Ramucirumab imc 1121 b ottobre 2010
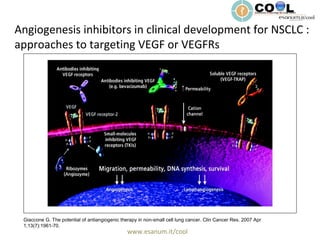
- 1. www.esanum.it/cool Giaccone G. The potential of antiangiogenic therapy in non-small cell lung cancer. Clin Cancer Res. 2007 Apr 1;13(7):1961-70. Angiogenesis inhibitors in clinical development for NSCLC : approaches to targeting VEGF or VEGFRs
- 2. www.esanum.it/cool s-s VEGF- B167 VEGF- B186 PlGF- 1,2 VEGF- A121 VEGF- A145 VEGF- A165 VEGF- A189 VEGF- A206 VEGF- C VEGF- D VEGFR1 (Flt-1) VEGFR2 (Flk-1/KDR) VEGFR3 (Flt-4) NRP-1 s-s NRP-2 IMC-1121B Vasculogenesis Angiogenesis Angiogenesis Lymphangiogenesis Ramucirumab (IMC-1121B), a fully human IgG1 monoclonal antibody, targets VEGFR2 www.researchvegf.com Youssoufian H, Hicklin DJ, Rowinsky EK. Review: monoclonal antibodies to the vascular endothelial growth factor receptor-2 in cancer therapy. Clin Cancer Res. 2007 Sep 15;13(18 Pt 2):5544s-5548s.
- 3. www.esanum.it/cool Studies of ramucirumab in NSCLC Ongoing trials Study ID Ph Eligibility/Line Arm(s) N (projected) NCT00735696 II Stage IIIB or IV (AJCC 6th ed.), first line IMC-1121B + paclitaxel + carboplatin 40 NCT01160744 II Stage IV , first line IMC-1121B + Premetrexed + Carboplatin or Cisplatin Premetrexed + Carboplatin or Cisplatin IMC-1121B + Gemcitabine + Carboplatin or Cisplatin Gemcitabine + Carboplatin or Cisplatin 280 NCT01168973 III Stage IV, second line Docetaxel + Ramucirumab Docetaxel + Placebo 1156 www.clinicaltrials.gov (accessed on Aug 28th , 2010)
- 4. www.esanum.it/cool NCT00735696 - Phase 2 study in NSCLC (1st -line: IMC-1121B + paclitaxel/carboplatin) Preliminary results shown at ASCO 2010 Camidge et al. ASCO (Poster) June 2010 D. R. Camidge, M. S. Ballas, S. Dubey, M. Haigentz, P. J. Rosen, J. F. Spicer, H. J. West, G. D. Shah, H. Youssoufian, A. C. Mita. A phase II, open-label study of ramucirumab (IMC-1121B), an IgG1 fully human monoclonal antibody (MAb) targeting VEGFR-2, in combination with paclitaxel and carboplatin as first-line therapy in patients (pts) with stage IIIb/IV non-small cell lung cancer (NSCLC). J Clin Oncol 28:15s, 2010 (suppl; abstr 7588) Patient Characteristics (n=35) Gender: male 35% , female 65% Performance Status: ECOG 0 38% , ECOG 1 62% Histology: adenocarcinoma w focal squamous 3%, adenocarcinoma97% Ramucirumab-related AEs in at Least 10% of Pts Across All Grades (N=35)
- 5. www.esanum.it/cool Waterfall Plot of Percent Change of Target Lesions from Baseline per RECIST v1.0 (from 28 pts*) Response by RECIST v1.0 (N=29 evaluable pts) NCT00735696 - Phase 2 study in NSCLC (1st -line: IMC-1121B + paclitaxel/carboplatin) Preliminary results shown at ASCO 2010 (cont’d) Camidge et al. ASCO (Poster) June 2010 D. R. Camidge, M. S. Ballas, S. Dubey, M. Haigentz, P. J. Rosen, J. F. Spicer, H. J. West, G. D. Shah, H. Youssoufian, A. C. Mita. A phase II, open-label study of ramucirumab (IMC-1121B), an IgG1 fully human monoclonal antibody (MAb) targeting VEGFR-2, in combination with paclitaxel and carboplatin as first-line therapy in patients (pts) with stage IIIb/IV non-small cell lung cancer (NSCLC). J Clin Oncol 28:15s, 2010 (suppl; abstr 7588) ♦ This pt had PD based on a new lesion ♠These 2 pts had an unconfirmed PR because confirmatory scans have not yet been performed * This plot was based on 28 pts with evaluable target lesions. There was 1 pt who had no follow-up target lesion measurements that were available in the clinical database and therefore is not presented in this plot; however, the Investigator considered the pt as SD at the time of this report.
- 6. www.esanum.it/cool •NSCLC stage IV progressing during or after one prior first-line platinum-based chemotherapy with or without maintenance therapy •All histologies •ECOG PS <=1 R A N D O M I Z E Docetaxel 75 mg/m2 q3w + IMC-1121B 10 mg/kg q3w Treat until PD, toxicity, consent withdrawal 2 1 Docetaxel 75 mg/m2 q3w + Placebo Randomized Phase 3 Study in NSCLC Docetaxel +/- IMC-1121B www.clinicaltrials.gov ; NCT01168973; accessed on Aug 28th , 2010