Case record...Dilated Virchow-Robin spaces associated with leukoaraiosis
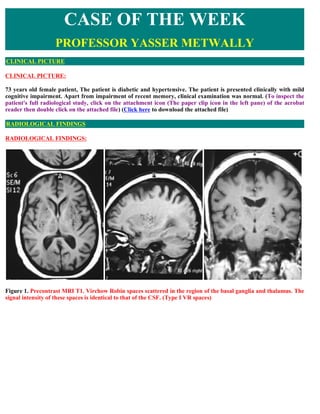
- 1. CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE: 73 years old female patient, The patient is diabetic and hypertensive. The patient is presented clinically with mild cognitive impairment. Apart from impairment of recent memory, clinical examination was normal. (To inspect the patient's full radiological study, click on the attachment icon (The paper clip icon in the left pane) of the acrobat reader then double click on the attached file) (Click here to download the attached file) RADIOLOGICAL FINDINGS RADIOLOGICAL FINDINGS: Figure 1. Precontrast MRI T1. Virchow Robin spaces scattered in the region of the basal ganglia and thalamus. The signal intensity of these spaces is identical to that of the CSF. (Type I VR spaces)
- 2. Figure 2. Precontrast MRI T2. Virchow Robin spaces scattered in the region of the basal ganglia and thalamus. The signal intensity of these spaces is identical to that of the CSF. Also notice the periventricular white matter changes (Leukoaraiosis) (Type I VR spaces) Figure 3. Precontrast MRI FLAIR images. Virchow Robin spaces scattered in the region of the basal ganglia and thalamus. The signal intensity of these spaces is identical to that of the CSF. Also notice the periventricular white matter changes (Leukoaraiosis) (Type I VR spaces)
- 3. Figure 4. MRI T2 images showing Vircho Robin spaces scsttered in the region of the midbrain. (Type III VR spaces) Virchow-Robin (VR) spaces surround the walls of vessels as they course from the subarachnoid space through the brain parenchyma. Small VR spaces appear in all age groups. With advancing age, VR spaces are found with increasing frequency and larger apparent sizes. At visual analysis, the signal intensity of VR spaces is identical to that of cerebrospinal fluid with all magnetic resonance imaging sequences. Dilated VR spaces typically occur in three characteristic locations: Type I VR spaces appear along the lenticulostriate arteries entering the basal ganglia through the anterior perforated substance. Type II VR spaces are found along the paths of the perforating medullary arteries as they enter the cortical gray matter over the high convexities and extend into the white matter. Type III VR spaces appear in the midbrain. Occasionally, VR spaces have an atypical appearance. They may become very large, predominantly involve one hemisphere, assume bizarre configurations, and even cause mass effect. Knowledge of the signal intensity characteristics and locations of VR spaces helps differentiate them from various pathologic conditions, including lacunar infarctions, cystic periventricular leukomalacia, multiple sclerosis, cryptococcosis, mucopolysaccharidoses, cystic neoplasms, neurocysticercosis, arachnoid cysts, and neuroepithelial cysts. Type I VR spaces Appear along the lenticulostriate arteries entering the basal ganglia through the anterior perforated substance. Type II VR spaces Are found along the paths of the perforating medullary arteries as they enter the cortical gray matter over the high convexities and extend into the white matter. Type III VR spaces Appear in the midbrain. DIAGNOSIS: DIAGNOSIS: ENLARGED VIRCHOW-ROBIN SPACES (PERIVASCULAR SPACES) ASSOCIATED WITH MICROVASCULAR BRAIN DISEASE (LEUKOARAIOSIS) DISCUSSION DISCUSSION: The Virchow-Robin (VR) space is named after Rudolf Virchow (German pathologist, 1821–1902) (1) and Charles Philippe Robin (French anatomist, 1821–1885) (2). VR spaces, or perivascular spaces, surround the walls of vessels as they course from the subarachnoid space through the brain parenchyma. VR spaces are commonly seen at magnetic resonance (MR) imaging and may sometimes be difficult to differentiate from pathologic conditions.
- 4. Knowledge of their signal intensity characteristics and localization helps in this differentiation, which is important for correct patient management. Anatomy VR spaces surround the walls of arteries, arterioles, veins, and venules as they course from the subarachnoid space through the brain parenchyma (Fig 1) (1–5). Electron microscopy and tracer studies have given insight into the location of VR spaces and clarified that the subarachnoid space does not communicate directly with the VR spaces (3–5). Figure 1. Photomicrograph (original magnification, x20; hematoxylin- eosin stain) of a coronal section through the anterior perforated substance shows two arteries (straight arrows) with surrounding VR spaces (curved arrows). Arteries in the cerebral cortex are coated by a layer of leptomeninges that is subtended from the pia mater; by this anatomic arrangement, the VR spaces of the intracortical arteries are in direct continuity with the VR spaces around arteries in the subarachnoid space (Fig 2). The lack of a similar coating of leptomeningeal cells around veins in the cerebral cortex suggests that VR spaces around veins are in continuity with the subpial space (4). Figure 2. Drawing shows a cortical artery with a surrounding VR space crossing from the subarachnoid and subpial spaces through the brain parenchyma. The magnified view on the right shows the anatomic relationship between the artery, VR space, subpial space, and brain parenchyma. In contrast to arteries in the cerebral cortex, arteries in the basal ganglia are surrounded by not one but two distinct coats of leptomeninges, separated by a VR space that is continuous with the VR space around arteries in the subarachnoid space. The inner layer of leptomeninges closely invests the adventitia of the vessel wall. The outer layer abuts on the glia limitans of the underlying brain and is continuous with the pia mater on the surface of the brain and the anterior perforated substance. Veins in the basal ganglia have no outer layer of leptomeninges (similar to cortical veins), which suggests that their VR spaces are continuous with the subpial space (5). Interstitial fluid within the brain parenchyma drains from the gray matter of the brain by diffusion through the extracellular spaces and by bulk flow along VR spaces. There is evidence from tracer studies and from pathologic
- 5. analysis of the human brain that VR spaces carry solutes from the brain and are, in effect, the lymphatic drainage pathways of the brain (6). Dilated VR Spaces Dilatation of VR spaces was described by Durant-Fardel (7) in 1843. These dilatations are regular cavities that always contain a patent artery. The mechanisms underlying expanding VR spaces are still unknown. Different theories have been postulated: segmental necrotizing angiitis of the arteries or another unknown condition causing permeability of the arterial wall (8–10), expanding VR spaces resulting from disturbance of the drainage route of interstitial fluid due to cerebrospinal fluid (CSF) circulation in the cistern (11,12), spiral elongation of blood vessels and brain atrophy resulting in an extensive network of tunnels filled with extracellular water (9,13), gradual leaking of the interstitial fluid from the intracellular compartment to the pial space around the metarteriole through the fenestrae in the brain parenchyma (14), and fibrosis and obstruction of VR spaces along the length of arteries and consequent impedance of fluid flow (5). Prevalence Small VR spaces (<2 mm) appear in all age groups. With advancing age, VR spaces are found with increasing frequency and larger apparent size (>2 mm) (15). Some studies found a correlation between dilated VR spaces and neuropsychiatric disorders (16–19), recent-onset multiple sclerosis (MS) (20), mild traumatic brain injury (21), and diseases associated with microvascular abnormalities (22). The prevalence of VR spaces at MR imaging is also dependent on the applied technique. Heavier T2-weighted imaging results in better visualization of VR spaces (23). In addition, the use of thinner sections will demonstrate more VR spaces as well (15,24). Also, high-field-strength MR imaging is expected to have an increased clinical impact in the near future; the current magnetic field (1.5 T) is likely to be switched to 3 or 4 T. The anticipated higher signal-to-noise ratio at higher magnetic field strengths may successfully improve spatial resolution and image contrast (25–27), leading to better visualization (and increased prevalence) of VR spaces on MR images. Appearance at MR Imaging Signal Intensity Characteristics Visually, the signal intensities of the VR spaces are identical to those of CSF with all pulse sequences. However, when signal intensities are measured, the VR spaces prove to have significantly lower signal intensity than the CSF- containing structures within and around the brain (28), a finding consistent with the fact that the VR spaces represent entrapments of interstitial fluid. This difference in signal intensity can also be explained by partial volume effects, since a VR space with accompanying vessel is smaller than the contemporary volume of a voxel on MR images. VR spaces show no restricted diffusion on diffusion-weighted images because they are communicating compartments. T1-weighted images with substantial flow sensitivity may show high signal intensity due to inflow effects, thereby helping confirm that one is indeed dealing with VR spaces (29). VR spaces do not enhance with contrast material. In patients with small to moderate dilatations of the VR spaces (2–5 mm), the surrounding brain parenchyma generally has normal signal intensity (30,31). Locations and Morphology Dilated VR spaces typically occur in three characteristic locations. The first type (type I) is frequently seen on MR images and appears along the lenticulostriate arteries entering the basal ganglia through the anterior perforated substance (Figs 3, 4) (15,32). Here, the tortuous lenticulostriate arteries change direction from a lateral to a dorsomedial path and are grouped closely together. A proximal VR space, containing several vessels, is the resulting physiologic finding (33).
- 6. Figure 3. Bilateral type I VR spaces in a 6-year-old boy. (a) Axial proton-density–weighted image (repetition time msec/echo time msec = 2375/100) shows hyperintense areas (arrows) in the anterior perforated substance on both sides. (b) Axial fluid-attenuated inversion-recovery (FLAIR) image (6606/100) obtained at the same level shows that these areas have CSF-like content (arrows). The signal intensity of the surrounding brain parenchyma is normal. (c, d) Diffusion-weighted image (2574/81; b factor = 1000 sec/mm2) (c) and corresponding apparent diffusion coefficient map (d) show no restricted diffusion in these areas (arrows).. Figure 4. Bilateral type I VR spaces in a 53-year-old woman. Coronal T1- weighted image (500/30) shows symmetrical hypointense areas (arrows) in the anterior perforated substance. The second type (type II) can be found along the path of the perforating medullary arteries as they enter the cortical gray matter over the high convexities and extend into the white matter (Figs 5, 6) (15,32).
- 7. Figure 5. Type II VR spaces in a 73-year-old woman. (a) Axial proton-density–weighted image (2376/100) shows multiple hyperintense foci in the centrum semiovale in both hemispheres. (b) On an axial FLAIR image (6614/100) obtained at the same level, the VR spaces are seen as hypointense dots without any surrounding high signal intensity. Note the two small lesions with a hypointense center and a hyperintense rim (arrows) in the left hemisphere; these lesions are not VR spaces but old lacunar infarctions. Figure 6. Type II dilated VR spaces in a 6-year-old boy. (a) Axial T2-weighted image (2620/100) shows linear to punctate hyperintense areas around the occipital horns, especially on the left side (arrow). (b) FLAIR image (7572/100) obtained at the same level shows no abnormal signal intensity (arrow), in accordance with the fact that these areas are true VR spaces.. The third type (type III) appears in the midbrain. In the lower midbrain, VR spaces at the pontomesencephalic junction surround the penetrating branches of the collicular and accessory collicular arteries (Figs 7, 8). They are mainly located between the cerebral peduncles in the axial plane and correspond to the level of the tentorial margin as seen in coronal sections. In the upper midbrain, where the VR spaces are visible at the mesencephalodiencephalic junction, they appear along the posterior (interpeduncular) thalamoperforating artery or the paramedian mesencephalothalamic artery and short and long circumferential arteries originating from the upper basilar artery or proximal posterior cerebral artery (23,34,35).
- 8. Figure 7. Type III VR space in a 25-year-old man. (a) Axial proton-density–weighted image (2620/100) shows a hyperintense spot in the brainstem (arrow). (b) Axial FLAIR image (7292/120) obtained at the same level shows that the spot has CSF-like content without abnormal surrounding signal intensity (arrow). These findings confirm that the spot is a VR space. Figure 8. Type III VR spaces in a 68-year-old man. (a) Axial proton-density–weighted image (2382/100) shows multiple punctate hyperintense areas in the brainstem (arrow). (b) Close-up T2-weighted image (4615/120) clearly shows the fine punctate pattern. (c) Axial FLAIR image (6609/100) shows the CSF-like content of the dots (arrow). No surrounding high signal intensity is seen. The typical configuration and the fact that no high signal intensity is seen on the FLAIR image confirm that the dots are VR spaces. VR spaces are mostly seen as well-defined oval, rounded, or tubular structures, depending on the plane in which they are intersected. They have smooth margins, commonly appear bilaterally, and usually measure 5 mm or less (32). Atypical VR Spaces It is reported that clusters of type II enlarged VR spaces may predominantly involve one hemisphere (36). There are even reports that describe the solely unilateral appearance of enlarged VR spaces in the high convexity (37,38). Occasionally, VR spaces appear markedly enlarged, cause mass effect, and assume bizarre cystic configurations that
- 9. may be misinterpreted as other pathologic processes, most often a cystic neoplasm. As most of these giant VR spaces border a ventricle or subarachnoid space, reports of such cases (39–41) have offered an extensive differential diagnosis that includes cystic neoplasms, parasitic cysts, cystic infarctions, nonneoplastic neuroepithelial cysts, and deposition disorders such as mucopolysaccharidosis. Salzman et al (42) presented a series of 37 patients with giant VR spaces. These spaces most often appear as clusters of variably sized cysts and are most common in the mesencephalothalamic region (Fig 9), in the territory of the paramedial mesencephalothalamic artery, and in the cerebral white matter. Giant VR spaces in the mesencephalothalamic region may cause hydrocephalus by direct compression of the third ventricle or the sylvian aqueduct (Fig 9), requiring surgical intervention (8,11,42–47). Figure 9. Giant VR spaces in the mesencephalothalamic region in a 19-year-old man. (a, b) Axial (a) and sagittal (b) T2-weighted images (5970/120) show a multicystic lesion in the mesencephalothalamic region. The lesion extends from the left cerebral peduncle to the left thalamus. The content of the cysts is CSF- like. The adjacent brain parenchyma has normal signal intensity. No solid components are identified. (c) Axial gadolinium-enhanced T1-weighted image (478/18) shows no enhancement. The process has caused obstruction of the sylvian aqueduct, resulting in hydrocephalus. The size of the lesion and the degree of hydrocephalus were unchanged compared with the appearance on MR images obtained 2 years earlier In one-half of cases, giant VR spaces that occur in the white matter may have surrounding signal intensity abnormality on T2-weighted or FLAIR images (42). This may be viewed as a worrisome finding and in some cases has prompted the performance of tissue biopsy. However, the abnormal signal intensity stems from reactive gliosis surrounding the enlarged VR spaces and is not an ominous finding (47). Dilated Virchow-Robin spaces as a marker of microvascular brain disease Virchow-Robin spaces (VRSs) are perivascular spaces that surround the perforating arteries that enter the brain. The spaces are normally microscopic, but when dilated, they may be seen on MR images. Even in the normal brain, some VRSs are usually seen in the area of the substantia innominata at the level of the anterior commissure, and a small number of dilated spaces may also be seen in the basal ganglia (BG) in up to 60% of individuals. Virchow- Robin Spaces can be identified by a combination of their typical location and their signal intensity characteristics. They are classically described as isointense to CSF on images obtained with all pulse sequences, and they are round
- 10. or linear depending on the imaging plane, although their characteristics may vary from this pattern for a number of reasons. First, the small size of the Virchow-Robin Spaces makes partial-volume effects common; therefore, measured signal intensities seldom equal those seen in pure CSF, although the changes in signal intensity between sequences are closely correlated. In addition, T1-weighted images with substantial flow sensitivity may show high signal intensity due to inflow effects. Even if we allow for these effects, the measured signal intensity in the VRS often slightly differs from that of true CSF. This finding has been attributed to the fact that Virchow-Robin Spaces around intracerebral arteries may represent interstitial fluid trapped in the subpial or interpial space. Pathologic dilatation of Virchow-Robin Spaces is most commonly associated with arteriolar abnormalities that arise due to aging, diabetes, hypercholesterolemia, smoking, and hypertension and other vascular risk factors. This dilatation forms part of a histologic spectrum of abnormalities, which include old, small infarcts (type 1 changes); scars from small hematomas (type 2 changes); and dilatations of Virchow-Robin Spaces (type 3 changes) (74). The presence of these abnormalities on histologic examination is believed to result from moderate-to-severe microangiopathy characterized by sclerosis, hyalinosis, and lipid deposits in the walls of small perforating arteries 50 – 400 `im in diameter (74, 75). As the severity of the microangiopathy increases, microvessels demonstrate increasingly severe changes, with arterial narrowing, microaneurysms and pseudoaneurysms, onion skinning, mural calcification, and thrombotic and fibrotic luminal occlusions (74–76) Although microvascular disease is common, few reliable surrogate imaging markers of its presence have been described. The extent and severity of deep white matter (WM) and periventricular hyperintensity on T2-weighted images have been widely studied as potential surrogate markers for small-vessel disease. However, the correlation between these abnormalities and clinical characteristics, such as diagnosis, vascular risk factor, or neuropsychological deficit, is often poor (77). Figure 10. MRI T2 (A), MRI FLAIR (B) and precontrast MRI T1 (C) images showing dilated Virchow-Robin Spaces associated with diffuse white matter changes (leukoaraiosis) More details about etiology and pathogenesis of dilatation of Virchow-Robin Spaces Virchow-Robin Spaces are potential perivascular spaces covered by pia that accompany arteries and arterioles as they perforate the brain substance. Deep in the brain, the Virchow-Robin Spaces are lined by the basement membrane of the glia limitans peripherally, while the outer surfaces of the blood vessels lie centrally. These pial layers form the Virchow-Robin Spaces as enclosed spaces filled with interstitial fluid and separated from the
- 11. surrounding brain and CSF . Dilatation of Virchow-Robin Spaces results in fluid filled perivascular spaces along the course of the penetrating arteries. Abnormal dilatation of Virchow-Robin Spaces is clinically associated with aging, dementia, incidental WM lesions, and hypertension and other vascular risk factors (73). Pathologically, this finding is most commonly associated with arteriosclerotic microvascular disease, which forms a spectrum of severity graded from 1 to 3 on the basis of histologic appearances (74, 76). Grade 1 changes include increased tortuosity and irregularity in small arteries and arterioles (74) Grade 2 changes include progress sclerosis, hyalinosis, lipid deposits, and regional loss of smooth muscle in the vessel wall associated with lacunar spaces that are histologically seen to consist of three subtypes. Type 1 lacunes are small, old cystic infarcts; type 2 are scars of old hematomas; and type 3 are dilated Virchow-Robin Spaces (79). Grade 3 microangiopathy represents the most severe stage and is especially related to severe chronic hypertension. Typical changes described in lower grades are accompanied by fibrotic thickening vessel wall with onion skinning, loss of muscularis and elastic lamina, and regional necrosis in the vessel walls. The brain parenchyma contains multiple lacunae, and diffuse abnormality of myelin is present in the deep hemispheric white matter. Several mechanisms for abnormal dilatation of Virchow-Robin Spaces have been suggested (80,81). These include mechanical trauma due to CSF pulsation or vascular ectasia (83), fluid exudation due to abnormalities of the vessel wall permeability (82), and ischemic injury to perivascular tissue causing a secondary ex vacuo effect (83). In the Western world, ischemic vascular dementia is seen in 8 –10% of cognitively impaired elderly subjects (84) and commonly associated with widespread small ischemic or vascular lesions throughout the brain, with predominant involvement of the basal ganglia, white matter, and hippocampus (84). Several groups have shown that a severe lacunar state and microinfarction due to arteriolosclerosis and hypertensive microangiopathy are more common in individuals with IVD than in healthy control subjects, and they have emphasized the importance of small vascular lesions in the development of dementia (84, 85). On CT or MR imaging, white matter lesions are commonly used as potential biomarkers of vascular abnormality. Many groups have suggested that simple scoring schemes for white matter lesion load and distribution are useful in the diagnosis of vascular dementia (86,87,88,89). Although white matter lesions are more severe in patients with vascular dementia (86), they are more prevalent in all groups with dementia than in healthy control subjects. Dilation of Virchow-Robin Spaces provides a potential alternative biomarker of microvascular disease (small vessel disease). Virchow-Robin Spaces in the centrum semiovale were significantly more frequent in patients with fronto- temporal dementia (FTD) than in control subjects (P .01). This finding is not associated with increases in basal ganglionic Virchow-Robin Spaces and is closely correlated with measures of forebrain atrophy, suggesting that these changes are probably representative of atrophy, which is more marked in this patient group than in those with other dementing conditions (78). SUMMARY SUMMARY Pathologic Findings Enlarged perivascular spaces, also known as Virchow-Robin spaces, are pial-lined interstitial fluid-filled structures that accompany penetrating arteries and veins. They do not communicate directly with the subarachnoid space. They are common, incidental, "leave me alone" lesions that should not be mistaken for more ominous disease. They frequently appear in the inferior basal ganglia, clustering around the anterior commissure and surrounding the
- 12. lenticulostriate arteries as they superiorly course through the anterior perforated substance. Other common locations include the midbrain, deep white matter, and subinsular cortex. They can also be found in the region of the thalami, dentate nuclei, corpus callosum, and cingulate gyrus . Microscopically, perivascular spaces consist of a single or double layer of invaginated pia. They are typically very small or inapparent as they pass through the cortex, enlarging in the subcortical white matter. They are typically not associated with gliosis in the surrounding parenchyma. Imaging Prominent perivascular spaces are considered a normal variant. Most appear as smoothly demarcated fluid-filled cysts, typically less than 5 mm in diameter, and often occur in clusters in the basal ganglia or midbrain. They are isointense to CSF at all sequences, including FLAIR. Most show normal signal intensity in the adjacent brain; 25% may have a small rim of slightly increased signal intensity. They do not enhance, cause focal mass effect, or restrict on diffusion-weighted images. In older patients, basal ganglia perivascular spaces sometimes become prominent and sievelike, a condition known as état criblé, or cribriform state. Occasionally perivascular spaces may become very large and appear bizarre. They are probably caused by the accumulation of interstitial fluid between the penetrating vessels and the pia. If interstitial fluid egress is blocked, fluid accumulates and the perivascular spaces dilate . These lesions cause focal mass effect and occasionally even hydrocephalus. Rarely, so-called giant or tumefactive perivascular spaces may be mistaken for more ominous disease. Differential Diagnosis Enlarged perivascular spaces are often mistaken for multiple lacunar infarcts, cystic neoplasms, and infectious cysts. Lacunar infarcts can usually be distinguished from perivascular spaces since many exhibit adjacent parenchymal hyperintensity (so-called état lacunaire). Cystic neoplasms rarely exhibit signal intensity exactly like the CSF. Neurocysticercosis cysts may have a scolex (parasite head), and the cyst walls often enhance. Neurocysticercosis cysts may be multiple but do not typically occur in clusters within the brain parenchyma. Addendum A new version of this PDF file (with a new case) is uploaded in my web site every week (every Saturday and remains available till Friday.) To download the current version follow the link "http://pdf.yassermetwally.com/case.pdf". You can also download the current version from my web site at "http://yassermetwally.com". To download the software version of the publication (crow.exe) follow the link: http://neurology.yassermetwally.com/crow.zip The case is also presented as a short case in PDF format, to download the short case follow the link: http://pdf.yassermetwally.com/short.pdf At the end of each year, all the publications are compiled on a single CD-ROM, please contact the author to know more details. Screen resolution is better set at 1024*768 pixel screen area for optimum display. Also to view a list of the previously published case records follow the following link (http://wordpress.com/tag/case-record/) or click on it if it appears as a link in your PDF reader To inspect the patient's full radiological study, click on the attachment icon (The paper clip icon in the left pane) of the acrobat reader then double click on the attached file. Click here to download the short case version of this case record in PDF format
- 13. REFERENCES References 1. Virchow R. Ueber die Erweiterung kleinerer Gefaesse. Archiv Pathol Anat Physiol Klin Med 1851; 3:427–462. 2. Robin C. Recherches sur quelques particularités de la structure des capillaires de l’encephale. J Physiol Homme Anim 1859;2:537–548. 3. Hutchings M, Weller RO. Anatomical relationships of the pia mater to cerebral blood vessels in man. J Neurosurg 1986;65:316–325. 4. Zhang ET, Inman CB, Weller RO. Interrelationship of the pia mater and the perivascular (Virchow-Robin) spaces in the human cerebrum. J Anat 1990;170:111–123. 5. Pollock H, Hutchings M, Weller RO, Zhang ET. Perivascular spaces in the basal ganglia of the human brain: their relationship to lacunes. J Anat 1997;191:337–346. 6. Schley D, Carare-Nnadi R, Please CP, Perry VH, Weller RO. Mechanisms to explain the reverse perivascular transport of solutes out of the brain. J Theor Biol 2006;238:962–974. 7. Durant-Fardel M. Traite du ramollissement du cerveau. Paris, France: Balliere, 1843. 8. Poirier J, Barbizet J, Gaston A, Meyrignac C. Thalamic dementia: expansive lacunae of the thalamo- paramedian mesencephalic area—hydrocephalus caused by stenosis of the aqueduct of Sylvius [in French]. Rev Neurol (Paris) 1983;139:349–358. 9. Benhaiem-Sigaux N, Gray F, Gherardi R, Roucayrol AM, Poirier J. Expanding cerebellar lacunes due to dilatation of the perivascular space associated with Binswanger’s subcortical arteriosclerotic encephalopathy. Stroke 1987;18:1087–1092. 10. Hughes W. Origin of lacunes. Lancet 1965;2:19–21. 11. Homeyer P, Cornu P, Lacomblez L, Chiras J, Derouesne C. A special form of cerebral lacunae: expanding lacunae. J Neurol Neurosurg Psychiatry 1996;61:200–202. 12. Mascalchi M, Salvi F, Godano U, et al. Expanding lacunae causing triventricular hydrocephalus: report of two cases. J Neurosurg 1999;91:669–674. 13. Awad IA, Johnson PC, Spetzler RF, Hodak JA. Incidental subcortical lesions identified on magnetic resonance imaging in the elderly. II. Postmortem pathological correlations. Stroke 1986;17: 1090–1097. 14. Adachi M, Hosoya T, Haku T, Yamaguchi K. Dilated Virchow-Robin spaces: MRI pathological study. Neuroradiology 1998;40:27–31. 15. Heier LA, Bauer CJ, Schwartz L, Zimmerman RD, Morgello S, Deck MD. Large Virchow-Robin spaces: MR- clinical correlation. AJNR Am J Neuroradiol 1989;10:929–936. 16. Rollins NK, Deline C, Morriss MC. Prevalence and clinical significance of dilated Virchow-Robin spaces in childhood. Radiology 1993;189:53–57. 17. Machado MA Jr, Matos AS, Goyanna F, Barbosa VA, Vieira LC. Dilatation of Virchow-Robin spaces in patients with migraine [in Portuguese]. Arq Neuropsiquiatr 2001;59:206–209. 18. MacLullich AM, Wardlaw JM, Ferguson KJ, Starr JM, Seckl JR, Deary IJ. Enlarged perivascular spaces are
- 14. associated with cognitive function in healthy elderly men. J Neurol Neurosurg Psychiatry 2004;75:1519–1523. 19. Taber KH, Shaw JB, Loveland KA, Pearson DA, Lane DM, Hayman LA. Accentuated Virchow-Robin spaces in the centrum semiovale in children with autistic disorder. J Comput Assist Tomogr 2004;28:263–268. 20. Achiron A, Faibel M. Sandlike appearance of Virchow-Robin spaces in early multiple sclerosis: a novel neuroradiologic marker. AJNR Am J Neuroradiol 2002;23:376–380. 21. Inglese M, Bomsztyk E, Gonen O, Mannon LJ, Grossman RI, Rusinek H. Dilated perivascular spaces: hallmarks of mild traumatic brain injury. AJNR Am J Neuroradiol 2005;26:719–724. 22. Patankar TF, Mitra D, Varma A, Snowden J, Neary D, Jackson A. Dilatation of the Virchow-Robin space is a sensitive indicator of cerebral microvascular disease: study in elderly patients with dementia. AJNR Am J Neuroradiol 2005;26: 1512–1520. 23. Saeki N, Sato M, Kubota M, et al. MR imaging of normal perivascular space expansion at midbrain. AJNR Am J Neuroradiol 2005;26:566–571. 24. Song CJ, Kim JH, Kier EL, Bronen RA. MR imaging and histologic features of subinsular bright spots on T2- weighted MR images: Virchow-Robin spaces of the extreme capsule and insular cortex. Radiology 2000;214:671–677. 25. Takahashi M, Uematsu H, Hatabu H. MR imaging at high magnetic fields. Eur J Radiol 2003;46: 45–52. 26. Uematsu H, Dougherty L, Takahashi M, et al. A direct comparison of signal behavior between 4.0 and 1.5 T: a phantom study. Eur J Radiol 2003; 45:154–159. 27. Sasaki M, Inoue T, Tohyama K, Oikawa H, Ehara S, Ogawa A. High-field MRI of the central nervous system: current approaches to clinical and microscopic imaging. Magn Reson Med Sci 2003; 2:133–139. 28. Ozturk MH, Aydingoz U. Comparison of MR signal intensities of cerebral perivascular (Virchow-Robin) and subarachnoid spaces. J Comput Assist Tomogr 2002;26:902–904. 29. Hirabuki N, Fujita N, Fujii K, Hashimoto T, Kozuka T. MR appearance of Virchow-Robin spaces along lenticulostriate arteries: spin-echo and two-dimensional fast low-angle shot imaging. AJNR Am J Neuroradiol 1994;15:277–281. 30. Braffman BH, Zimmerman RA, Trojanowski JQ, Gonatas NK, Hickey WF, Schlaepfer WW. Brain MR: pathologic correlation with gross and histopathology. 1. Lacunar infarction and Virchow-Robin spaces. AJR Am J Roentgenol 1988;151: 551–558. 31. Demaerel P, Wilms G, Baert AL, Van den Bergh V, Sainte T. Widening of Virchow-Robin spaces. AJNR Am J Neuroradiol 1996;17:800–801. 32. Jungreis CA, Kanal E, Hirsch WL, Martinez AJ, Moossy J. Normal perivascular spaces mimicking lacunar infarction: MR imaging. Radiology 1988; 169:101–104. 33. Pullicino PM, Miller LL, Alexandrov AV, Ostrow PT. Infraputaminal ‘lacunes’: clinical and pathological correlations. Stroke 1995;26:1598–1602. 34. Elster AD, Richardson DN. Focal high signal on MR scans of the midbrain caused by enlarged perivascular spaces: MR-pathologic correlation. AJR Am J Roentgenol 1991;156:157–160. 35. Duvernoy HM. Human brainstem vessels. Berlin, Germany: Springer-Verlag, 1978; 16–66.
- 15. 36. Ogawa T, Okudera T, Fukasawa H, et al. Unusual widening of Virchow-Robin spaces: MR appearance. AJNR Am J Neuroradiol 1995;16:1238–1242. 37. Sawada M, Nishi S, Hashimoto N. Unilateral appearance of markedly dilated Virchow-Robin spaces. Clin Radiol 1999;54:334–336. 38. Shiratori K, Mrowka M, Toussaint A, Spalke G, Bien S. Extreme, unilateral widening of Virchow-Robin spaces: case report. Neuroradiology 2002; 44:990–992. 39. Davis G, Fitt GJ, Kalnins RM, Mitchell LA. Increased perivascular spaces mimicking frontal lobe tumor. J Neurosurg 2002;97:723. 40. Romi F, Tysnes OB, Krakenes J, Savoiardo M, Aarli JA, Bindoff L. Cystic dilation of Virchow-Robin spaces in the midbrain. Eur Neurol 2002; 47:186–188. 41. Cakirer S. MR imaging findings in tumefactive perivascular spaces. Acta Radiol 2003;44:673–674. 42. Salzman KL, Osborn AG, House P, et al. Giant tumefactive perivascular spaces. AJNR Am J Neuroradiol 2005;26:298–305. 43. Kanamalla US, Calabro R, Jinkins JR. Cavernous dilatation of mesencephalic Virchow-Robin spaces with obstructive hydrocephalus. Neuroradiology 2000;42:881–884. 44. Papayannis CE, Saidon P, Rugilo CA, et al. Expanding Virchow Robin spaces in the midbrain causing hydrocephalus. AJNR Am J Neuroradiol 2003;24:1399–1403. 45. Rohlfs J, Riegel T, Khalil M, et al. Enlarged perivascular spaces mimicking multicystic brain tumors: report of two cases and review of the literature. J Neurosurg 2005;102:1142–1146. 46. Longatti PL, Fiorindi A, Carteri A, Caroli F, Martinuzzi A. Expanding cerebral cysts (lacunae): a treatable cause of progressive midbrain syndrome. J Neurol Neurosurg Psychiatry 2003;74:393–394. 47. House P, Salzman KL, Osborn AG, MacDonald JG, Jensen RL, Couldwell WT. Surgical considerations regarding giant dilations of the perivascular spaces. J Neurosurg 2004;100:820–824. 48. Fisher CM. Lacunes: small, deep cerebral infarcts. Neurology 1965;15:774–784. 49. Fisher CM. Lacunar strokes and infarcts: a review. Neurology 1982;32:871–876. 50. Bokura H, Kobayashi S, Yamaguchi S. Distinguishing silent lacunar infarction from enlarged Virchow-Robin spaces: a magnetic resonance imaging and pathological study. J Neurol 1998;245: 116–122. 51. Regli L, Regli F, Maeder P, Bogousslavsky J. Magnetic resonance imaging with gadolinium contrast agent in small deep (lacunar) cerebral infarcts. Arch Neurol 1993;50:175–180. 52. Baker LL, Stevenson DK, Enzmann DR. End-stage periventricular leukomalacia: MR evaluation. Radiology 1988;168:809–815. 53. Flodmark O, Lupton B, Li D, et al. MR imaging of periventricular leukomalacia in childhood. AJR Am J Roentgenol 1989;152:583–590. 54. Pretorius PM, Quaghebeur G. The role of MRI in the diagnosis of MS. Clin Radiol 2003;58:434–448. 55. Mathews VP, Alo PL, Glass JD, Kumar AJ, McArthur JC. AIDS-related CNS cryptococcosis: radiologic- pathologic correlation. AJNR Am J Neuroradiol 1992;13:1477–1486.
- 16. 56. Tien RD, Chu PK, Hesselink JR, Duberg A, Wiley C. Intracranial cryptococcosis in immunocompromised patients: CT and MR findings in 29 cases. AJNR Am J Neuroradiol 1991;12:283–289. 57. Wehn SM, Heinz ER, Burger PC, Boyko OB. Dilated Virchow-Robin spaces in cryptococcal meningitis associated with AIDS: CT and MR findings. J Comput Assist Tomogr 1989;13:756–762. 58. Miszkiel KA, Hall-Craggs MA, Miller RF, et al. The spectrum of MRI findings in CNS cryptococcosis in AIDS. Clin Radiol 1996;51:842–850. 59. Lee C, Dineen TE, Brack M, Kirsch JE, Runge VM. The mucopolysaccharidoses: characterization by cranial MR imaging. AJNR Am J Neuroradiol 1993;14:1285–1292. 60. Matheus MG, Castillo M, Smith JK, Armao D, Towle D, Muenzer J. Brain MRI findings in patients with mucopolysaccharidosis types I and II and mild clinical presentation. Neuroradiology 2004;46:666–672. 61. Takahashi Y, Sukegawa K, Aoki M, et al. Evaluation of accumulated mucopolysaccharides in the brain of patients with mucopolysaccharidoses by (1)H-magnetic resonance spectroscopy before and after bone marrow transplantation. Pediatr Res 2001;49:349–355. 62. Tien RD, Felsberg GJ, Friedman H, Brown M, MacFall J. MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. AJR Am J Roentgenol 1994;162: 671–677. 63. Noguchi K, Watanabe N, Nagayoshi T, et al. Role of diffusion-weighted echoplanar MRI in distinguishing between brain abscess and tumour: a preliminary report. Neuroradiology 1999;41:171–174. 64. Desprechins B, Stadnik T, Koerts G, Shabana W, Breucq C, Osteaux M. Use of diffusion-weighted MR imaging in differential diagnosis between intracerebral necrotic tumors and cerebral abscesses. AJNR Am J Neuroradiol 1999;20:1252–1257. 65. do Amaral LL, Ferreira RM, da Rocha AJ, Ferreira NP. Neurocysticercosis: evaluation with advanced magnetic resonance techniques and atypical forms. Top Magn Reson Imaging 2005;16: 127–144. 66. Dumas JL, Visy JM, Belin C, Gaston A, Goldlust D, Dumas M. Parenchymal neurocysticercosis: follow-up and staging by MRI. Neuroradiology 1997;39:12–18. 67. Noujaim SE, Rossi MD, Rao SK, et al. CT and MR imaging of neurocysticercosis. AJR Am J Roentgenol 1999;173:1485–1490. 68. Van Tassel P, Cure JK. Nonneoplastic intracranial cysts and cystic lesions. Semin Ultrasound CT MR 1995;16:186–211. 69. Andrews BT, Halks-Miller M, Berger MS, Rosenblum ML, Wilson CB. Neuroepithelial cysts of the posterior fossa: pathogenesis and report of two cases. Neurosurgery 1984;15:91–95. 70. Guermazi A, Miaux Y, Majoulet JF, Lafitte F, Chiras J. Imaging findings of central nervous system neuroepithelial cysts. Eur Radiol 1998;8:618–623. 71. Sherman JL, Camponovo E, Citrin CM. MR imaging of CSF-like choroidal fissure and parenchymal cysts of the brain. AJNR Am J Neuroradiol 1990;11:939–945. 72. Metwally, MYM: Textbook of neuroimaging, A CD-ROM publication, (Metwally, MYM editor) WEB-CD agency for electronic publication, version 11.4a. October 2010 73. Heier LA, Bauer CJ, Schwartz L, Zimmerman RD, Morgello S, Deck MD. Large Virchow-Robin spaces: MR- clinical correlation. AJNR Am J Neuroradiol 1989;10:929 -936
- 17. 74. Hommel M, Gray F. Microvascular pathology. In: Caplan L, ed. Brain Ischaemia: Basic Concepts and Clinical Relevance. New York: Springer-Verlag Berlin; 1995:215-223 75. Furuta A, Ishii N, Nishihara Y, Horie A. Medullary arteries in aging and dementia. Stroke 1991;22:442- 446 76. Brun A, Fredriksson K, Gustafson L. Pure subcortical arterioscle- rotic encephalopathy (Binswanger’s disease): a clinicopathological study. Part 2: Pathological features. Cerebrovasc Dis 1992;2:87-92 77. Thomas AJ, O’Brien JT, Davis S, et al. Ischemic basis for deep white matter hyperintensities in major depression: a neuropatho- logical study. Arch Gen Psychiatry 2002;59:785-792 78. Thacker NA, Varma AR, Bathgate D, et al. Dementing disor- ders: volumetric measurement of cerebrospinal fluid to distin- guish normal from pathologic findings: feasibility study. Radi- ology 2002;224:278 -285 79. Poirier J, Derouesne C. Cerebral lacunae: a proposed new classi- fication [letter]. Clin Neuropathol 1984;3:266 80. Fazekas F, Kleinert R, Offenbacher H, et al. The morphologic correlate of incidental punctate white matter hyperintensities on MR images. AJNR Am J Neuroradiol 1991;12:915-921 81. Ogawa T, Okudera T, Fukasawa H, et al. Unusual widening of Virchow-Robin spaces: MR appearance. AJNR Am J Neuroradiol 1995;16:1238 -1242 82. Hughes W. Origin of lacunes. Lancet 1965 1:19 -21 83. Benhaiem-Sigaux N, Gray F, Gherardi R, Roucayrol AM, Poirier J. Expanding cerebellar lacunes due to dilatation of the perivascular space associated with Binswanger’s subcortical arteriosclerotic en- cephalopathy. Stroke 1987;18:1087-1092 84. Derouesne C, Gray F, Escourolle R, Castaigne P. "Expanding cerebral lacunae" in a hypertensive patient with normal pressure hydrocephalus. Neuropathol Appl Neurobiol 1987;13:309 -320 85. Pullicino PM, Miller LL, Alexandrov AV, Ostrow PT. Infrapu- taminal "lacunes": clinical and pathological correlations. Stroke 1995;26:1598 -1602 86. Jellinger KA. The pathology of ischemic-vascular dementia: an update. J Neurol Sci 2002; 203-204:153-157 87. Hulette C ND, McKeel D, Morris K, Mirras SS, Sumi SM, et. a. Clinical-neuropathologic findings in multi- infarct dementia: a re- portof six autopsied cases. Neurology 1997;48:668 – 672 88. Erkinjuntti T. Diagnosis and management of vascular cognitive impairment and dementia. J Neural Transm Suppl 2002:91-109 89. Hentschel F, Kreis M, Damian M, Krumm B. Microangiopathic lesions of white matter: quantitation of cerebral MRI findings and correlation with psychological tests. Nervenarzt 2003;74:355-361
