ = 2πr means ? • • • • Orbit/circumference - exact multiples of electron wavelength Circumference of orbit- equal t0 1x wavelength, 2x wavelength, 3x wavelength or multiple of its wavelength, nλ Electron as standing wave around the nucleus Wavelength fits around the circumference of the orbit h
- 2. Electron Wavelength around orbit • • • • Electron acts as standing wave surrounding the nucleus Wavelength fits around the circumference of the orbit Orbit/circumference - exact multiples of electron wavelength Circumference of orbit- equal the wavelength, 2x wavelength, 3x wavelength or multiple of its wavelength, nλ nλ = 2πr n=1 1λ = 2πr1 ONE wavelength λ fits the 1st orbit n=2 2λ = 2πr2 TWO wavelength λ fits the 2nd orbit n=3 3λ = 2πr3 THREE wavelength λ fits the 3rd orbit Standing wave around the circumference/circle 1λ ONE wavelength λ fits the 1st orbit 1st Orbit 2λ TWO wavelength λ fits the 2nd orbit 3λ 2nd Orbit THREE wavelength λ fits the 3rd orbit 3rd Orbit Relationship between wavelength and circumference a o = 0.0529nm/Bohr radius 1λ n=1 n=2 ONE wavelength λ TWO wavelength λ r n = n2 a 0 1λ1 = 2πr1 2λ 2λ2 = 2πr2 λ1 = 6.3 ao - 1st orbit r n = n2 a 0 λ2= 12.6 ao - 2nd orbit 3λ n=3 THREE wavelength λ 3λ3 = 2πr3 r n = n2 a 0 λ3 = 18.9 ao - 3rd orbit
- 3. Electron Wavelength around orbit • • • • Electron acts as standing wave around the nucleus Wavelength fits around circumference of orbit Orbit/circumference - exact multiples of electron wavelength Circumference of orbit- equal t0 1x wavelength, 2x wavelength, 3x wavelength or multiple of its wavelength, nλ nλ = 2πr ONE wavelength λ fits the 1st orbit n=1 λ = 2πr1 n=2 2λ = 2πr2 TWO wavelength λ fits the 2nd orbit n=3 3λ = 2πr3 THREE wavelength λ fits the 3rd orbit Standing wave around the circumference /circle λ ONE wavelength λ fits the 1st orbit 1st Orbit λ TWO wavelength λ fits the 2nd orbit λ 2nd Orbit THREE wavelength λ fits the 3rd orbit Click here to view video Click here to view notes Click here - electron wave simulation
- 4. Models for electronic orbitals 1927 1925 1913 Bohr Model De Broglie wavelength Electron in fixed orbits Heisenberg Uncertainty principle • Electron form a standing wave Impossible to determine both the position and velocity of electron at the same time. • Applies to electron, small and moving fast.. If we know position, x very precisely – we don’t know its momentum, velocity Δp electron Δx Big hole electron Δx electron Δx Probability/chance/likelyhood to find electron in space ORBITAL is used to replace orbit Small hole Reduce the hole smaller, x Know precisely x, electron position Uncertainty Δx is small ( Δx, Δp) Δp is high so Δx Δp > h/2 Δp high – uncertainty in its velocity is high Position of electron is unknown! Δp = mass x velocity Velocity is unknown Δx = uncertainty in position Δp = uncertainty in momentum/velocity (ħ)= reduced plank constant Probability/likelyhood to find an electron in space
- 5. Uncertainty for electron in space Bohr Model Electron in fixed orbits 1927 1925 1913 De Broglie wavelength Electron form a standing wave Heisenberg Uncertainty principle • Impossible to determine both the position and velocity of electron at the same time. • Applies to electron, small and moving fast.. If we know position, x very precisely – we don’t know its momentum, velocity Probability/chance/likelyhood to find an electron ORBITAL is used to replace orbit Excellent video on uncertainty principle Click here video on uncertainty principle Video on uncertainty principle Click here to view uncertainty principle Δx = uncertainty in position Δp = uncertainty in momentum/velocity (ħ)= reduced plank constant
- 6. Schrödinger's wave function. 1927 Schrödinger's wave function. •Mathematical description of electron given by wave function •Amplitude – probability of finding electron at any point in space/time High probability finding electron electron density • • • Bohr Model ✗ • • • Probability finding electron in space Position electron unknown ✗ is used Orbital ✔ NOT orbit Schrödinger's wave function. Probability find electron distance from nucleus Probability density used- Ψ2 Orbital NOT orbit is used ✔ ORBITAL is used to replace orbit ORBITAL•Mathematical description wavelike nature electron •Wavefunction symbol – Ψ •Probability finding electron in space better description electron behave Click here to view simulation Click here to view simulation Click here to view simulation
- 7. Four Quantum Numbers • • • Electrons arrange in specific energy level and sublevels Orbitals of electrons in atom differ in size, shape and orientation. Allow states call orbitals, given by four quantum number 'n', 'l', 'm l' and ’ms’ - (n, l, ml, ms) 1 Principal Quantum Number (n): n = 1, 2, 3,.. ∞ •Energy of electron and size of orbital/shell •Distance from nucleus, (higher n – higher energy) •Larger n - farther e from nucleus – larger size orbital • n=1, 1stprincipal shell ( innermost/ground shell state) 2 Angular Momentum Quantum Number (l): l = 0 to n-1. •Orbital Shape •Divides shells into subshells/sublevels. •Letters (s, d, p, f) s orbital p orbital No TWO electron have same 4 quantum number 3 4 Magnetic Quantum Number (ml): ml = -l, 0, +l. •Orientation orbital in space/direction •mℓ range from −ℓ to ℓ, •ℓ = 0 -> mℓ = 0 –> s sublevel -> 1 orbital •ℓ = 1 -> mℓ = -1, 0, +1 -> p sublevel -> 3 diff p orbitals •ℓ = 2 -> mℓ = -2, -1, 0, +1, +2 -> d sublevel -> 5 diff d orbitals •(2l+ 1 ) quantum number for each ℓ value Spin Quantum Number (ms): ms = +1/2 or -1/2 •Each orbital – 2 electrons, spin up/down •Pair electron spin opposite direction •One spin up, ms = +1/2 •One spin down, ms = -1/2 •No net spin/cancel out each other– diamagnetic electron writing electron spin electron spin up/down d orbital
- 8. Principal and Angular Momentum Quantum numbers • • • Electrons arrange in specific energy level and sublevels Orbitals of electrons in atom differ in size, shape and orientation. Allow states call orbitals, given by four quantum number 'n', 'l', 'm l' and ’ms’ - (n, l, ml, ms) 1 Principal Quantum Number (n): n = 1, 2, 3, …, ∞ •Energy of electron and size of orbital /shell •Distance from nucleus, (higher n – higher energy) •Larger n - farther e from nucleus – larger size orbital • n=1, 1stprincipal shell ( innermost/ground shell state) 2 Angular Momentum Quantum Number (l): l = 0, ..., n-1. •Orbital Shape •Divides shells into subshells (sublevels) •Letters (s,p,d,f) •< less than n-1 Sublevels, l Quantum number, n and l l=1 2p sublevel l=0 2s sublevel n= 2 n= 1 1 Principal Quantum #, n (Size , energy) l=0 2 1s sublevel Angular momentum quantum number, l (Shape of orbital) 2p sublevel – contain 2p orbital 2nd energy level Has TWO sublevels 2s sublevel – contain 2s orbital 1st energy level Has ONE sublevel 1s sublevel – contain 1s orbital 1 Principal Quantum Number (n) 2 Angular Momentum Quantum Number (l)
- 9. Electronic Orbitals Simulation Electronic Orbitals n = 1, 2, 3,…. Allowed values l = 0 to n-1 Allowed values ml = -l, 0, +l- (2l+ 1 ) for each ℓ value ml =+2 ml =+1 l=2 3d sublevel ml = 0 Energy Level ml =-1 ml =-2 ml =+1 n= 3 l=1 3p sublevel ml = 0 ml =-1 l=0 3s sublevel ml = 0 ml =+1 l=1 ml = 0 2p sublevel ml =-1 n= 2 l=0 1 Principal Quantum #, n (Size , energy) 2 ml =0 l=0 n= 1 2s sublevel 1s sublevel ml =0 Angular momentum quantum number, l (Shape of orbital) 3 3dx2 – y2 orbital 3dyz orbital 3dz2 orbital 3dxz orbital 3dxy orbital Click here to view simulation 3pz orbital 3py orbital 3px orbital 3s orbital 2py orbital 2pz orbital Click here to view simulation 2px orbital 2s orbital 1s orbital Magnetic Quantum Number (ml) (Orientation orbital) Click here to view simulation
- 10. Quantum Numbers and Electronic Orbitals ml =+2 3dx2 – y2orbital Simulation Electronic Orbitals Energy Level ml =+1 3d sublevel ml = 0 3dz2 orbital ml =-1 l=2 3dyz orbital 3dxz orbital Click here to view simulation n= 3 ml =-2 3dxy orbital ml =+1 3p sublevel ml = 0 3pz orbital ml =-1 l=1 3py orbital 3px orbital Click here to view simulation l=0 2p sublevel n= 2 ml = 0 3s orbital ml =+1 l=1 3s sublevel 2py orbital ml = 0 2pz orbital ml =-1 2px orbital l=0 n= 1 2s sublevel ml =0 2s orbital l=0 1s sublevel ml =0 1s orbital Click here to view simulation
- 11. Concept Map No TWO electron have same 4 quantum number Quantum number Quantum number = genetic code for electron What are these 4 numbers? (1, 0, 0, +1/2) or (3, 1, 1, +1/2) 4 numbers n l ml ms Size/distance Shape Orientation Electron has special number codes Electron spin Number + letter 1 Electron with quantum number given below (n,l,ml,,ms) – (1, 0, 0, +1/2) (n,l,ml,,ms) – (3, 1, 1, +1/2) 2 1s orbital 3py orbital What values of l, ml, allow for n = 3? How many orbitals exists for n=3? Video on Quantum numbers For n=3 -> l = n -1 =2 -> ml = -l, 0, +l -> -2, -1, 0, +1, +2 •mℓ range from −ℓ to ℓ, •ℓ = 0 -> mℓ = 0 –> s sublevel -> 1 orbital •ℓ = 1 -> mℓ = -1, 0, +1 -> p sublevel -> 3 diff p orbitals •ℓ = 2 -> mℓ = -2, -1, 0, +1, +2 -> d sublevel -> 5 diff d orbitals •(2l+ 1 ) quantum number for each ℓ value Answer = nine ml values – 9 orbitals/ total # orbitals = n 2 Click here video on quantum number Click here video on quantum number
