Poster main revised
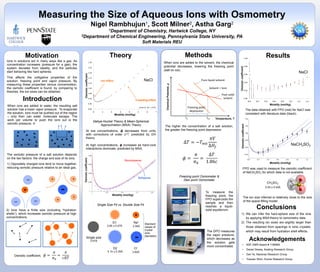
- 1. When ions are added to water, the resulting salt solution has a lower vapor pressure. To evaporate the solution, ions must be pushed out of the region – only then can water molecules escape. The work per volume to push the ions out is the osmotic pressure, π. Theory Results Acknowledgements Introduction Methods Measuring the Size of Aqueous Ions with Osmometry Nigel Rambhujun1, Scott Milner2, Astha Garg2 1Department of Chemistry, Hartwick College, NY 2Department of Chemical Engineering, Pennsylvania State University, PA Soft Materials REU The osmotic pressure of a salt solution depends on the two factors: the charge and size of its ions. 1) Oppositely charged ions tend to move together, reducing osmotic pressure relative to an ideal gas. Debye-Huckel Theory & Mean-Spherical Approximation (MSA) Theory 2) Ions have a finite size (including “hydration shells”), which increases osmotic pressure at high concentrations. Motivation 0.85 0.90 0.95 1.00 1.05 1.10 1.15 1.20 1.25 1.30 0 1 2 3 4 5 6 7 Osmoticcoefficient Molality (mol/kg) NaClIon effect Crowding effect 𝜙 = 𝜋 𝜋0 = 𝜋 𝑐𝑅𝑇 At low concentrations, ϕ decreases from unity with corrections of order c1/2, predicted by DH theory. At high concentrations, ϕ increases as hard-core interactions dominate, predicted by MSA. Freezing point Osmometer & Dew point Osmometer Single Size Fit vs. Double Size Fit 0.5 1.0 1.5 2.0 0.94 0.96 0.98 1.00 Molality (mol/kg) Osmoticcoefficient Monodisperse Bidisperse Na+ 2.04Å Cl- 3.62Å D1 2.85 ± 0.47Å Single size 3.57Å D2 4.14 ± 0.35Å The higher the concentration of a salt solution, the greater the freezing point depression. Pure liquid solvent Solvent + Ions Pure solid solvent Temperature, T ChemicalPotential,μ Freezing point depression Tm0Tm’ When ions are added to the solvent, the chemical potential decreases, lowering the freezing point (salt on ice). • NSF DMR-Award # 1156960 • Daniel Dewey, Keating Research Group • Dan Ye, Maranas Research Group • Yuexiao Shen, Kumar Research Group Standard values of crystal ionic diameters 0.90 0.92 0.94 0.96 0.98 1.00 0.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 Osmoticcoefficient Molality (mol/kg) NaCl To measure the freezing point, the FPO supercools the sample and then reaches a liquid- solid equilibrium. The DPO measures the vapor pressure, which decreases as the solution gets more concentrated. Hamer W., Wu Y. (1972) Conclusions 0.0 0.2 0.4 0.6 0.8 1.0 1.2 0.94 0.96 0.98 1.00 1.02 1.04 CH3SO3 - 5.54 ± 0.40Å NaCH3SO3 Ions in solutions act in many ways like a gas. As concentration increases (pressure for a gas), the system deviates from ideality, and the particles start behaving like hard spheres. This affects the colligative properties of the solution: freezing point and vapor pressure. By measuring these properties versus concentration, the osmotic coefficient is found; by comparing to theories, the ion sizes can be obtained. 1) We can infer the hard-sphere size of the ions by applying MSA theory to osmometry data. 2) The resulting ion sizes are slightly larger than those obtained from spacings in ionic crystals, which may result from hydration shell effects. The data obtained with FPO (red) for NaCl was consistent with literature data (black). FPO was used to measure the osmotic coefficient of NaCH3SO3 for which data is not available. Molality (mol/kg) Osmoticcoefficient The ion size inferred is relatively close to the size of the space-filling model. Osmotic coefficient, Δ𝑇 = −𝑇 𝑚0 𝜋𝑉 Δ𝐻𝑓 𝜙 = 𝜋 𝜋0 = Δ𝑇 1.86𝑐