lecture18.pdf
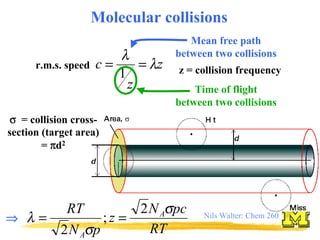
- 1. Nils Walter: Chem 260 σ σ σ σ = collision cross- section (target area) = π π π πd2 Molecular collisions r.m.s. speed z z c λ λ = = 1 Mean free path between two collisions Time of flight between two collisions z = collision frequency RT pc N z p N RT A A σ σ λ 2 ; 2 = = ⇒ ⇒ ⇒ ⇒
- 2. Nils Walter: Chem 260 In reality: Gases have attractive and repulsive forces Lennard-Jones 6-12 potential ⇒ ⇒ ⇒ ⇒ e.g., CO2 At high T: perfect gas isotherms At low T: liquefaction
- 3. Nils Walter: Chem 260 The critical point: Gas and liquid density become equal ⇒ ⇒ ⇒ ⇒ Application: Extraction of caffeine from coffee with supercritical CO2 At critical point (for water 373oC @ 218 atm!) the boundary is lost Heating a liquid in a container
- 4. Nils Walter: Chem 260 Describing the deviation from the perfect gas Introducing the compression factor Z: molar volume of real gas molar volume of perfect gas Z= RT pV p RT V V V Z m m perfect m m = = = Z = 1 ⇒ perfect gas Z < 1 ⇒ molecules cluster, attractive forces are dominant Z > 1 ⇒ molecules repel each other, repulsive forces are dominant
- 5. Nils Walter: Chem 260 The virial equation of state Empirically: ... 1 2 + + + = m m V C V B Z virial coefficients B > 0 ⇒ Z > 1, e.g., H2 B < 0 ⇒ Z < 1, e.g., CH4, NH3 C > 0 ⇒ Z > 1 at high pressure (Vm small) + + + = ... 1 2 2 V C n V nB V nRT p RT pV Z m = ⇒ ⇒ ⇒ ⇒ and very accurate
- 6. Nils Walter: Chem 260 Physically more palpable: The van der Waals equation [Johannes van der Waals 1873] ( ) nRT nb V V an p = − + 2 2 ⇒ ⇒ ⇒ ⇒ molecules have a non-zero volume ⇒ ⇒ ⇒ ⇒ additional volume needed: nb molecules have attractive forces ⇒ ⇒ ⇒ ⇒ reduction in exerted pressure: a(n/V)2 [molecules strike less frequently and with reduced force] Lennard-Jones 6-12 potential
- 7. Nils Walter: Chem 260 Plotting the van der Waals equation: In reasonable agreement with reality only the van der Waals loops are unrealistic in 3D p V T
- 8. Nils Walter: Chem 260 Liquefaction of real gases: The Joule-Thomson effect Real gases have attractive forces Linde refrigerator ⇒ ⇒ ⇒ ⇒ if they are allowed to expand through a throttle without outside heat entering (“adiabatic” process) they will use their kinetic (heat) energy to escape each other’s attraction ⇒ ⇒ ⇒ ⇒ they will cool down