The Emerging Role of ADCs in Lung Cancer Treatment
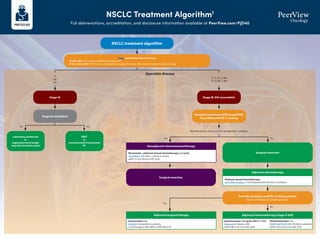
- 1. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PJD40 NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Stage IA Surgical candidate? Lobectomy (preferred) or Segmentectomy/wedge resection (in select cases) SBRT or conventionally fractionated RT Surgical resection Consider mutation and PD-L1 testing results EGFR ex19del/ex21 L858R present? Surgical resection T1 N0 M0 Operable disease Yes Yes Yes No No No Multidisciplinary discussion for neoadjuvant candidacy Stage IB-IIIA (resectable) Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing T1–2, N1–2, M0 T3–4, N0–1, M0 Neoadjuvant chemoimmunotherapy Nivolumab + platinum-based chemotherapy x 3 cycles CheckMate -816: Nivo + chemo vs chemo mEFS: 31.6 vs 20.8 mo (HR, 0.63) Adjuvant chemotherapy Platinum-based chemotherapy LACE Meta-analysis: 5-y OS improvement of 5.4% vs no chemo Adjuvant immunotherapy (stage II-IIIA) Atezolizumab x 16 cycles (PD-L1 1%) IMpower010: Atezo vs BSC mDFS: NR vs 35.3 mo (HR, 0.66) Adjuvant targeted therapy Osimertinib x 3 y ADAURA: Osimertinib vs placebo 2-y DFS (stage II-IIIA): 90% vs 44% (HR, 0.17) Pembrolizumab x 1 y PEARLS/KEYNOTE-091 :Pembro vs placebo mDFS: 53.6 vs 42.0 mo (HR, 0.76)
- 2. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PJD40 Stage IIIA (unresectable) or IIIB/C Definitive chemoradiation → durvalumab Concurrent platinum-based chemotherapy and radiation with consolidation durvalumab PACIFIC: Durvalumab vs placebo mPFS: 16.8 vs 5.6 mo (HR, 0.52) BRAF V600E Dabrafenib + trametiniba BRF113928: Dabrafenib + trametinib single arm ORR: 64% (95% CI, 46–79) 2nd line: KRAS G12C Sotorasib CodeBreaK100: Sotorasib single arm ORR: 37.1% (95%CI, 29-46); mPFS: 6.8 mo ALK Alectiniba ALEX: Alectinib vs crizotinib 1-y PFS: 68.4% vs 48.7% (HR, 0.47) Brigatiniba ALTA-1L: Brigatinib vs crizotinib mPFS: 24 vs 11.1 mo (HR, 0.48) Lorlatiniba CROWN: Lorlatinib vs crizotinib mPFS: NR vs 9.3 mo, (HR, 0.28); 1-y PFS: 78% vs 39% Ceritinib ASCEND-4: Ceritinib vs chemo mPFS: 16.6 vs 8.1 mo (HR, 0.55) Crizotinib PROFILE 1007: Crizotinib vs chemo mPFS: 7.7 vs 3 mo (HR, 0.49) NTRK Larotrecteniba Entrectiniba ALKA/STARTRK: Entrectinib single arm ORR: 70% (NSCLC) RET Selpercatiniba LIBRETTO-001: Selpercatinib single arm ORR: 64%; mDOR: 17.5 mo Pralsetiniba ARROW: Pralsetinib single arm ORR: 61% (95% CI, 50–71) 2nd line: EGFR (ex20) Amivantamab CHRYSALIS: Amivantamab single arm CBR: 74% (95%CI, 63-83); mPFS: 8.3 mo Mobocertinib AP32788-15-101: Mobocertinib single arm DCR: 78% (95% CI, 69-85); mPFS: 7.3 mos ROS1 Crizotiniba PROFILE 1001: Crizotinib single arm ORR: 72% (95% CI, 58–84) Entrectiniba ALKA STARTRK: Entrectinib single arm ORR: 67.1%; mPFS: 19 mo Ceritinib YONSEI: Ceritinib single arm ORR: 67% (95% CI, 48–81) EGFR (ex19 del or L858R) Osimertiniba FLAURA: Osimertinib vs erlotinib/gefitinib mPFS: 18.9 vs 10.2 mo (HR, 0.46) Erlotinib EURTAC: Erlotinib vs chemo mPFS: 9.7 vs 5.2 mo (HR, 0.37) Afatinib LUX-Lung 3: Afatinib vs cis/pemetrexed mPFS: 13.6 vs 6.9 mo (HR, 0.47) Gefitinib IFUM: Gefitinib single arm mPFS: 9.7 mo Dacomitinib ARCHER 1050: Dacomitinib vs geftinib mOS: 34.1 vs 27 mo (HR, 0.75) Erlotinib + ramucirumab RELAY: Erlotinib + ramucirumab vs elotinib mPFS: 19.4 vs 12.4 mo (HR, 0.59) Erlotinib + bevacizumab ARTEMIS-CTONG1509: Erlotinib + bevacizumab vs erlotinib mPFS: 17.9 vs 11.2 mo (HR, 0.55) MET (exon 14) Capmatiniba GEOMETRY mono-1: Capmatinib single arm mPFS: 12.4 mo Tepotiniba VISION: Tepotinib single arm mPFS: 8.5–11 mo 2nd line: HER2 Trastuzumab deruxtecan DESTINY-Lung01 T-DXd single arm ORR: 55% (95% CI, 44-65); mPFS: 8.2 mo T1-2, N2–3, M0 T3, N1–3, M0 T4, N0–3, M0 Tx Nx M1 Actionable mutation detected • EGFR (ex19, ex20ins) • ALK • ROS1 • BRAF V600E • RET • MET (ex14) • HER2 • NTRK1/2/3 • KRAS G12C Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Please see the next page for recommendations if no actionable mutation is detected Stage IV KRYSTAL-1: Adagrasib single arm ORR: 43% (95% CI, 34-53); mDOR: 8.5 mo
- 3. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/PJD40 a Denotes NCCN-preferred regimens. 1. Created by Aakash Desai, MBBS, MPH, and Matthew Ho, MD, PhD. Used with permission from the authors. PD-L1 1% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52), 12-mo; OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycle) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) IMMUNOTHERAPY MONOTHERAPY Pembrolizumab KEYNOTE-042: Pembro vs plat-based chemo mOS: 16.7 vs 12.1 mo (HR, 0.81) Ramucirumab + docetaxela REVEL: Ram/docetaxel vs docetaxel; mOS: 10.5 vs 9.1 mo (HR, 0.86) Docetaxela TAX320: Docetaxel vs vinorelbine/ifosfamide; 1-y OS: 32% vs 19% Gemcitabine DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo OS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) PD-L1 1%-49% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab+chemotherapya (carboplatin+paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150 : Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) • Cemiplimab + chemotherapy (carboplatin + pemetrexed) EMPOWER-Lung 3: Cemi + chemo vs chemo mOS: 21.9 vs 13 mo (HR, 0.7) PD-L1 50% IMMUNOTHERAPY MONOTHERAPY Pembrolizumaba KEYNOTE-024: Pembro vs platinum-based chemo mPFS: 10.3 vs 6 mo (HR, 0.50) Atezolizumaba IMpower110: Atezo vs platinum-based chemo mOS: 20.1 vs 13.1 mo (HR, 0.59) Cemiplimaba EMPOWER-Lung1: Cemi vs platinum-based chemo mPFS: 8.2 vs 5.7 mo; mOS: NR vs 14.2 mo (HR, 0.57) IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + pemetrexed) KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) No actionable mutation detected (stratify based on PD-L1 staining %) Second-line therapy
- 4. The Emerging Role of ADCs in Lung Cancer Full abbreviations, accreditation, and disclosure information available at PeerView.com/PJD40 1. Fu Z et al. Signal Transduct Target Ther. 2022;7:93. 2. Desai A et al. Lung Cancer. 2022;163:96-106. 3. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-fam-trastuzumab-deruxtecan-nxki-her2-mutant-non-small-cell-lung. Antibody–Drug Conjugates (ADCs): What Are They?1 Landscape of ADCs Under Study in Lung Cancer2 New and First FDA Approval of an ADC for HER2-Mutant NSCLC3 Target antigen Antibody Guidance system for cytotoxic drugs Bridge between antibody and drugs to control the release of drugs inside cancer cells Linker Cytotoxic drug Warhead for destroying cancer cells Recognition of target cancer cells Key functions NSCLC SCLC Ado-Trastuzumab Emtansine (T-DM1) Trastuzumab Deruxtecan (T-DXd) Patritumab Deruxtecan (HER3-DXd) Datopotamab Deruxtecan (Dato-DXd) Sacituzumab Govitecan (SN-38) SAR408701 (DM4) Telisotuzumab Vedotin (MMAE) Rovalpituzumab Tesirine (PYRROLO-BZD) Lorvotuzumab Mertansine (DM1) Sacituzumab Govitecan (SN-38) HER2 HER3 HER2 TROP2 TROP2 CEACAM5 C-MET DLL-3 CD-56 1. Drug internalized 2. ADC in endosome 3. Drug release from ADC in lysosome CYTOTOXIC EFFECT • On August 11, 2022, the FDA granted accelerated approval to fam-trastuzumab deruxtecan-nxki for adult patients with unresectable or metastatic NSCLC whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy • This is the first drug approved for HER2-mutant NSCLC, and also the first ADC approved in NSCLC • FDA also approved the Oncomine™ Dx Target Test (tissue) and the Guardant360® CDx (plasma) as companion diagnostics for trastuzumab deruxtecan; if no mutation is detected in a plasma specimen, the tumor tissue should be tested
