Periodic table intro2
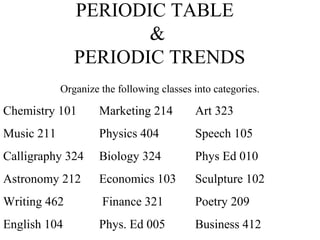
- 1. PERIODIC TABLE & PERIODIC TRENDS Organize the following classes into categories. Chemistry 101 Marketing 214 Art 323 Music 211 Physics 404 Speech 105 Calligraphy 324 Biology 324 Phys Ed 010 Astronomy 212 Economics 103 Sculpture 102 Writing 462 Finance 321 Poetry 209 English 104 Phys. Ed 005 Business 412
- 3. Where did the Periodic Table come from? • 1829 – J.W. Dobereiner • Triad Classification • 1864 – John Newlands • Law of Octaves • 1869 – Dmitri Mendeleev • Credited with modern Periodic Table
- 4. Still Not Perfect Potassium Atomic Mass: 39.10 amu • Highly reactive Metal Argon Atomic Mass: 39.95 amu • Inert gas *Important fact you may want to right down* The Modern Periodic Table is based on atomic number and electron configuration, not atomic mass.
- 5. Periodic Law • When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. Ya’ll best line up d’em elements in order of increasing proton number to pick out the properties!
- 6. METALS • Have only a few electrons in outer most energy level. • Lose e- to form positive ions (X+ ) • Good Conductors • Malleable (sheets) • Ductile (wire) • Often Lusterous
- 7. NONMETALS • More than 3 e- in their outermost orbit • Gain electrons to form negative ions (X- ) • Dull in appearance • Brittle
- 8. METALLOIDS • Have properties of metals & nonmetals • Positioned on metal / nonmetal line • B, Si, Ge, As, Sb, Te, Po, At
- 9. A Groups = Main Group Elements ALKALI ALKALINEEARTH NOBLEGASES HALOGENS CHALCOGENS IA IIA VIAVIIA VIIIA IB – VIIIB TRANSITION METALS INNER TRANSITION METALS (Lanthanide & Actinide Series) VAIVAIIIA 3B 4B 5B 6B 7B 8B 1B 2B Coinage Metals
- 10. Pick a Trend, Any Trend 1. Atomic Size 2. Ionization Energy 3. Electron Affinity 4. Electronegativity
- 11. Atomic Radius: No definite shape, but scientist can get a rough measure.
- 12. Atomic Size It doesn’t depend on Weight Watchers
- 13. Ionization Energy The energy required to remove an electron from an atom in the gas phase. A(g)+ Energy A+ (g) + e- Ionization energy measures how tightly an electron is held in the atom.
- 14. Ionization Energy (cont.) • More and more energy is required to move each electron from an atom • Metals generally have low IE. • Nonmetals have high IE. • IE increases as you move across a period and decreases as you go down a group or family.
- 15. Electron Affinity EA – It’s NOT a clothing company • The energy used or released for a gaseous atom to gain an electron. A(g) + e- A- (g) + Energy
- 16. Electron Affinity (cont.) In general. . . • EA increases (becomes more negative) as you go across a period and decreases as you go down a group or family. • The greater the electron affinity, the greater the IE. – Metals have lower EAs – Nonmetals have higher EAs
- 17. Electronegativity A comparative scale relating the abilities of elements to attract electrons when their atoms are combined. • Active metals (IA) have the lowest Ens • Most nonmetals have the highest Ens
- 19. And Now a guest appearance by Mr. Tom Lehrer
Notas del editor
- Because atoms with similar electron configurations react similarly, we can see trends or patterns in the periodic table.
- There is no sharp boundary to an atom, but we can get a rough measure of atomic size from the most probable electron distance from the nucleus, the area that contains 90% of the electron density. This distance is called the atomic radius.
- Atomic Radius is effected by “Effective Nuclear Charge” – the charge of the nucleus felt by an electron. As ENC increases, the atomic radius decreases. I.E. a strong positive nucleus will pull in more negative electrons. Atomic radius Decreases as you move across a period and increases as you move down a group
- Both electron affinity and ionization energy deal with isolated atoms, where as Electronegativity allows us to compare an elements attractive force to electrons when their atoms are combined.