Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM
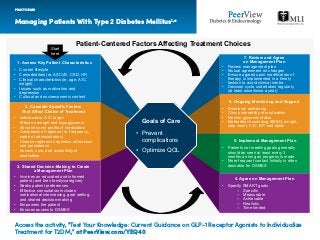
- 1. Managing Patients With Type 2 Diabetes Mellitus1,a PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 1. Assess Key Patient Characteristics • Current lifestyle • Comorbidities (ie, ASCVD, CKD, HF) • Clinical characteristics (ie, age, A1C, weight) • Issues such as motivation and depression • Cultural and socioeconomic context 2. Consider Specific Factors That Affect Choice of Treatment • Individualize A1C target Effect on weight and hypoglycemia • Adverse event profile of medication • Complexity of regimen (ie, frequency, mode of administration) • Choose regimen to optimize adherence and persistence • Access, cost, and availability of medication 3. Shared Decision-Making to Create a Management Plan • Involves an educated and informed patient (and their family/caregiver) • Seeks patient preferences • Effective consultation includes motivational interviewing, goal setting, and shared decision-making • Empowers the patient • Ensures access to DSMES 7. Review and Agree on Management Plan • Review management plan • Mutual agreement on changes • Ensure agreed upon modification of therapy is implemented in a timely fashion to avoid clinical inertia • Decision cycle undertaken regularly (at least once/twice a year) 6. Ongoing Monitoring and Support • Emotional well-being • Check tolerability of medication • Monitor glycemic status • Biofeedback including SMBG, weight, step count, A1C, BP, and lipids 5. Implement Management Plan • Patients not meeting goals generally should be seen at least every 3 months as long as progress is made • More frequent contact initially is often desirable for DSMES 4. Agree on Management Plan • Specify SMART goals – Specific – Measurable – Achievable – Realistic – Time limited Goals of Care • Prevent complications • Optimize QOL Patient-Centered Factors Affecting Treatment Choices Start here
- 2. Managing Patients With Type 2 Diabetes Mellitus1,a PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 First-line therapy is metformin and comprehensive lifestyle modifications (including weight management and physical activity) Consider independently of baseline A1C or individualized A1C target ASCVD predominates If A1C above target HF or CKD predominates If A1C above target Indicators of high risk or established ASCVD, CKD, or HF Compelling need to minimize hypoglycemia If A1C above target If A1C above target If A1C above target If A1C above target If A1C above target Cost is a major issue If A1C above target If A1C above target To avoid therapeutic inertia, reassess and modify treatment regularlyNO • Established ASCVD • Indicators of high ASCVD risk (age >55 years with coronary, carotid, or lower extremity artery stenosis >50% or LVH) Preferably GLP-1 RA with proven CVD benefit or SGLT2i with proven CVD benefit if eGFR adequate If further intensification is required or patient is now unable to tolerate GLP-1 RA and/or SGLT2i, choose agents demonstrating CV safety: • For patients on GLP-1 RA, considering adding SGLT2i with proven CVD benefit; DPP-4i if not on GLP-1 RA; basal insulin; TZD; or SU • Particularly HFrEF (LVEF <45%) • CKD: specifically eGFR 30-60 mL/min/1.73 m2 or UACR >30 mg/g, particularly UACR >300 mg/g Preferably SGLT2i with evidence of reducing HF/CKD progression in CVOTs if eGFR adequate or If SGLT2i not tolerated/ contraindicated or if eGFR less than adequate add GLP-1 RA with proven CVD benefit Avoid TZD in the setting of HF; choose agents demonstrating CV safety: for patients on SGLT2i, consider adding GLP-1 RA with proven CVD benefit; DPP-4i (not saxagliptin) in the setting of HF (not on GLP-1 RA); basal insulin; or SU DPP-4i GLP-1 RA SGLT2i TZD SGLT2i or TZD SGLT2i or TZD GLP-1 RA or DPP-4i or TZD SGLT2i or DPP-4i or GLP-1 RA Continue with addition of other agents as outlined above Consider the addition of SU or basal insulin: • Choose later-generation SU with lower risk of hypoglycemia • Consider basal insulin with lower risk of hypoglycemia GLP-1 RA with good efficacy for weight loss SGLT2i SGLT2i GLP-1 RA with good efficacy for weight loss If quadruple therapy required, or SGLT2i and/or GLP-1 RA not tolerated/ contraindicated, use regimen with lowest risk of weight gain Preferably DPP-4i (if not on GLP-1 RAs) based on weight neutrality If DPP-4i not tolerated/contraindicated or patient already on GLP-1 RA, cautious addition of SU, TZD, or basal insulin SU TZD TZD SU • Insulin therapy: basal insulin with lowest acquisition cost or • Consider DPP-4i or SGLT2i with lowest acquisition cost ADA: Treatment Algorithm YES Compelling need to minimize weight gain or promote weight loss If A1C above individualized target proceed as below
- 3. Managing Patients With Type 2 Diabetes Mellitus1,a PRACTICE AID a These algorithms have been adapted from the original source; see full article for additional information on these recommendations. ADA: American Diabetes Association; ASCVD: atherosclerotic cardiovascular disease; BP: blood pressure; CKD: chronic kidney disease; CVD: cardiovascular disease; CVOTs: cardiovascular outcomes trials; DPP-4i: dipeptidyl peptidase-4 inhibitor; DSMES: Diabetes Self-Management Education and Support; eGFR: estimated glomerular filtration rate; FPG: fasting plasma glucose; HFrEF: heart failure with reduced ejection fraction; LVEF: left ventricular ejection fraction; LVH: left ventricular hypertrophy; NPH: neutral protamine Hagedorn; SGLT2i: sodium-glucose cotransporter 2 inhibitor; SMBG: self-monitored blood glucose; SU: sulfonylurea; TZD: thiazolidinediones; UACR: urine albumin-to-creatinine ratio. 1. American Diabetes Association. Diabetes Care. 2020;43(suppl 1):s1-s212. Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 To avoid clinical inertia reassess and modify treatment regularly (3-6 months) ADA: Injectable Therapy Treatment Algorithm Consider GLP-1 RA in most patients prior to insulin INITIATION:Initiate appropriate starting dose for agent selected (varies within class) TITRATION:Gradual titration to maintenance dose (varies within class) If injectable therapy is needed to reduce A1C Add basal insulin Choice of basal insulin should be based on patient-specific considerations, including cost Add basal analog or bedtime NPH insulin INITIATION:Start 10 IU a day or 0.1-2.0 IU/kg a day TITRATION: • Set up FPG target • Choose evidence-based titration algorithm (eg, increase 2 units every 3 days to reach FPG target without hypoglycemia) • For hypoglycemia, determine cause; if no clear reason, lower dose by 10%-20% Add prandial insulin Usually one dose with the largest meal or meal with greatest FPG excursion; prandial insulin can be dosed individually or mixed with NPH as appropriate INITIATION: • 4 IU a day or 10% of basal insulin dose • If A1C <8% (64 mmol/mol) consider lowering the basal dose by 4 IU a day or 10% of basal dose TITRATION: • Increase dose by 1-2 IU or 10%-15% twice weekly • For hypoglycemia, determine cause; if no clear cause lower corresponding dose by 10%-20% If above A1C target If already on GLP-1 RA or if GLP-1 RA not appropriate or insulin preferred If above A1C target despite adequately titrated basal analog or bedtime NPH or once basal dose >0.5 IU/kg or FPG at target If on bedtime NPH, consider converting to twice-daily NPH regimen INITIATION:Total dose = 80% of current bedtime dose; two-thirds given in the morning and one-third given at bedtime TITRATION:Titrate based on individualized needs If above A1C target Proceed to full basal-bolus regimen (ie, basal insulin and prandial insulin with each meal) Stepwise additional injections of prandial insulin (ie, two, then three additional injections) Consider self-mixed/split insulin regimen INITIATION:Total NPH dose = 80% of current NPH dose; two-thirds given before breakfast, one-third TITRATION:Titrate each component of the regimen based on individualized needs Consider twice daily premix insulin regimen INITIATION:Usually unit per unit at the same total insulin dose, but may TITRATION:Titrate based on individualized needs If above A1C target given before dinner; add 4 IU of short/rapid-acting insulin to each injection or 10% of reduced NPH dose require adjustment to individual needs
- 4. Counseling Patients About Treatments for Type 2 Diabetes Mellitus (T2DM) PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 How effective at reducing BG? Might prevent heart attacks? Might increase the risk of HF? Might slow the loss of renal function? Suppresses appetite? Might cause weight loss? Hypoglycemia is a common AE? Nausea is a common AE? Administered as an injection? Can be given once weekly? Generic available? Highly Some do No Some may Yes Yes No Yes Usually Some can Not yet Highly No No No No No Yes No Always No Not yet Highly No No No No No Yes No Never No Yes Highly Some might Yes No No No No No Never No Yes Moderately Some do No Some may No Yes No No Never No Not yet Moderately No Some do No No No No No Never No Not yet Characteristic GLP-1 RAs Basal insulin DPP-4is SGLT2is TZDs SUs Adverse Events Associated With T2DM Treatments1,2
- 5. Counseling Patients About Treatments for Type 2 Diabetes Mellitus (T2DM) PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 • Twice-daily injection • 60 minutes before morning and evening meals Exenatide BID Liraglutide • Daily injection • Any time of day Lixisenatide • Daily injection • 60 minutes before first meal of the day Semaglutide (oral) • Daily oral tablet • 30 minutes before morning meal GLP-1 RAs: Frequency of Administration and Dosing With Respect to Meals2-6 Dulaglutide • Weekly injection • Any time of the day Exenatide ER • Weekly injection • Any time of day Semaglutide (injectable) • Weekly injection • Any time of day Shorter-acting agents Longer-acting agents
- 6. Counseling Patients About Treatments for Type 2 Diabetes Mellitus (T2DM) a Initial dose is intended to decrease GI-related AEs only, it does not provide effective glycemic control. BG: blood glucose; BID: twice daily; ER: extended release; GLP-1 RAs: glucagon-like peptide-1 receptor agonists; IR: immediate release; SGLT2is: sodium glucose cotransporter 2 inhibitors; SUs: sulfonylureas; TZDs: thiazolidinediones. 1. American Diabetes Association. Diabetes Care. 2020;43(suppl 1):S1-S212. 2. https://www.accessdata.fda.gov/scripts/cder/daf/. 3. Hinnen D. Diabetes Spectr. 2017;30:202-210. 4. Abd El Aziz MS et al. Diabetes Obes Metab. 2017;19:216-227. 5. Kalra S et al. Diabetes Ther. 2019;10:1645-1717. 6. Bain SC et al. Diabetes Obes Metab. 2019;21:499-508. PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 GLP-1 RAs: Practical Aspects2 Frequency Starting dose Next higher dose Next higher dose Screw on needle Separate prescription for needle Autoinjector Daily 10 mcg 20 mcg — — Lixisenatide Daily 3 mga 7 mg 14 mg — — — Semaglutide (oral) Auto Tray /pen Weekly 2 mg — — — — — — — — Exenatide ER Twice daily 5 mcg 10 mcg — — Exenatide BID Daily 0.6 mga 1.2 mg 1.8 mg — Liraglutide Weekly 0.75 mg 1.5 mg — — — Dulaglutide Weekly 0.25 mga 0.5 mg 1.0 mg — — Semaglutide (injectable) = FDA indication for CV risk reduction
- 7. GLP-1 RAs: Prescribing Information Highlights1 PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 Adjunct to diet and exercise to improve glycemic control in adults with T2DM Adjunct to diet and exercise to improve control in patients aged ≥10 years Reduce risk of MACE in adults with T2DM and established CVD Reduce risk of MACE in adults with T2DM and CV risk factors Exenatide BID Liraglutide DulaglutideLixisenatide Semaglutide (oral) Exenatide ER Semaglutide (injectable) Indications
- 8. GLP-1 RAs: Prescribing Information Highlights1 PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 Do not use in patients with a history of hypersensitivity to this agent or any product components Personal or family history of MTC or patients with MEN2 History of drug- induced immune- mediated thrombocytopenia from exenatide products Exenatide BID Liraglutide DulaglutideLixisenatide Semaglutide (oral) Exenatide ER Semaglutide (injectable) Contraindications
- 9. GLP-1 RAs: Prescribing Information Highlights1 a Liraglutide is currently the only GLP-1 RA with a pediatric indication from the US FDA. b Should not be used in patients with severe renal impairment. c Should not be used in patients with end-stage renal disease. d Not recommended in patients with end-stage renal disease. e Not recommended for use in patients with eGFR <45 mL/min/1.73m2 . AKI: acute kidney injury; CVD: cardiovascular disease; DR: diabetic retinopathy; ER: extended release; GLP-1 RAs: glucagon-like peptide-1 receptor agonists; MACE: major adverse cardiovascular events; MEN2: multiple endocrine neoplasia type 2; MTC: medullary thyroid carcinoma; T2DM: type 2 diabetes. 1. https://www.accessdata.fda.gov/scripts/cder/daf/. PRACTICE AID Access the activity, “Test Your Knowledge: Current Guidance on GLP-1 Receptor Agonists to Individualize Treatment for T2DM,” at PeerView.com/YEQ40 Exenatide BID Liraglutide DulaglutideLixisenatide Semaglutide (oral) Exenatide ER Semaglutide (injectable) Warnings and Precautions Pancreatitis Hypoglycemia with insulin secretagogue or insulin Hypoglycemia in pediatric patients, regardless of other agents Hypersensitivity, including anaphylaxis, angioedema AKI or caution in patients with renal impairment Severe GI disease (not studied) Diabetic retinopathy (monitor patients with history of DR) Drug-induced immune- mediated thrombocytopenia Immunogenicity Injection-site reactions Acute gallbladder disease edb,c Thyroid C-cell tumors (boxed warning) a
