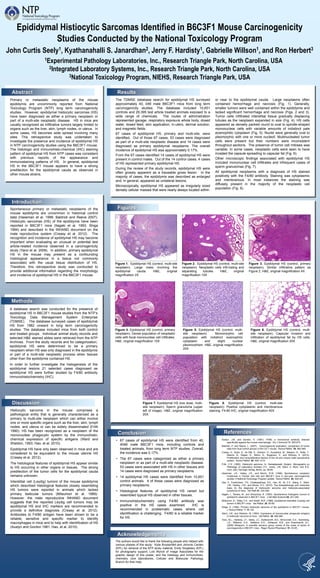
Epididymal Histiocytic Sarcomas in B6C3F1 Mice
- 1. Austyn, J.M. and Gordon, S. (1981). F4/80, a monoclonal antibody directed specifically against the mouse macrophage. Eur J Immunol 11, 805-815. Baldrick, P. and Reeve, L. (2007). Carcinogenicity evaluation: comparison of tumor data from dual control groups in the CD-1 mouse. Toxicol Pathol 35, 562-569. Creasy, D., Bube, A., de Rijk, E., Kandori, H., Kuwahara, M., Masson, R., Nolte, T., Reams, R., Regan, K., Rehm, S., Rogerson, P., and Whitney, K. (2012). Proliferative and nonproliferative lesions of the rat and mouse male reproductive system. Toxicol Pathol 40, 40S-121S. Frith, C.H. (1990). Histiocytic sarcoma. In: Hematopoietic System. Monographs on Pathology of Laboratory Animals (T.C. Jones, J.M. Ward, U. Mohr, and R.D. Hunt, eds.) Springer-Verlag, Berlin, pp. 58-65. Haseman, J.K., Hailey, J.R., and Morris, R.W. (1998). Spontaneous neoplasm incidences in Fischer 344 rats and B6C3F1 mice in two-year carcinogenicity studies: A National Toxicology Program update. Toxicol Pathol 26, 428-441. Hao, X., Fredrickson, T.N., Chattopadhyay, S.K., Han, W., Qi, C-F., Wang, Z., Ward, J.M., Hartley, J.W., and Morse, H.C. (2010). The histopathologic and molecular basis for the diagnosis of histiocyctic sarcoma and histiocyte-associated lymphoma of mice. Vet Path 47, 434-445. Itagaki, I., Tanaka, M., and Shinomiya, K. (1993). Spontaneous histiogenic tumors of epididymis observed in B6C3F1 mice. J Vet Med Science 55, 241-246. Mitsumori, K., Talley, F.A., and Elwell, M.R. (1989). Epididymal interstitial (Leydig) cell tumors in B6C3F1 mice. Vet Pathol 26, 65-69. Shiga, A. (1994). Primary histiocytic sarcoma of the epididymis in B6C3F1 mouse. J Toxicol Pathol 7, 95-102. Ward, J.M., and Sheldon, W. (1993). Expression of mononuclear phagocyte antigens in histiocytic sarcoma of mice. Vet Pathol 30, 560-565. Yano, B.L., Hardisty, J.F., Seely, J.C., Butterworth, B.E., McConnell, E.E., Swenberg, J.A., Williams, G.A., Stebbins, K.E., Gollapudi, B.B., and Eisenbrandt, D.L. (2008) Nitrapyrin: A scientific advisory group review of the mode of action of carcinogenicity in B6C3F1 mice. Regul Toxicol Pharmacol 51, 53-65. Epididymal Histiocytic Sarcomas Identified in B6C3F1 Mouse Carcinogenicity Studies Conducted by the National Toxicology Program John Curtis Seely1, Kyathanahalli S. Janardhan2, Jerry F. Hardisty1, Gabrielle Willson1, and Ron Herbert3 1Experimental Pathology Laboratories, Inc., Research Triangle Park, North Carolina, USA 2Integrated Laboratory Systems, Inc., Research Triangle Park, North Carolina, USA 3National Toxicology Program, NIEHS, Research Triangle Park, USA Spontaneous primary or metastatic neoplasms of the mouse epididymis are uncommon in historical control data (Haseman et al. 1998; Baldrick and Reeve 2007). Histiocytic sarcomas (HS) of the epididymis have been reported in B6C3F1 mice (Itagaki et al. 1993; Shiga 1994) and described in the INHAND document on the male reproductive system (Creasy et al. 2012). The recognition and incidence of epididymal HS may become important when evaluating an unusual or potential test article-related incidence observed in a carcinogenicity study (Yano et al. 2008). In addition, primary epididymal HS in the mouse may present as a confounding histological appearance in a tissue not commonly associated with the usual tissue distribution of HS. Therefore, this retrospective study was conducted to provide additional information regarding the morphology and incidence of epididymal HS in the B6C3F1 mouse. Introduction Figures References A database search was conducted for the presence of epididymal HS in B6C3F1 mouse studies from the NTP’s Toxicology Data Management System Enterprise (TDMSE). The database surveyed cases of epididymal HS from 1982 onward in long term carcinogenicity studies. The database included mice from both control and treated groups. Individual animal study records and selected H&E stained slides were retrieved from the NTP Archives. From the study records and for categorization, epididymal HS were determined to be a primary neoplasm when HS was only diagnosed in the epididymis or part of a multi-site neoplastic process when tissues other than the epididymis contained HS. In order to further investigate the histogenesis of the epididymal lesions 21 selected cases diagnosed as epididymal HS were further studied by F4/80 antibody immunohistochemistry (IHC). Methods Primary or metastatic neoplasms of the mouse epididymis are uncommonly reported from National Toxicology Program (NTP) long term carcinogenicity studies. However, epididymal histiocytic sarcomas (HS) have been diagnosed as either a primary neoplasm or part of a multi-site neoplastic disease. HS in mice are usually recognized as infiltrative tumors largely limited to organs such as the liver, skin, lymph nodes, or uterus. In some cases, HS becomes wide spread involving many sites. This retrospective study was undertaken to investigate the presence and incidence of epididymal HS in NTP carcinogenicity studies using the B6C3F1 mouse. The histologic and immunohisto-chemical (IHC) staining pattern of epididymal HS from NTP cases was consistent with previous reports of the appearance and immunostaining patterns of HS. In general, epididymal HS in the B6C3F1 mouse appears to have a strong predilection for the epididymal cauda as observed in other mouse strains. Abstract • 67 cases of epididymal HS were identified from 40, 4046 male B6C3F1 mice, including controls and treated animals, from long term NTP studies. Overall, the incidence was 0.17%. • The 67 cases were categorized as either a primary neoplasm or as part of a multi-site neoplastic disease. 53 cases were associated with HS in other tissues and 14 cases were diagnosed as primary neoplasms. • 14 epididymal HS cases were identified from 10,651 control animals. 4 of these cases were diagnosed as primary neoplasms. • Histological features of epididymal HS varied but resembled typical HS observed in other tissues. • Immunohistochemistry using F4/80 antibody was positive in the HS cases examined. IHC is recommended in problematic cases where cell identification is challenging. F4/80 is a reliable marker for HS. Conclusion The TDMSE database search for epididymal HS surveyed approximately 40, 046 male B6C3F1 mice from long term carcinogenicity studies. The database included 10,651 controls and 29,395 test article treated animals exposed to a wide range of chemicals. The routes of administration represented gavage, respiratory exposure whole body, dosed water, dosed feed, skin application, in-utero, dermal solution, and magnetic fields. 67 cases of epididymal HS, primary and multi-site, were identified. Out of those 67 cases, 53 cases were diagnosed as part of a multi-site neoplastic disease and 14 cases were diagnosed as primary epididymal neoplasms. The overall incidence of epididymal HS was approximately 0.17%. From the 67 cases identified 14 cases of epididymal HS were present in control males. Out of the 14 control cases, 4 cases of HS represented primary epididymal HS. During the review of the study records, epididymal HS were often grossly apparent as a traceable gross lesion. In the majority of cases, the epididymis was described as enlarged and, in general, appeared as unilateral lesions. Microscopically, epididymal HS appeared as irregularly sized densely cellular masses that were nearly always located within or near to the epididymal cauda. Large neoplasms often contained hemorrhage and necrosis (Fig. 1). Generally, smaller tumors were well contained within the epididymis and lacked significant hemorrhage and necrosis (Figs. 2 and 3). Tumor cells infiltrated interstitial tissue gradually displacing tubules as the neoplasm expanded in size (Fig. 4). HS cells appeared as densely packed round to oval to spindle-shaped mononuclear cells with variable amounts of indistinct pale eosinophilic cytoplasm (Fig. 5). Nuclei were generally oval to pleomorphic with one or more nucleoli. Multinucleated tumor cells were present but their numbers were inconsistent throughout sections. The presence of tumor cell mitoses was variable. In some cases, neoplastic cells were seen to have invaded the capsule spreading to capsular fat (Fig. 6). Other microscopic findings associated with epididymal HS included mononuclear cell infiltrates and infrequent cases of sperm granulomas (Fig. 7). All epididymal neoplasms with a diagnosis of HS stained positively with the F4/80 antibody. Staining was cytoplasmic and membranous. In most instances the staining was diffusely present in the majority of the neoplastic cell population (Fig. 8). Results The authors would like to thank the following people who helped with various phases of this study: Kylie Brockenfelt and Johnnie Carden (EPL) for retrieval of the NTP study material, Emily Singletary (EPL) for photography support, Lois Wyrick of Image Associates for the graphic design of this poster, and the histology and immunohisto- chemistry core laboratories, Cellular and Molecular Pathology Branch for their help. Acknowledgements Histiocytic sarcoma in the mouse comprises a pathological entity that is generally characterized as a primary to multi-site neoplasm which can either involve one or more specific organs such as the liver, skin, lymph nodes, and uterus or can be widely disseminated (Frith 1990). HS has been recognized as a neoplasm of the mononuclear phagocytic system by the immunohisto- chemical expression of specific antigens (Ward and Sheldon, 1993; Hao, et al. 2010). Epididymal HS have only been observed in mice and are considered to be equivalent to the mouse uterine HS (Creasy et al. 2012). The histological features of epididymal HS appear similar to HS occurring in other organs or tissues. The strong predilection of the tumor cells for the epididymal cauda remains unknown. Interstitial cell (Leydig) tumors of the mouse epididymis which described histological features closely resembling HS tumors were reported in animals which lacked primary testicular tumors (Mitsumori et al. 1989). However, the male reproductive INHAND document suggests that the reported Leydig cell tumors may be epididymal HS and IHC markers are recommended to provide a definitive diagnosis (Creasy et al. 2012). Antibodies to F4/80 antigen have been shown to be a reliable, sensitive and specific marker to identify macrophages in mice and to help with identification of HS (Austyn and Gordon 1981; Hao, et al. 2010). Discussion Figure 1. Epididymal HS (control, multi-site neoplasm). Large mass involving the epididymal cauda. H&E, original magnification 2X Figure 2. Epididymal HS (control, multi-site neoplasm). Neoplastic cells infil-trating and separating tubules. H&E, original magnification 10X Figure 3. Epididymal HS (control, primary neoplasm). Similar infiltrative pattern as Figure 2. H&E, original magnification 4X Figure 4. Epididymal HS (control, primary neoplasm). Dense population of neoplastic cells with focal mononuclear cell infiltrates. H&E, original magnification 10X Figure 5. Epididymal HS (control, multi- site neoplasm). Monomorphic cell population with indistinct eosinophilic cytoplasm and slight nuclear pleomorphism. H&E, original magnification 20X Figure 6. Epididymal HS (control, multi- site neoplasm). Capsular invasion and infiltration of epididymal fat by HS cells. H&E, original magnification 20X Figure 7. Epididymal HS (low dose, multi- site neoplasm). Sperm granuloma (upper left of image). H&E, original magnification 20X Figure 8. Epididymal HS (control, multi-site neoplasm). Positive cytoplasmic and membranous staining. F4-80 IHC, original magnification 40X
