Identify Acid-Fast Bacteria Using Staining Techniques
- 1. Acid-fast staining Anup Muni Bajracharya
- 2. Acid-fast staining • is a differential staining • used to differentiate acid fast and non acid fast bacteria. • used to identify acid-fast organisms such as members of the genus Mycobacterium . • also known as Ziehl-Neelsen method • first introduced by Paul Ehrlich later modified by two German doctors bacteriologist Franz Ziehl (1859–1926) and the pathologist Friedrich Neelsen (1854–1898). A.B
- 3. History • In 1882 Robert Koch discovered the etiology of tuberculosis. • Soon after Koch’s discovery, Paul Ehrlich developed a stain for mycobacterium tuberculosis, called the alum hematoxylin stain. • Franz Ziehl then altered Ehrlich’s staining technique by using carbolic acid as the mordant. • Friedrich Neelsen kept Ziehl’s choice of mordant but changed the primary stain to carbol fuchsin. • Ziehl and Neelsen’s modifications together have developed the Ziehl-Neelsen stain. • Another acid-fast satin was developed by Joseph Kinyoun by using the Ziehl-Neelsen staining technique but removing the heating step from the procedure. • This new stain from Kinyoun was named the Kinyoun stain. A.B
- 4. What are acid fast bacteria? • those which have a high content of mycolic acids in their cell walls. • are characterized by • wax-like, nearly impermeable cell walls; • contain mycolic acid and large amounts of fatty acids, waxes, and complex lipids. • are highly resistant to disinfectants and dry conditions. • Due to their waxy cell wall components, Mycobacterium are acid fast; that is, they retain the red dye, carbol fuchsin, after rinsing with acid solvents. A.B
- 5. Acid fast organisms Acid-fast organisms like Mycobacterium contain large amounts of lipid substances,mycolic acids. which resist staining by ordinary methods such as a Gram stain. Require acid-fast staining that is used to screen for the Mycobacterium, Nocardia, and Legionella species in body tissues and fluids. Acid-fastness is an uncommon characteristic shared by the genera Mycobacterium and Nocardia (weakly acid-fast). Because of this feature, this stain is extremely helpful in identification in diseases caused by acid-fast bacteria, particularly tuberculosis and leprosy. A.B
- 6. Principle • Because the cell wall containing mycolic acid is so waxy and resistant to most compounds, acid-fast organisms require a special staining technique. • The primary stain used in acid-fast staining, carbol fuchsin, is lipid-soluble and contains phenol, which helps the stain penetrate the cell wall. This is further assisted by the addition of heat. • The smear is then rinsed with a very strong decolorizer, which strips the stain from all non-acid-fast cells but does not permeate the cell wall of acid-fast organisms. • The decolorized non-acid-fast cells then take up the counterstain. • Acid fast bacteria will be red, while nonacid fast bacteria will stain green. A.B
- 7. A.B
- 8. Requirements Reagents • Primary Stain: Carbol-fuchsin. • Decoloriser: (HCl+Ethanol) • Counterstain: Methylene blue. Materials Slides Sample Inoculating loop Bunsen burner Cotton Microscope A.B
- 9. Preparation of reagents • Primary Stain: 0.3% Carbol-fuchsin. • Dissolve 50 g phenol in 100 mL ethanol (95%). • Dissolve 3 g Basic fuchsin in the mixture and add distilled water to bring the volume to 1 L. Decolorization Solution: (HCl+Ethanol) • Add 30 mL hydrochloric acid to 1 L of 95% denatured alcohol. • Cool and mix well before use. Counterstain: 0.3% Methylene blue. • Dissolve 3 g methylene blue in 1 L distilled water. A.B
- 10. Procedure • Take a clean grease free slide. • Prepare the smear from provided sample. • Air dry and heat fixed the smear. • Place a small strip of filter paper over the top of smear and place the slide over a boiling hot water bath on a mesh surface. • Cover the filter paper with the primary stain, carbol fuchsin. • Leave the slide on the water bath for 5 minutes or more. • Note- Continue to apply stain if the filter paper begins to dry. • Remove the filter paper and rinse the slide with water until the solution runs clear. • Run acid-alcohol decolorizer over the slide for approximately 10 to 15 seconds. • Rinse the slide with water. • Cover the smear with the counterstain, methylene blue, for 1 minute. • Gently rinse the slide with water. • Blot the slide dry with bibulous paper. • Observe under microscope. A.B
- 11. Procedure A.B
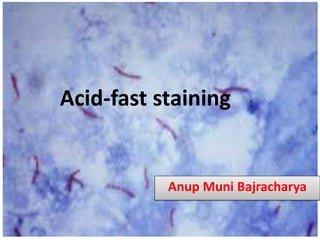
- 12. Observation under Microscope Acid fast: Red, straight or slightly curved rods, occurring singly or in small groups, may appear beaded Non-acid fast: Blue color; In addition, background material stain blue. A.B
- 13. Organisms stained by acid fast staining A.B
- 14. Kinyoun stain • Unlike the (Z-N stain), the Kinyoun method of staining does not require heating. • In the ZN stain, heat acts as a physical mordant while phenol (carbol of carbol fuschin) acts as the chemical mordant. • Since the Kinyoun stain is a cold method (no heat applied), the concentration of carbol fuschin used is increased. A.B
