Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook.
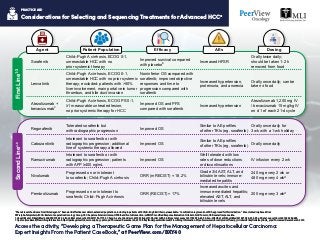
- 1. Considerations for Selecting and Sequencing Treatments for Advanced HCCa a Phase 3 level of evidence for all listed agents. b Real-world effectiveness data in extended populations, including in patients with Child–Pugh B cirrhosis, are available. c Combination regimen is not FDA approved for this indication. d Dose studied in phase 2 trial. AFP: alpha-fetoprotein; ECOG: Eastern Cooperative Oncology Group; HCC: hepatocellular carcinoma; HFSR: hand-foot skin reaction; mRECIST: modified Response Evaluation Criteria In Solid Tumors; ORR: overall response rate. 1. Llovet JM et al. NEnglJMed. 2008;359:378-390. 2. Kudo M et al. Lancet. 2018;391:1163-1173. 3. Cheng A-L et al. AnnOncol. 2019;30(suppl 9): Abstract LBA3. 4. Bruix J et al. Lancet. 2017;389:56-66. 5. Abou-Alfa GK et al. NEnglJMed. 2018;379:54-63. 6. Zhu AX et al. LancetOncol. 2019;20:282-296. 7. Opdivo (nivolumab) Prescribing Information. https://packageinserts.bms.com/pi/pi_opdivo.pdf. Accessed December 20, 2019. 8. Keytruda (pembrolizumab) Prescribing Information. https://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. Accessed December 20, 2019. PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Regorafenib Tolerated sorafenib but with radiographic progression Improved OS Similar to AE profiles of other TKIs (eg, sorafenib) Orally once daily for 3 wk with a 1-wk holiday Cabozantinib Intolerant to sorafenib or with radiographic progression; additional line of systemic therapy allowed Improved OS Similar to AE profiles of other TKIs (eg, sorafenib) Orally once daily Ramucirumab Intolerant to sorafenib or with radiographic progression; patients with AFP ≥400 ng/mL Improved OS Well tolerated with low rates of dose reductions or discontinuations IV infusion every 2 wk Nivolumab Progressed on or intolerant to sorafenib; Child–Pugh A cirrhosis ORR (mRECIST) = 18.2% Grade 3/4 AST, ALT, and bilirubin levels; immune- mediated hepatitis 240 mg every 2 wk or 480 mg every 4 wkd Pembrolizumab Progressed on or intolerant to sorafenib; Child–Pugh A cirrhosis ORR (RECIST) = 17% Increased ascites and immune-mediated hepatitis; elevated AST, ALT, and bilirubin levels 200 mg every 3 wkd Child–Pugh A cirrhosis, ECOG 0-1, unresectable HCC with no prior systemic therapy Improved survival compared with placebob Increased HFSR Orally twice daily; should be taken 1-2 h removed from food Lenvatinib Child–Pugh A cirrhosis, ECOG 0-1, unresectable HCC with no prior systemic therapy; excluded: patients with >50% liver involvement, main portal vein tumor thrombus, and bile duct invasion Noninferior OS compared with sorafenib; improved objective responses and time to progression compared with sorafenib Increased hypertension, proteinuria, and anorexia Orally once daily; can be taken ± food Atezolizumab + bevacizumabc Child–Pugh A cirrhosis, ECOG PS 0-1, ≥1 measurable untreated lesion, no prior systemic therapy for HCC Improved OS and PFS compared with sorafenib Increased hypertension Atezolizumab 1,200 mg IV / bevacizumab 15 mg/kg IV on d 1 of each 21-d cycle Sorafenib FirstLine1-3SecondLine4-8 EfficacyAgent Patient Population AEs Dosing
- 2. Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Selected Ongoing Clinical Trials in HCC1 PRACTICE AID Immunotherapeutic Agents HIMALAYAa (NCT03298451) IMbrave150 (NCT03434379) 3 3 Durvalumab ± tremelimumab vs sorafenib Atezolizumab + bevacizumab vs sorafenib First-LineAdvancedHCC COSMIC-312 (NCT03755791) LEAP-002 (NCT03713593) 3 3 Cabozantinib ± atezolizumab vs sorafenib Pembrolizumab + lenvatinib vs placebo + lenvatinib NCT03412773a Tislelizumab (BGB-A317) vs sorafenib 3 CheckMate -9DX (NCT03383458) KEYNOTE-937 (NCT03867084) 3 3Nivolumab (adjuvant) vs placebo Pembrolizumab (adjuvant) vs placebo Resectedor LocallyAdvancedHCC EMERALD-1 (NCT03778957) EMERALD-2 (NCT03847428) 3 3 Durvalumab ± bevacizumab + TACE vs placebo + TACE Durvalumab ± bevacizumab vs placebo IMbrave050 (NCT04102098) Atezolizumab plus bevacizumab (adjuvant) vs active surveillance 3 NCT03916627 Cemiplimab 2 CheckMate -9DW (NCT04039607) 3 Nivolumab + ipilimumab vs sorafenib or lenvatinib NCT03638141 2 TACE + durvalumab + tremelimumab Intermediate HCC IMMUTACE (NCT03572582) 2Nivolumab + TACE NCT03347292 1Regorafenib + pembrolizumab Trial Name Trial Arms Phase NCT03841201 2Nivolumab + lenvatinib
- 3. Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Selected Ongoing Clinical Trials in HCC1 PRACTICE AID a Active, not recruiting. HCC: hepatocellular carcinoma; SBRT: stereotactic body radiation therapy; TACE: transarterial chemoembolization; TTFields: tumor treating fields. 1. https://clinicaltrials.gov. Accessed January 13, 2020. Other Treatment Modalities and Strategies ResectedorLocally AdvancedHCC NCT02762266 SBRT vs TACE 3 NCT02182687 SBRT vs TACE 2 Trial Name Trial Arms Phase Intermediateand AdvancedHCC NCT01730937 SBRT + sorafenib vs sorafenib 3 HEPANOVA (NCT03606590) TTFields + sorafenib 2
- 4. Clinical Monitoring Recommendations for Advanced HCC Treatments1 Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 PRACTICE AID Complete Blood Count (CBC) Comprehensive Metabolic Panel • Sorafenib: B/L, every 2 wk for 2 mo, then mo • Lenvatinib: B/L, every 2 wk for 2 mo, then mo • Regorafenib: B/L, every 2 wk for 2 mo, then mo • Cabozantinib: B/L, every 2 wk for 2 mo, then mo • Ramucirumab: Every 2 wk • Nivolumab: B/L, every 2-4 wk during Tx • Pembrolizumab: B/L and every 3 wk during Tx Thyroid-Stimulating Hormone Sorafenib: B/L, then every 2-3 mo Lenvatinib: B/L, then mo Regorafenib: B/L, then every 2-3 mo Cabozantinib: B/L, then every 2-3 mo Ramucirumab: — Nivolumab: B/L, every 4-6 wk on Tx, every 6-12 wk after Tx Pembrolizumab: B/L, every 4-6 wk on Tx, every 6-12 wk after Tx Clinical Evaluation While on Therapy • Sorafenib: Every 1-2 wk for 6 wk, then mo • Lenvatinib: Every 1-2 wk for 6 wk, then mo • Regorafenib: Every 1-2 wk for 6 wk, then mo • Cabozantinib: Every 1-2 wk for 6 wk, then mo • Ramucirumab: Every 2-4 wk • Nivolumab: Every 2-4 wk • Pembrolizumab: Every 3-6 wk • Sorafenib: B/L, at 2 wk, then mo • Lenvatinib: B/L, at 2 wk, then mo • Regorafenib: B/L, at 2 wk, then mo • Cabozantinib: B/L, at 2 wk, then mo • Ramucirumab: Every 2 wk • Nivolumab: Every 2-4 wk • Pembrolizumab: Every 3 wk
- 5. Clinical Monitoring Recommendations for Advanced HCC Treatments1 a Use caution about applicability of data, because these agents, except for nivolumab, have not formally been tested in patients with Child–Pugh B scores. B/L: baseline. 1. Grieb BC et al. AmSocClinOncolEducBook. 2019;39:248-260. Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 PRACTICE AID Dose Adjustment in Child–Pugh Ba Urinalysis Electrocardiogram (ECG) Blood Pressure • Sorafenib: Every 1-2 wk for up to 8 wk, then mo • Lenvatinib: Every 1-2 wk for up to 8 wk, then mo • Regorafenib: Every 1-2 wk for 6 wk, then mo • Cabozantinib: Every 1-2 wk for up to 8 wk, then mo • Ramucirumab: Every 2 wk • Nivolumab: — • Pembrolizumab: — • Sorafenib: Regularly • Lenvatinib: Regularly • Regorafenib: Regularly • Cabozantinib: Regularly • Ramucirumab: Regularly • Nivolumab: — • Pembrolizumab: — • Sorafenib: B/L, 2-4 wk after start, then every 3 mo • Lenvatinib: B/L, 2-4 wk after start, then every 3 mo • Regorafenib: B/L, 2-4 wk after start, then every 3 mo • Cabozantinib: B/L, 2-4 wk after start, then every 3 mo • Ramucirumab: B/L, 2-4 wk after start, then every 3 mo • Nivolumab and pembrolizumab: — • Sorafenib: None • Lenvatinib: 8 mg daily • Regorafenib: No dose adjustment for bilirubin ≤3 ULN • Cabozantinib: Dose reduce with caution • Ramucirumab: Not indicated; increased hepatic toxicity • Nivolumab: None • Pembrolizumab: No data
- 6. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 HCC in Cirrhotic Liver1 Solitary 2-3 nodules 3 cm tnalpsnarT candidate Resection No Yes No tnalpsnarT Ablation Chemoembolization Systemic therapyAblation BSC Optimal surgical candidate Yes Very early stage (0) Single <2 cm Preserved liver function PS 0 Early stage (A) 3-2roelgniS nodules <3 cm Preserved liver function PS 0 Intermediate stage (B) ,raludonitluM unresectable Preserved liver function PS 0 Advanced stage (C) Portal invasion/ extrahepatic spread Preserved liver function PS 1-2 Terminal stage (D) Not transplantable egats-dnE liver function PS 3-4 HCC Patient Cases
- 7. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 1 Therapy Considerations for Early-Stage HCC Clinical Presentation and Testing Early-Stage (BCLC A) HCC2-11 • 32-year-old man with chronic HBV, on tenofovir, and with compensated cirrhosis • Undergoing HCC surveillance and found to have liver mass • MRI shows 3.5-cm LR-5 lesion in segment VI • Child–Pugh A: Bili 0.7, Alb 4.0, INR 1.0 • PLT: 217 • ECOG 0 • Patient undergoes robotic liver resection without complication • Returns to clinic, at which time, you reinforce risk of recurrence and need for continued surveillance • Recurrence: 70%-80% at 5 years • No proven adjuvant therapy to decrease HCC recurrence • Ongoing phase 3 trials are testing adjuvant immunotherapy monotherapy and combinations (eg, CheckMate -9DX, KEYNOTE-937, EMERALD-2, IMbrave050)
- 8. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 2 Therapy Considerations for Intermediate- Stage HCC Clinical Presentation and Testing Intermediate-Stage (BCLC B) HCC12-13 • AASLD guidance: TACE (level 1 evidence) • Suboptimal long-term survival, particularly in patients with larger tumor burden; offers opportunity for combinations with systemic therapies • Ongoing trials are testing immunotherapy in combination with LRT (eg, phase 3 trial EMERALD-1) • Consideration of transition to systemic therapy is important if patient fails TACE multiple times • 64-year-old man with NASH and compensated cirrhosis • Found to have incidental liver mass on imaging • MRI shows 6.5-cm LR-5 lesion with two satellite nodules • No vascular invasion and no metastatic disease • Child–Pugh A: Bili 0.9, Alb 3.7, INR 1.0 • ECOG 0
- 9. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 3 Appropriate First-Line Options for Newly Diagnosed Advanced HCC Clinical Presentation and Testing Newly Diagnosed Advanced (BCLC C) HCC14-20 • 59-year-old man with NASH cirrhosis • Child–Pugh A; AFP 178 ng/mL • No ascites or encephalopathy • ECOG-PS 0 • Previously treated with two rounds of TACE • Follow-up MRI shows multifocal HCC (LR-5) with three lesions, largest 6.5 cm, with invasion into the right portal vein • Sorafenib • Lenvatinib • Emerging option: atezolizumab/bevacizumab (IMbrave150 results) • Phase 3 clinical trials Checkpoint inhibitor + TKI (ie, LEAP-002, COSMIC-312) PD-1/L1 inhibitor + CTLA-4 inhibitor (ie, CheckMate -9DW, HIMALAYA)
- 10. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 4 Selecting a Second-Line Targeted Therapy Option Clinical Presentation and Testing Previously Treated Advanced (BCLC C) HCC21-23 • Consider AE profile, AFP levels, liver function, comorbidities, and prior treatment history Regorafenib Did not receive prior sorafenib Ramucirumab AFP levels <400 ng/mL Cabozantinib Allowed up to two lines of prior therapy Favorable data in patients with HBV • 54-year-old Asian woman with chronic HBV on entecavir with undetectable viral load • Treated with TACE for a 6-cm right lobe liver tumor 6 months ago • MRI now shows enlarging 8-cm right lobe tumor, HCC (LR-5), with two satellite lesions and new right branch portal vein tumor thrombus • Child–Pugh A; ECOG-PS 0; AFP level 142 ng/mL • Started on lenvatinib 12 mg daily Required dose reduction to 8 mg/d for fatigue and HTN Demonstrated initial tumor regression on restaging imaging for 6 months Developed lung metastases after 6 months; AFP has risen to 233 ng/mL
- 11. Therapeutic Options in HCC: A Patient Casebook for Physicians PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 5 Clinical Presentation and Testing Previously Treated Advanced (BCLC C) HCC • 69-year-old man with chronic HCV was treated and cured with direct-acting antiviral therapy 2 years ago • He then presents to his PMD with right upper quadrant pain • He was found to have an 8-cm mass and satellite lesions in the right lobe; he was hypervascular with delayed washout, and he has right portal vein thrombosis • Hb 12.3; MCV 90; PLT 95; bili 1.8; Alb 3.1; Cr 0.9; AFP 78 ng/mL • History: HCV, T2DM, HTN • No ascites; no encephalopathy; PS 1 • Staging: no extrahepatic disease; BCLC C • Started on sorafenib 400 mg twice daily; dose reduced to 400 mg daily after 3 weeks because of fatigue, weight loss, and anorexia • Continued for 2 months but developed worsening pain and rising AFP, prompting restaging imaging Tumors found on the left lobe, and the right lobe masses are larger Extension of PVT
- 12. Therapeutic Options in HCC: A Patient Casebook for Physicians AFP: alpha-fetoprotein; Alb: albumin; BCLC: Barcelona Clinic Liver Cancer staging system; Bili: bilirubin; Cr: creatinine; ECOG-PS: Eastern Cooperative Oncology Group performance status; Hb: hemoglobin; HTN: hypertension; INR: international normalized ratio; MCV: mean corpuscular volume; NASH: nonalcoholic steatohepatitis; PLT: platelets; PMD: primary medical doctor; T2DM: type 2 diabetes mellitus; TACE: transarterial chemoembolization; TKI: tyrosine kinase inhibitor 1. Galle PR et al. J Hepatol. 2018;69:182-236. 2. Ikai I et al. Cancer. 2004;101:796-802. 3. Vauthey JN et al. J Clin Oncol. 2002;20:1527-1536. 4. Shi M et al. Ann Surg. 2007;245:36-43. 5. Katz SC et al. Ann Surg. 2009;249:617-623. 6. Tabrizian P et al. Ann Surg. 2015;261:947-855. 7. Bruix J et al. Lancet Oncol. 2015;16:1344-1354. 8. https://clinicaltrials.gov/ct2/show/NCT03383458. Accessed January 23, 2020. 9. https://clinicaltrials.gov/ct2/show/NCT03867084. Accessed January 23, 2020. 10. https://clinicaltrials.gov/ct2/show/NCT03847428. Accessed January 23, 2020. 11. https://clinicaltrials.gov/ct2/show/NCT04102098. Accessed January 23, 2020. 12. Marrero JA et al. Hepatology. 2018;68:723-750. 13. https://clinicaltrials.gov/ct2/show/NCT03778957. Accessed January 23, 2020. 14. Llovet JM et al. N Engl J Med. 2008;359:378-390. 15. Kudo M et al. Lancet. 2018;391:1163-1173. 16. Cheng A-L et al. ESMO Asia 2019. Abstract LBA3. 17. https://clinicaltrials.gov/ct2/show/NCT03713593. Accessed January 23, 2020. 18. https://clinicaltrials.gov/ct2/show/NCT03755791. Accessed January 23, 2020. 19. https://clinicaltrials.gov/ct2/show/ NCT04039607. Accessed January 23, 2020. 20. https://clinicaltrials.gov/ct2/show/NCT03298451. Accessed January 23, 2020. 21. Bruix J et al. Lancet. 2017;389:56-66. 22. Abou-Alfa G et al. N Engl J Med. 2018;379:54-63. 23. Zhu AX et al. Lancet Oncol. 2019;20:282-296. 24. El-Khoueiry AB et al. Lancet. 2017;389:2492-2502. 25. Zhu AX et al. Lancet Oncol. 2018;19:940-952. PRACTICE AID Access the activity, “Developing a Therapeutic Game Plan for the Management of Hepatocellular Carcinoma: Expert Insights From the Patient CaseBook,” at PeerView.com/BXY40 Case 5 Consider Checkpoint Inhibitor Immunotherapy as Second- or Third-Line Therapy Previously Treated Advanced (BCLC C) HCC (Cont’d)24,25 When to consider checkpoint inhibitor therapy as a treatment option: • Patients who have not received immunotherapy in first line • If rapid progression and/or intolerance to TKI first-line therapy • If increasing degrees of hepatic dysfunction Prospective cohort and retrospective case series show acceptable safety and efficacy of nivolumab in patients with Child–Pugh B HCC • If contraindications to antiangiogenic therapy (eg, nonhealing wounds, active venous thromboembolism, bleeding complications)
