Sulphur cycle
•Download as PPTX, PDF•
5 likes•1,554 views
stpm biology sulphur cycle
Report
Share
Report
Share
Recommended
Recommended
Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient Uptake

Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient UptakeVasantrao Nail Marathwada Krishi Vidyapeeth, Parbhani
More Related Content
What's hot
What's hot (18)
Viewers also liked
Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient Uptake

Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient UptakeVasantrao Nail Marathwada Krishi Vidyapeeth, Parbhani
Viewers also liked (18)
Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient Uptake

Effect of Nitrogen and Sulphur on Growth, Yield, Nutrient Uptake
B Sc Agri II Sc,Sf & Nm, U 1 Soil And Plant Nutrient

B Sc Agri II Sc,Sf & Nm, U 1 Soil And Plant Nutrient
Similar to Sulphur cycle
Similar to Sulphur cycle (20)
Chemisty project _9_F_100383_Acid Rain [Autosaved].pptx![Chemisty project _9_F_100383_Acid Rain [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Chemisty project _9_F_100383_Acid Rain [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Chemisty project _9_F_100383_Acid Rain [Autosaved].pptx
Recently uploaded
Recently uploaded (20)
❤Jammu Kashmir Call Girls 8617697112 Personal Whatsapp Number 💦✅.

❤Jammu Kashmir Call Girls 8617697112 Personal Whatsapp Number 💦✅.
Botany krishna series 2nd semester Only Mcq type questions

Botany krishna series 2nd semester Only Mcq type questions
Hubble Asteroid Hunter III. Physical properties of newly found asteroids

Hubble Asteroid Hunter III. Physical properties of newly found asteroids
Disentangling the origin of chemical differences using GHOST

Disentangling the origin of chemical differences using GHOST
Stunning ➥8448380779▻ Call Girls In Panchshil Enclave Delhi NCR

Stunning ➥8448380779▻ Call Girls In Panchshil Enclave Delhi NCR
Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency

Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency
Unlocking the Potential: Deep dive into ocean of Ceramic Magnets.pptx

Unlocking the Potential: Deep dive into ocean of Ceramic Magnets.pptx
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
Forensic Biology & Its biological significance.pdf

Forensic Biology & Its biological significance.pdf
Pests of mustard_Identification_Management_Dr.UPR.pdf

Pests of mustard_Identification_Management_Dr.UPR.pdf
CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡

CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡
Spermiogenesis or Spermateleosis or metamorphosis of spermatid

Spermiogenesis or Spermateleosis or metamorphosis of spermatid
All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...

All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...
Sulphur cycle
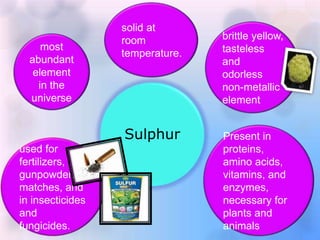
- 1. Sulphur most abundant element in the universe solid at room temperature. brittle yellow, tasteless and odorless non-metallic element Present in proteins, amino acids, vitamins, and enzymes, necessary for plants and animals used for fertilizers, gunpowder, matches, and in insecticides and fungicides.
- 2. ELEMENTS INVOLVED IN CYCLE
- 3. +O2 Burning of fossil fuel volcanic eruptions +H20 +Ammonia +O2 +O2 Certain marine algae anaerobic decay of sulfur-containg biological material in swamps Aerobic O2 requiring bacteria (Thiobacillus sp) incorporate with sulfur decompose by Desulphovibrio sp. (organic S H2S) Oxidise by Chromatium Wind carries Sulfuric acid and ammonium salts which falls to earth in form of Precipitation. Rain The remaining sulphur is lost into the oceans depth combining with iron to form Ferrous Sulfide which is responsible for the black color of most marine sediments.
- 4. Effect irritates human respiratory tract increased incidences of respiratory illnesses such as bronchitis , asthma, and heart disease Low level of O2 in blood Causes major damage to vegetation in areas surrounding the source of emission Reduced plant growth Acid rain
- 5. Acid Rain Low pH level of soil Nutrient leaching Low: Macro High: Micro High toxicity (Al, Fe, Manganese toxicity) Affects activity of microorganis ms Reduced humus production, mineralization and nutrient fixation Nutrient deficient soils
