Chemiosmotic theory
•Download as PPTX, PDF•
3 likes•3,944 views
ATP synthase, Protons, acidity, F1 and F0 domain
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Biological oxidation (part - III) Oxidative Phosphorylation

Biological oxidation (part - III) Oxidative Phosphorylation
Electron transport chain( Oxidative phosphorylation)

Electron transport chain( Oxidative phosphorylation)
Oxidative phosphorylation and electron transport chain

Oxidative phosphorylation and electron transport chain
Oxidative phosphorylation and photophosphorylation

Oxidative phosphorylation and photophosphorylation
MITOCHONDRIA ,STRUCTURE ,Mt DNA ,PROTEIN TRANSPORT,ETC,OXIDATIVE PHOSPHORYLATION

MITOCHONDRIA ,STRUCTURE ,Mt DNA ,PROTEIN TRANSPORT,ETC,OXIDATIVE PHOSPHORYLATION
Similar to Chemiosmotic theory
Chemiosmotic regeneration of ATP during respiration, fo f1 model of ATpase & ...

Chemiosmotic regeneration of ATP during respiration, fo f1 model of ATpase & ...gohil sanjay bhagvanji
Similar to Chemiosmotic theory (20)
Chemiosmotic regeneration of ATP during respiration, fo f1 model of ATpase & ...

Chemiosmotic regeneration of ATP during respiration, fo f1 model of ATpase & ...
Biochem Respiratory chain and Oxidative phosphorylation

Biochem Respiratory chain and Oxidative phosphorylation
BIOLOGICAL OXIDATION L4 (Oxidative phosphorylation)

BIOLOGICAL OXIDATION L4 (Oxidative phosphorylation)
The direct energy source that drives ATP synthesis during respiratory.pdf

The direct energy source that drives ATP synthesis during respiratory.pdf
3- Electron Transport Chain pdf which is related to biochemistry

3- Electron Transport Chain pdf which is related to biochemistry
6_2018_12_21!01_04_11_AM ELECTRON TRANSPORT AND OXIDATIVE PHOSPHORILATION.ppt

6_2018_12_21!01_04_11_AM ELECTRON TRANSPORT AND OXIDATIVE PHOSPHORILATION.ppt
More from mariagul6
More from mariagul6 (20)
oxidation of alpha, beta fatty acid and unsaturated fatty acid 

oxidation of alpha, beta fatty acid and unsaturated fatty acid
Regulation of translation or gene expression lec 33

Regulation of translation or gene expression lec 33
Metabolism of essential and non essential amino acids 20

Metabolism of essential and non essential amino acids 20
Recently uploaded
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...Association for Project Management
Mehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
Recently uploaded (20)
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...
ICT role in 21st century education and it's challenges.

ICT role in 21st century education and it's challenges.
Food safety_Challenges food safety laboratories_.pdf

Food safety_Challenges food safety laboratories_.pdf
Salient Features of India constitution especially power and functions

Salient Features of India constitution especially power and functions
Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...

Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...
Unit-IV; Professional Sales Representative (PSR).pptx

Unit-IV; Professional Sales Representative (PSR).pptx
Chemiosmotic theory
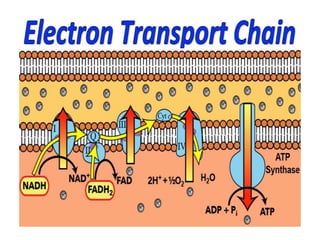
- 3. The transfer of electrons down the electron transport chain is energetically favored because NADH is a strong electron donor and molecular oxygen is an avid electron acceptor. However, the flow of electrons from NADH to oxygen does not directly result in ATP synthesis. A) Chemiosmotic hypothesis The chemiosmotic hypothesis (also known as the Mitchell hypothesis) explains how the free energy generated by the transport of electrons by the electron transport chain is used to produce ATP from ADP + Pi. 1. Proton pump: Electron transport is coupled to the phosphorylation of ADP by the transport (“pumping”) of protons (H+) across the inner mitochondrial membrane from the matrix to the inter membrane space at Complexes I, III, and IV. This process creates an electrical gradient (with more positive charges on the outside of the membrane than on the inside) and a pH gradient (the outside of the membrane is at a lower pH than the inside. The energy generated by this proton gradient is sufficient to drive ATPirectly result in ATP synthesis.
- 4. synthesis. Thus, the proton gradient serves as the common intermediate that couples oxidation to phosphorylation. 2. ATP synthase: The enzyme complex ATP synthase (Complex V) synthesizes ATP using the energy of the proton gradient generated by the electron transport chain. [Note: It is also called F1/Fo ATPase because the isolated enzyme can catalyze the hydrolysis of ATP to ADP and Pi.] The chemiosmotic hypothesis proposes that after protons have been pumped to the cytosolic side of the inner mitochondrial membrane, they reenter the matrix by passing through a channel in the membrane-spanning domain (Fo ) of Complex V, driving rotation of Fo and, at the same time, dissipating the pH and electrical gradients. Fo rotation causes conformational changes in the extra-membranous F1 domain that allow it to bind ADP + Pi, phosphorylate ADP to ATP, and release ATP.