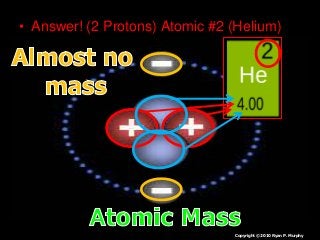
More Related Content More from www.sciencepowerpoint.com (20) 1. • Answer! (2 Protons) Atomic #2 (Helium)
Copyright © 2010 Ryan P. Murphy
3. • RED SLIDE: These are notes that are very
important and should be recorded in your
science journal.
Copyright © 2010 Ryan P. Murphy
4. -Nice neat notes that are legible and use indents when
appropriate.
-Example of indent.
-Skip a line between topics
-Make visuals clear and well drawn. Please label.
Neutron
Proton
Electron
5. • RED SLIDE: These are notes that are very
important and should be recorded in your
science journal.
• BLACK SLIDE: Pay attention, follow
directions, complete projects as described
and answer required questions neatly.
Copyright © 2010 Ryan P. Murphy
8. The Atoms and Periodic Table Unit
Copyright © 2010 Ryan P. Murphy
10. • Activity! Bringing things down to size.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
11. • Activity! Bringing things down to size.
– Take one sheet of paper 8 by 11.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
12. • Activity! Bringing things down to size.
– Take one sheet of paper 8 by 11.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
13. • Activity! Bringing things down to size.
– Take one sheet of paper 8 by 11.
– Cut it in half as precisely as possible.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
14. • Activity! Bringing things down to size.
– Take one sheet of paper 8 by 11.
– Cut it in half as precisely as possible.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
15. • Activity! Bringing things down to size.
– Take one sheet of paper 8 by 11.
– Cut it in half as precisely as possible.
– Cut in half again and again. Keep track.
Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
1
16. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
2
17. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
2
Line up scraps from large to small.
18. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
3
19. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
4
20. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
5
21. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
6
22. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
7
23. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
8
24. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
9
25. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
10
26. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
11
27. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
12
28. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
13
29. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
14
30. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
15
31. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
16
32. Copyright © 2010 Ryan P. Murphy
This is the product of Ryan
Murphy Copyright 2010
www.sciencepowerpoint.com
17
40. • Answer: If you were to cut the paper in half
about 90 times, you would be around the
size of the atom.
90
41. • Answer: If you were to cut the paper in half
about 90 times, you would be around the
size of the atom. The atom is incredibly small.
Copyright © 2010 Ryan P. Murphy
90
42. • Answer: If you were to cut the paper in half
about 90 times, you would be around the
size of the atom.
Copyright © 2010 Ryan P. Murphy
90
43. • About a million atoms stacked on top of
each other = the thickness of a sheet of
paper.
Copyright © 2010 Ryan P. Murphy
44. • About 5 million atoms make up a period at
the end of a sentence.
Copyright © 2010 Ryan P. Murphy
45. • Link! Size Scale of the Universe (Flash)
• From Sub-Atomic to the Universe.
– http://inciswf.com/589217_scale_of_universe_
enhanced.swf
48. • Journal Question?
– Please use the round Petri-dish to create a circle.
– Inside the circle, write everything you know about
the atom.
Copyright © 2010 Ryan P. Murphy
49. • Video Link! The size of atoms
– http://www.youtube.com/watch?v=yQP4UJhNn0I
Copyright © 2010 Ryan P. Murphy
50. • Video Link, Atoms, Googol’s, and the
Googolplex. (7 minutes)
– http://www.youtube.com/watch?v=gh4F5BQ8hgw
Copyright © 2010 Ryan P. Murphy
51. • Is this you billions of years ago?
Copyright © 2010 Ryan P. Murphy
52. • Answer! The atomic particles spinning
around billions of years ago are the same
atoms that make up planets and all that
are on them. Yes, your atoms were
stardust.
Answer! The atomic particles spinning
around billions of years ago are the same
atoms that make up planets and all that are
on them. Yes, you were once a gas cloud
spinning around the universe.
53. • Can we see atoms?
– Is this drawing accurate?
Copyright © 2010 Ryan P. Murphy
54. • These pictures just represent what the
atom might look like.
– They are simple models meant for
understanding.
Copyright © 2010 Ryan P. Murphy
55. • These pictures just represent what the
atom might look like.
– They are simple models meant for
understanding.
Copyright © 2010 Ryan P. Murphy
56. • These pictures just represent what the
atom might look like.
– They are simple models meant for
understanding.
– Today I will attempt to help you understand
the universe.
Copyright © 2010 Ryan P. Murphy
57. • These pictures just represent what the
atom might look like.
– They are simple models meant for
understanding.
– Today I will attempt to help you understand
the universe.
– I unfortunately must use simple pictures
because as we know atoms are very small
and they are mostly empty space.
Copyright © 2010 Ryan P. Murphy
59. • We can’t see individual atoms.
– We can see billions and billions of them
bonded together.
60. • We can’t see individual atoms.
– We can see billions and billions of them
bonded together.
63. • Video Link! A Boy and His Atom
– Smallest movie ever made from IBM.
– http://www.youtube.com/watch?v=oSCX78-8-q0
Copyright © 2010 Ryan P. Murphy
64. • Video Link! A Boy and His Atom
– Smallest movie ever made from IBM.
– http://www.youtube.com/watch?v=oSCX78-8-q0
Copyright © 2010 Ryan P. Murphy
Making of (Optional)
http://www.youtube.com/watch?v=z_8LHqwYMKY
65. • Video! A look at the atoms in steel.
• Look closely to get a good look atom .
– http://www.youtube.com/watch?v=dNvdrpEmS48
Copyright © 2010 Ryan P. Murphy
66. • An atom has charged particles, this means
it has a (+) and a (-) charge.
Copyright © 2010 Ryan P. Murphy
67. • An atom has charged particles, this means
it has a (+) and a (-) charge.
– Atoms and some of the particles they are
made of carry a charge.
Copyright © 2010 Ryan P. Murphy
68. • (Optional) Recommended Link! Khan
Academy Introduces the Atom.
• 21 minutes
• http://www.khanacademy.org/video/introduc
tion-to-the-atom?playlist=Chemistry
70. • Early experiments realized that that atoms
were charged particles. One of those
experiments is a Crookes tube.
Copyright © 2010 Ryan P. Murphy
71. • Early experiments realized that that atoms
were charged particles. One of those
experiments is a Crookes tube.
Copyright © 2010 Ryan P. Murphy
“Hoot” “Hoot” I can’t
wait to see how this
Crookes tube thing
works.”
72. • Early experiments realized that that atoms
were charged particles. One of those
experiments is a Crookes tube.
Copyright © 2010 Ryan P. Murphy
73. • Video! Cathode Ray tube.
– Record a picture of it in your journal and how it
worked to help show that atoms carry a charge.
– http://www.youtube.com/watch?v=XU8nMKkzbT8
Copyright © 2010 Ryan P. Murphy
76. Copyright © 2010 Ryan P. Murphy
+
-
Like charges (-) (-) repel.
The Electron is negative.
80. Copyright © 2010 Ryan P. Murphy
Opposite charges attract (+) (-).
The electrons are negative.
81. • What did this study find?
• It helped lead to J.J. Thompson to realizing
that this ray is negatively charged. (electron)
Copyright © 2010 Ryan P. Murphy
82. • What did this study find?
– It helped lead J.J. Thompson to realizing that this
ray is negatively charged. (electron)
Copyright © 2010 Ryan P. Murphy
83. • What did this study find?
– It helped lead J.J. Thompson to realizing that this
ray is negatively charged. (electron)
Copyright © 2010 Ryan P. Murphy
Learn more:
http://explorable.com/cathode-
ray-experiment.html
86. • Knowing that an atom had a charge was
just the first step. Scientists still didn’t
know the structure of the atom.
Copyright © 2010 Ryan P. Murphy
87. • Knowing that an atom had a charge was
just the first step. Scientists still didn’t
know the structure of the atom.
– J.J. Thompsons early plum pudding model of
an atom.
Copyright © 2010 Ryan P. Murphy
88. • Knowing that an atom had a charge was
just the first step. Scientists still didn’t
know the structure of the atom.
– J.J. Thompsons early plum pudding model of
an atom. (This was incorrect)
Copyright © 2010 Ryan P. Murphy
90. • Ernest Rutherford’s experiment used particles
and reflection to determine the structure of the
atom.
– What is the mystery shape in the next slide, using your
knowledge of reflection?
Copyright © 2010 Ryan P. Murphy
91. • Ernest Rutherford’s experiment used particles
and reflection to determine the structure of the
atom.
– What is the mystery shape in the next slide. Use your
knowledge of reflection?
Copyright © 2010 Ryan P. Murphy
92. • Try and guess the mystery shape below
based on how objects would reflect off it.
Copyright © 2010 Ryan P. Murphy
94. • Ernest Rutherford had to make sense of a
puzzle similar to this to figure out the
structure of the atom.
Copyright © 2010 Ryan P. Murphy
95. • Ernest Rutherford had to make sense of a
puzzle similar to this to figure out the
structure of the atom.
Copyright © 2010 Ryan P. Murphy
96. • Ernest Rutherford had to make sense of a
puzzle similar to this to figure out the
structure of the atom.
Copyright © 2010 Ryan P. Murphy
99. • Video! Rutherford’s Gold Foil Experiment
– http://www.youtube.com/watch?v=5pZj0u_XMbc
Copyright © 2010 Ryan P. Murphy
100. • In Rutherford's experiment, a radioactive source
shot a stream of alpha particles at a sheet of very
thin gold foil which stood in front of a screen.
Copyright © 2010 Ryan P. Murphy
101. • In Rutherford's experiment, a radioactive source
shot a stream of alpha particles at a sheet of very
thin gold foil which stood in front of a screen.
– The alpha particles would make small flashes of light
where they hit the screen.
Copyright © 2010 Ryan P. Murphy
102. • Since some of the positive alpha particles
were substantially deflected,
Copyright © 2010 Ryan P. Murphy
103. • Since some of the positive alpha particles
were substantially deflected, Rutherford
concluded that there must be something
inside an atom for the alpha particles to
bounce off of,
Copyright © 2010 Ryan P. Murphy
104. • Since some of the positive alpha particles
were substantially deflected, Rutherford
concluded that there must be something
inside an atom for the alpha particles to
bounce off of, that must be small, dense,
and positively charged.
Copyright © 2010 Ryan P. Murphy
105. • Since some of the positive alpha particles
were substantially deflected, Rutherford
concluded that there must be something
inside an atom for the alpha particles to
bounce off of, that must be small, dense,
and positively charged. The Nucleus
Copyright © 2010 Ryan P. Murphy
147. Copyright © 2010 Ryan P. Murphy
The history of the atom. Learn more at…
http://www.nobeliefs.com/atom.htm
Ernest Rutherford: Learn more…
http://www.rutherford.org.nz/
149. An Atom is the smallest part of an element
which can take part in a chemical reaction.
Copyright © 2010 Ryan P. Murphy
150. The atom consists of three fundamental
particles
-
-
-
Copyright © 2010 Ryan P. Murphy
151. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
152. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
153. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
+
154. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
155. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
+
+
156. Proton + (positive charge)
Copyright © 2010 Ryan P. Murphy
+
+
157. Neutron 0 (neutral charge / no charge)
Copyright © 2010 Ryan P. Murphy
158. Neutron 0 (neutral charge / no charge)
Copyright © 2010 Ryan P. Murphy
159. Neutron 0 (neutral charge / no charge).
Copyright © 2010 Ryan P. Murphy
160. Neutron 0 (neutral charge / no charge).
Copyright © 2010 Ryan P. Murphy
161. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
Copyright © 2010 Ryan P. Murphy
162. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
Copyright © 2010 Ryan P. Murphy
163. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
Copyright © 2010 Ryan P. Murphy
164. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
– A neutron walks into a video store.
Copyright © 2010 Ryan P. Murphy
165. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
– He asks the clerk “How much are the movies?”
Copyright © 2010 Ryan P. Murphy
166. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
– He asks the clerk “How much are the movies?”
Copyright © 2010 Ryan P. Murphy
“For you…”
“No Charge.”
167. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
– He asks the clerk “How much are the movies?”
Copyright © 2010 Ryan P. Murphy
“For you…”
“No Charge.”
168. • Neutron: A particle that appears in the nucleus of
all atoms except hydrogen.
– Neutrons have no electrical charge and just a bit more
mass than a proton.
– He asks the clerk “How much are the movies?”
Copyright © 2010 Ryan P. Murphy
169. Electron – (negative charge)
Copyright © 2010 Ryan P. Murphy
178. Nucleus: The positively charged center of
the atom.
Copyright © 2010 Ryan P. Murphy
179. Nucleus: The positively charged center of
the atom.
The nucleus has an incredibly high density.
Copyright © 2010 Ryan P. Murphy
180. Nucleus: The positively charged center of
the atom.
The nucleus has an incredibly high density.
Copyright © 2010 Ryan P. Murphy
Equal to a million million million kg m-3 or
a thousand million million tonnes m-3
181. Nucleus: The positively charged center of
the atom.
The nucleus has an incredibly high density.
Copyright © 2010 Ryan P. Murphy
Or 6 billion or so cars stuffed into a small
cardboard box.
182. • Nucleus: The positively charged center of
the atom.
– The nucleus has an incredibly high density.
Copyright © 2010 Ryan P. Murphy
198. • Quiz Wiz! 1-10 Name that part of the Atom.
– Proton, Neutron, Electron, Nucleus
Copyright © 2010 Ryan P. Murphy
210. • Answers! 1-10 Name that part of the Atom.
• Proton, Neutron, Electron, Nucleus
Copyright © 2010 Ryan P. Murphy
236. • What is this a picture of? Our Solar System
237. • What is this a picture of? Our Solar System
• Why am I showing you this?
238. • What is this a picture of? Our Solar System
• Why am I showing you this? To show the
incorrect behavior of subatomic particles.
239. • Activity! Pin the tail on the electron!
– One volunteer needs to tape the tail to the
electron. Blindfold not needed.
240. • Activity! Pin the tail on the electron!
– One volunteer needs to tape the tail to the
electron. Blindfold not needed.
241. • Activity! Pin the tail on the electron!
– What is the point in this activity?
242. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next. That
makes it impossible to plot an orbit for an
electron around a nucleus.
Copyright © 2010 Ryan P. Murphy
243. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next.
Copyright © 2010 Ryan P. Murphy
244. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next. That
makes it impossible to plot an orbit for an
electron around a nucleus.
Copyright © 2010 Ryan P. Murphy
245. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next. That
makes it impossible to plot an orbit for an
electron around a nucleus.
Copyright © 2010 Ryan P. Murphy
246. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next. That
makes it impossible to plot an orbit for an
electron around a nucleus. This is also true
for the Proton and Neutron.
Copyright © 2010 Ryan P. Murphy
247. • Heisenberg Uncertainty Principle
– You can't know with certainty both where an
electron is and where it's going next. That
makes it impossible to plot an orbit for an
electron around a nucleus. This is also true
for the Proton and Neutron.
Copyright © 2010 Ryan P. Murphy
Learn more (advanced) at…
http://science.howstuffworks.com/innovation/science-
questions/quantum-suicide2.htm
263. Could you pin point the
exact location of any single
particle?
264. Could you pin point the
exact location of any single
particle? Where will the
electron be on the next slide
– Put a marker to see if we
will be right.
266. You can’t pinpoint the exact
location of any single
particle according to The
Hinesburg Uncertainty
Principle.
268. Could you provide a general
region you might find a
particular particle? Yes, the
nucleus was usually found
around here.
269. Could you provide a general
region you might find a
particular particle? Yes, the
nucleus was usually found
around here.
270. Could you provide a general
region you might find a
particular particle? The
Electron was usually found
in this region.
271. Could you provide a general
region you might find a
particular particle? The
Electron was usually found
in this region.
272. • These type of models are just to help us
understand.
Copyright © 2010 Ryan P. Murphy
273. • Heisenburg Uncertainty Principle.
– Video Link! Hanks explains.
– Preview for language.
• http://www.youtube.com/watch?v=noZWLPpj3to
274. • Neils Bohr Model (1913):
Copyright © 2010 Ryan P. Murphy
275. • Neils Bohr Model (1913): Depicts the atom
as a small, positively charged nucleus
surrounded by electrons that travel in circular
orbits around the nucleus
– .
Copyright © 2010 Ryan P. Murphy
276. • Neils Bohr Model (1913): Depicts the atom
as a small, positively charged nucleus
surrounded by electrons that travel in circular
orbits around the nucleus
– .
Copyright © 2010 Ryan P. Murphy
277. • Neils Bohr Model (1913): Depicts the atom
as a small, positively charged nucleus
surrounded by electrons that travel in circular
orbits around the nucleus
– .
Copyright © 2010 Ryan P. Murphy
Because of its simplicity,
the Bohr model is still
commonly taught to
introduce students to
quantum mechanics.
278. • Neils Bohr Model (1913): Depicts the atom
as a small, positively charged nucleus
surrounded by electrons that travel in circular
orbits around the nucleus
– .
Copyright © 2010 Ryan P. Murphy
Because of its simplicity,
the Bohr model is still
commonly taught to
introduce students to
quantum mechanics.
We will touch upon this later in the unit.
280. • Activity – Creating an accurate model of an
atom. Atomic Cloud model.
Copyright © 2010 Ryan P. Murphy
281. • Activity – Creating an accurate model of an
atom. Atomic Cloud model.
– Create a small nucleus (3 protons) and then
make 500 hundred dots (Three Electrons moving
at the speed of light around the nucleus.)
Copyright © 2010 Ryan P. Murphy
282. • Activity – Creating an accurate model of an
atom. Atomic Cloud model.
– Create a small nucleus (3 protons) and then
make 500 hundred dots (Three Electrons moving
at the speed of light around the nucleus.)
– Label model as Atomic Cloud
Copyright © 2010 Ryan P. Murphy
285. Copyright © 2010 Ryan P. Murphy
These are
the three
electrons in
the Lithium
Atom
286. Copyright © 2010 Ryan P. Murphy
These are
the three
electrons in
the Lithium
Atom
They are
moving at
the speed
of light
287. Copyright © 2010 Ryan P. Murphy
These are
the three
electrons in
the Lithium
Atom
They are
moving at
the speed
of light
299,792, 458 m / s in a vacuum
290. Copyright © 2010 Ryan P. Murphy
They are everywhere and nowhere?
An atom is mostly empty space
292. Copyright © 2010 Ryan P. Murphy
E M P T Y S P A C E
Everything is composed
chiefly of nothing
298. Copyright © 2010 Ryan P. Murphy
It has to do with the uncertainty principle.
299. Copyright © 2010 Ryan P. Murphy
It has to do with the uncertainty principle.
-The electron cannot have a defined position
in the nuclei of atoms means that it must occupy
every other space within the atom in a wave of
possibilities.
300. Copyright © 2010 Ryan P. Murphy
It has to do with the uncertainty principle.
-The electron cannot have a defined position
in the nuclei of atoms means that it must occupy
every other space within the atom in a wave of
possibilities.
Pauli exclusion principle: The principle that two fermions
of a given type, such as electrons, protons, or neutrons,
cannot occupy the same quantum state. It does not apply
to bosons. This principle plays a key role in the electron
orbital structure of atoms, since it prevents more than two
electrons from occupying any given orbital (two are
allowed, since they may have opposite spin, and thus be
in different quantum states)
More difficult explanation.
310. • You should have completed page 2 of the
bundled homework package.
311. • You should have completed page 2 of the
bundled homework package.
312. • You should have completed page 2 of the
bundled homework package.
313. • You should have completed page 2 of the
bundled homework package.
321. Atoms always have the same number of
protons and electrons, this is called the
atomic number.
Copyright © 2010 Ryan P. Murphy
322. Atoms always have the same number of
protons and electrons, this is called the
atomic number.
Copyright © 2010 Ryan P. Murphy
323. Atoms always have the same number of
protons and electrons, this is called the
atomic number.
Copyright © 2010 Ryan P. Murphy
324. Atoms always have the same number of
protons and electrons, this is called the
atomic number.
Copyright © 2010 Ryan P. Murphy
325. Atoms always have the same number of
protons and electrons, this is called the
atomic number.
Copyright © 2010 Ryan P. Murphy
326. • What atom is this How can you tell?
Copyright © 2010 Ryan P. Murphy
327. • Answer! (2 Protons) Atomic #2
Copyright © 2010 Ryan P. Murphy
328. • Answer! (2 Protons) Atomic #2 (Helium)
Copyright © 2010 Ryan P. Murphy
329. • Answer! (2 Protons) Atomic #2 (Helium)
Copyright © 2010 Ryan P. Murphy
330. • Answer! (2 Protons) Atomic #2 (Helium)
Copyright © 2010 Ryan P. Murphy
331. • Answer! (2 Protons) Atomic #2 (Helium)
Copyright © 2010 Ryan P. Murphy
342. Atomic Number
Atomic Symbol
Name
Atomic Mass
Copyright © 2010 Ryan P. Murphy
Note: Only the
first letter is
capitalized.
Also note that the atomic
symbol for Silver is Si . Si is
the atomic Symbol for Silicon.
343. The Nucleus has almost all the mass of the
atom.
Copyright © 2010 Ryan P. Murphy
344. The Nucleus has almost all the mass of the
atom.
It’s made up of protons (+)
Copyright © 2010 Ryan P. Murphy
+
+
345. The Nucleus has almost all the mass of the
atom.
It’s made up of protons (+) and neutrons (O)
(0).
Copyright © 2010 Ryan P. Murphy
+
+ o
o
346. The Nucleus has almost all the mass of the
atom.
It’s made up of protons (+) and neutrons (O)
(0).
Copyright © 2010 Ryan P. Murphy
Mostly empty space
except for dense nucleus
+
+ o
o
347. The Nucleus has almost all the mass of the
atom.
It’s made up of protons (+) and neutrons (O)
(0).
Copyright © 2010 Ryan P. Murphy
Mostly empty space
except for dense nucleus
+
+ o
o
348. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
349. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
350. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
351. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
352. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
353. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
354. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
355. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
356. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
357. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
358. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
1 0 1
359. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
360. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
361. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
2 2 2
362. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
2 2 2
363. • The smallest atoms are Hydrogen (H) and
Helium (He).
Copyright © 2010 Ryan P. Murphy
2 2 2
364. Isotope: Atom with the same number
of protons and electrons but different
numbers of neutrons.
Copyright © 2010 Ryan P. Murphy
365. Isotope: Atom with the same number
of protons and electrons but different
numbers of neutrons.
Copyright © 2010 Ryan P. Murphy
366. Isotope: Atom with the same number
of protons and electrons but different
numbers of neutrons.
Copyright © 2010 Ryan P. Murphy
367. Isotope: Atom with the same number
of protons and electrons but different
numbers of neutrons.
Copyright © 2010 Ryan P. Murphy
368. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
369. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
370. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
371. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
372. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
Two
extra
neutrons
373. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
Two
extra
neutrons
374. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
Two
extra
neutrons
Carbon will
always have 6
protons in its
nucleus
375. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
Two
extra
neutrons
Carbon will
always have 6
protons in its
nucleus
376. • Two Carbon Isotopes, C-12, and C-14
Copyright © 2010 Ryan P. Murphy
Two
extra
neutrons
Carbon will
always have 6
protons in its
nucleus
378. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
379. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
Atomic Mass is the number of
Protons and Neutrons
380. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
Remember, electrons are so small
they don’t add much to atomic mass
381. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
382. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
383. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
384. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
14 - 6 =
385. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
14 - 6 = 8
386. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
14 - 6 = 8
387. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
14 - 6 = 8 amu (atomic mass units)
388. To find the number of neutrons: Subtract the
atomic number from the atomic mass.
Copyright © 2010 Ryan P. Murphy
14 - 6 = 8 amu (atomic mass units)
Atomic Number. Learn more at
http://education.jlab.org/qa/pen_
number.html
389. • Activity! Atomic Mass and Isotopes
Simulator.
• http://phet.colorado.edu/en/simulation/isot
opes-and-atomic-mass
390. • Count up the Protons, and then count up the
neutrons. What element is this? What would
be a rough guess for its atomic mass.
+
+ +
Copyright © 2010 Ryan P. Murphy
391. • Answer! This is Lithium, atomic number = 3.
+
+ +
Copyright © 2010 Ryan P. Murphy
392. • Answer! This is Lithium, atomic number = 3.
+
+ +
Copyright © 2010 Ryan P. Murphy
3 Protons so it must have…
393. • Answer! This is Lithium, atomic number = 3.
+
+ +
Copyright © 2010 Ryan P. Murphy
3 Protons so it must have…
3 Electrons
394. • Answer! This is Lithium, atomic number = 3.
There are 4 neutrons however.
+
+ +
Copyright © 2010 Ryan P. Murphy
395. • Answer! This is Lithium, atomic number = 3.
There are 4 neutrons however.
+
+ +
Copyright © 2010 Ryan P. Murphy
396. • Answer! This is Lithium, atomic number = 3.
There are 4 neutrons however. The atomic
mass would be roughly 7 as electrons don’t
weigh very much.
+
+ +
Copyright © 2010 Ryan P. Murphy
397. • Answer! This is Lithium, atomic number = 3.
There are 4 neutrons however. The atomic
mass would be roughly 7 as electrons don’t
weigh very much.
+
+ +
Copyright © 2010 Ryan P. Murphy
Atomic Mass: 6.941 amu.
399. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
400. • What is the atomic
symbol? F
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
401. • What is the atomic
symbol? F
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
402. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
403. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
404. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
405. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
406. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
407. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
408. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
409. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
410. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
19
Copyright © 2010 Ryan P. Murphy
411. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
19 -
Copyright © 2010 Ryan P. Murphy
412. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
19 - 9
Copyright © 2010 Ryan P. Murphy
413. • What is the atomic
symbol? F
• What is the atomic
number? 9
– How many Protons? 9
– How many Electrons? 9
• What is the atomic
Mass? 19 amu
– How many Neutrons?
19 - 9 = 10
Copyright © 2010 Ryan P. Murphy
415. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
416. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
opyright © 2010 Ryan P. Murphy
417. • What is the atomic
symbol? Ar
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
418. • What is the atomic
symbol? Ar
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
419. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
420. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
421. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
422. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
423. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass?
– How many Neutrons?
424. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass?
– How many Neutrons?
425. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
426. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
427. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
428. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
39.95
429. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
39.95 -18 =
430. • What is the atomic
symbol? Ar
• What is the atomic
number? 18
– How many Protons? 18
– How many Electrons? 18
• What is the atomic
Mass? 39.95 amu
– How many Neutrons?
39.95 -18 = 21.95
432. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
433. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
434. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
435. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
The number of
Electrons is the
Atoms Atomic #
436. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
The number of
Electrons is the
Atoms Atomic #
437. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
The number of
Electrons is the
Atoms Atomic #
6
438. • What is the atomic
symbol? C (Carbon)
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
The number of
Electrons is the
Atoms Atomic #
6
439. • What is the atomic
symbol? C (Carbon)
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
The number of
Electrons is the
Atoms Atomic #
6
440. • What is the atomic
symbol? C
• What is the atomic
number? 6
– How many Protons? 6
– How many Electrons?6
• What is the atomic
Mass? 12.01 amu
– How many Neutrons?
6
Copyright © 2010 Ryan P. Murphy
442. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
443. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. Murphy
444. • What is the atomic
symbol? Li
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
445. • What is the atomic
symbol? Li
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
446. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
447. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
448. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. Murphy
449. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
450. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
451. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
452. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass? 6.94 amu
– How many Neutrons?
Copyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. Murphy
453. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass? 6.94 amu
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
454. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass? 6.94 amu
– How many Neutrons?
3.94
Copyright © 2010 Ryan P. Murphy
455. • What is the atomic
symbol? Li
• What is the atomic
number? 3
– How many Protons? 3
– How many Electrons? 3
• What is the atomic
Mass? 6.94 amu
– How many Neutrons?
3.94
Copyright © 2010 Ryan P. Murphy
456. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
457. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
458. • What is the atomic
symbol? Fe
• What is the atomic
number? 26
– How many Protons? 26
– How many Electrons? 26
• What is the atomic
Mass? 55.84 amu
– How many Neutrons?
29.84
Copyright © 2010 Ryan P. Murphy
461. • Meet the Elements Activity Sheet.
– Periodic table needed
– Complete the page on the back using your
periodic table for the upcoming fun quiz.
467. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
468. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
469. • What is the atomic
symbol? Zn
• What is the atomic
number? 30
– How many Protons? 30
– How many Electrons? 30
• What is the atomic
Mass? 65.37 amu
– How many Neutrons? 35
Copyright © 2010 Ryan P. Murphy
470. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
471. • What is the atomic
symbol?
• What is the atomic
number?
– How many Protons?
– How many Electrons?
• What is the atomic
Mass?
– How many Neutrons?
Copyright © 2010 Ryan P. Murphy
472. • What is the atomic
symbol? B
• What is the atomic
number? 5
– How many Protons? 5
– How many Electrons? 5
• What is the atomic
Mass? 10.81 amu
– How many Neutrons?
5.81
Copyright © 2010 Ryan P. Murphy
473. • What is the atomic
symbol? B
• What is the atomic
number? 5
– How many Protons? 5
– How many Electrons? 5
• What is the atomic
Mass? 10.81 amu
– How many Neutrons?
5.81
Copyright © 2010 Ryan P. Murphy
474. • Class Quiz (1-10)
– #10 We haven’t learned yet but give it a try.
– http://www.mcwdn.org/chemist/atom/atomquiz.html
+
+ +
Copyright © 2010 Ryan P. Murphy
477. Hydrogen Helium
Neonn
Krypton, Argon, Xenon,
Helium Copper
Yttrium Tinn Krypton Francium
Zinc, Neon,
Gold, Lead Europium Americium
Mercury, Plutonium,
Uranium, Neptunium,
Einsteinium Californium Nobelium
Calcium
Arsenic Mercury Tungsten Potassium
478. • Quiz Wiz! Exploring some of the Periodic
Table because we have it out.
– Have your Periodic Table of the Elements
handy. Quiz Wiz for fun….
Copyright © 2010 Ryan P. Murphy
479. • This element only has three letters in its
name?
Copyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. Murphy
1
480. • These elements are a form of U.S.
currency that we carry around?
Copyright © 2010 Ryan P. Murphy
2
481. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”?
Copyright © 2010 Ryan P. Murphy
3
482. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”?
– Note, this is the picture that came up when I
Googled “Nodar”
Copyright © 2010 Ryan P. Murphy
3
483. • What are the names of the two elements
named after countries in Europe?
Copyright © 2010 Ryan P. Murphy
4
484. • What is the atomic number of the element
named after a famous peace prize?
Copyright © 2010 Ryan P. Murphy
5
485. • How many electrons are in the element
named for a famous gold foil
experiment?
Copyright © 2010 Ryan P. Murphy
6
486. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
Copyright © 2010 Ryan P. Murphy
7
487. • What’s the atomic number that’s associated
with my element?
Copyright © 2010 Ryan P. Murphy
8
488. • I am a the only element named after a
university in California?
Copyright © 2010 Ryan P. Murphy
9
489. • This is the only element named after a
U.S. State?
10
490. • Bonus - If any element existed that could
destroy superman, it would be…
– This is just movie stuff and the element is a
gas in real life?
Copyright © 2010 Ryan P. Murphy
491. • Answers to the Quiz Wiz – Exploring the
Periodic Table.
Copyright © 2010 Ryan P. Murphy
493. • This element only has three letters in its
name? Tin # 50, Symbol Sn
Copyright © 2010 Ryan P. MurphyCopyright © 2010 Ryan P. Murphy
1
494. • These elements are a form of U.S.
currency that we carry around?
Copyright © 2010 Ryan P. Murphy
2
495. • These elements are a form of U.S.
currency that we carry around? Nickel
#28, Ni
Copyright © 2010 Ryan P. Murphy
2
496. • These elements are a form of U.S.
currency that we carry around? Also Gold
Au #79
Copyright © 2010 Ryan P. Murphy
2
497. • These elements are a form of U.S.
currency that we carry around? Also Gold
Au #79
Copyright © 2010 Ryan P. Murphy
2
498. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”?
– Note, this is the picture that came up when I
searched “Nodar”
Copyright © 2010 Ryan P. Murphy
3
499. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”? Radon, #86,
– Note, this is the picture that came up when I
searched “Nodar”
Copyright © 2010 Ryan P. Murphy
3
500. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”? Radon, #86, amu=222
• 222 - 86 = 136 Neutrons
– Note, this is the picture that came up when I
searched “Nodar”
Copyright © 2010 Ryan P. Murphy
3
501. • How many neutrons does the dangerous
gas when spelled backwards spells
“Nodar”? Radon, #86, amu=222
• 222 - 86 = 136 Neutrons
– Note, this is the picture that came up when I
searched “Nodar”
Copyright © 2010 Ryan P. Murphy
3
502. • What are the names of the two elements
named after countries in Europe?
Copyright © 2010 Ryan P. Murphy
4
503. • What are the names of the two elements
named after countries in Europe?
Copyright © 2010 Ryan P. Murphy
4
504. • What are the names of the two elements
named after countries in Europe?
– Francium #87, Fr
Copyright © 2010 Ryan P. Murphy
4
505. • What are the names of the two elements
named after countries in Europe?
– Francium #87, Fr
– Germanium #32, Ge
Copyright © 2010 Ryan P. Murphy
4
506. • What is the atomic number of the element
named after a famous peace prize?
Copyright © 2010 Ryan P. Murphy
5
507. • What is the atomic number of the element
named after a famous peace prize?
– Nobelium, #102, No
– The Nobel Peace Prize
Copyright © 2010 Ryan P. Murphy
5
508. • What is the atomic number of the element
named after a famous peace prize?
– Nobelium, #102, No
– The Nobel Peace Prize
Copyright © 2010 Ryan P. Murphy
5
509. • How many electrons are in the element
named for a famous gold foil
experiment?
Copyright © 2010 Ryan P. Murphy
6
510. • How many electrons are in the element
named for a famous gold foil
experiment? Rutherfordium, #104, Rf
Copyright © 2010 Ryan P. Murphy
6
511. • How many electrons are in the element
named for a famous gold foil
experiment? Rutherfordium, #104, Rf
• 104 Electrons
Copyright © 2010 Ryan P. Murphy
6
512. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
Copyright © 2010 Ryan P. Murphy
7
513. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
– Mercury, #80, Hg
Copyright © 2010 Ryan P. Murphy
7
514. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
– Mercury, #80, Hg
– Plutonium, #94, Pu
Copyright © 2010 Ryan P. Murphy
7
515. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
– Mercury, #80, Hg
– Plutonium, #94, Pu
– Uranium, #92, U
Copyright © 2010 Ryan P. Murphy
7
516. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
– Mercury, #80, Hg
– Plutonium, #94, Pu
– Uranium, #92, U
– Neptunium, #93, Np
Copyright © 2010 Ryan P. Murphy
7
517. • Name at least two elements that names
could be associated with a Planet or
Dwarf Planet?
– Mercury, #80, Hg
– Plutonium, #94, Pu
– Uranium, #92, U
– Neptunium, #93, Np
Copyright © 2010 Ryan P. Murphy
7Also
Tellerium
“Earth” #52
Te
518. • What is the atomic number that associated
with my element?
Copyright © 2010 Ryan P. Murphy
519. • What is the atomic number that associated
with my element? 99, Einsteinium, #99, Es
Copyright © 2010 Ryan P. Murphy
8
520. • I am a the only element named after a
university in California?
Copyright © 2010 Ryan P. Murphy
521. • I am a the only element named after a
university in California? Berkelium, #97, Bk
Copyright © 2010 Ryan P. Murphy
9
522. • This is the only element named after a
U.S. State?
10
523. • This is the only element named after a
U.S. State? Californium, #98, Cf
10
524. • Bonus - If any element existed that could
destroy superman, it would be…,
although this is just movie stuff and the
element is a gas in real life?
Copyright © 2010 Ryan P. Murphy
525. • Bonus - If any element existed that could
destroy superman, it would be…,
although this is just movie stuff and the
element is a gas in real life?
• Krypton, #36, Kr
Copyright © 2010 Ryan P. Murphy
527. • Video Link! Nucleus Crash Course.
– (Optional and Advanced)
– http://www.youtube.com/watch?v=FSyAehMdpyI&
list=PL8dPuuaLjXtPHzzYuWy6fYEaX9mQQ8oGr
528. • Try and be the first to figure out the hidden
picture beneath the boxes.
– Raise your hand when you think you know, you
only get one guess.
Copyright © 2010 Ryan P. Murphy
539. • Try Again! Be the first to figure out the
hidden picture beneath the boxes.
– Raise your hand when you think you know,
you only get one guess.
Copyright © 2010 Ryan P. Murphy
553. • Try Again! Be the first to figure out the
hidden picture beneath the boxes.
– Raise your hand when you think you know,
you only get one guess.
Copyright © 2010 Ryan P. Murphy
565. Can you guess the atom
based on the number of
orbiting electrons?
573. The energy involved in the binding together
of the nucleons effects the mass of the atom
and is known as the mass deficit
574. • Try Again! Can you name the picture
hidden beneath the boxes?
– Raise your hand when you think you know.
You only get one guess.
– These two box games are work bonus points
on the balancing chemical equations sheet.
Copyright © 2010 Ryan P. Murphy
586. • You should have completed page 2 of the
bundled homework package.
587. • You should have completed page 2 of the
bundled homework package.
588. • You can also complete this portion of the
homework
589. • You can now neatly label in the white
spaces around each picture and color as
desired to the following…
610. • Video Link! History of the Atom.
– http://www.youtube.com/watch?v=njGz69B_p
Ug
611. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up (school dress code enforced) with
hand made costume)
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
– Be prepared to present. Presentation is a part
of your grade so get creative.
– Learn more about Cosplay at…
http://en.wikipedia.org/wiki/Cosplay
612. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up (school dress code enforced) with
hand made costume)
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
– Be prepared to present. Presentation is a part
of your grade so get creative.
– Learn more about Cosplay at…
http://en.wikipedia.org/wiki/Cosplay
614. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
615. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
616. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
Lots of
information
written all
around the
character
with element
specifics
617. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
• No toy or real weapons, school dress code enforced!
618. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
• No toy or real weapons, school dress code enforced!
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
– Be prepared to present. Presentation is a part
of your grade so get creative.
– Learn more about Cosplay at…
http://en.wikipedia.org/wiki/Cosplay
619. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
• No toy or real weapons, school dress code enforced!
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
– Be prepared to present. Presentation is a part
of your grade so get creative.
– Learn more about Cosplay at…
http://en.wikipedia.org/wiki/Cosplay
620. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
• No toy or real weapons, school dress code enforced!
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
621. • Possible Project (3 weeks from now)
– Element Cosplay (Create composite sketch or
dress-up with hand made costumes).
• No toy or real weapons, school dress code enforced!
– Must choose one element from the Periodic
Table of Elements
– You must support your character with at least 8
factoids / characteristics / uses of that element.
– Be prepared to present. Presentation is a part
of your grade so get creative.
• Learn more about Cosplay at…
http://en.wikipedia.org/wiki/Cosplay
Note: Image search for Cosplay not recommend as some cosplayers
wear revealing costumes.
622. • “AYE” Advance Your Exploration ELA and
Literacy Opportunity Worksheet
– Visit some of the many provided links or..
– Articles can be found at (w/ membership to
NABT and NSTA)
• http://www.nabt.org/websites/institution/index.php?p=
1
• http://learningcenter.nsta.org/browse_journals.aspx?j
ournal=tst
Please visit at least one of the
“learn more” educational links
provided in this unit and complete
this worksheet
623. • “AYE” Advance Your Exploration ELA and
Literacy Opportunity Worksheet
– Visit some of the many provided links or..
– Articles can be found at (w/ membership to and
NSTA)
• http://www.sciencedaily.com/
• http://www.sciencemag.org/
• http://learningcenter.nsta.org/browse_journals.aspx?jo
urnal=tst
627. http://sciencepowerpoint.com/Atoms_Periodic_Table_of_Elements_Unit.html
Areas of Focus within The Atoms and Periodic Table Unit:
Atoms (Atomic Force Microscopes), Rutherford’s Gold Foil Experiment, Cathode
Tube, Atoms, Fundamental Particles, The Nucleus, Isotopes, AMU, Size of
Atoms and Particles, Quarks, Recipe of the Universe, Atomic Theory, Atomic
Symbols, #’;s, Valence Electrons, Octet Rule, SPONCH Atoms, Molecules,
Hydrocarbons (Structure), Alcohols (Structure), Proteins (Structure), Atomic
Bonds, Ionic Bonds, Covalent Bonds, Metallic Bonds, , Precipitation Reactions,
Acids and Bases, Electron Negativity, Polar Bonds, Chemical Change,
Exothermic Reactions, Endothermic Reactions, Laws Conservation of Matter,
Balancing Chemical Equations, Oxidation and Reduction, Periodic Table of the
Elements, Organization of Periodic Table, Transition Metals, Acids and Bases,
Non-Metals, Metals, Metalloids, Ionization.
633. • This PowerPoint roadmap is one small part of
my Atoms and Periodic Table Unit.
• This unit includes a four part 2000+ slide
PowerPoint roadmap.
• 13 page bundled homework that chronologically
follows slideshow
• 14 pages of unit notes with visuals.
• 3 PowerPoint review games.
• Activity sheets, rubrics, advice page, curriculum
guide, materials list, and much more.
• http://sciencepowerpoint.com
635. • Please visit the links below to learn more
about each of the units in this curriculum
– These units take me about four years to complete
with my students in grades 5-10.
Earth Science Units Extended Tour Link and Curriculum Guide
Geology Topics Unit http://sciencepowerpoint.com/Geology_Unit.html
Astronomy Topics Unit http://sciencepowerpoint.com/Astronomy_Unit.html
Weather and Climate Unit http://sciencepowerpoint.com/Weather_Climate_Unit.html
Soil Science, Weathering, More http://sciencepowerpoint.com/Soil_and_Glaciers_Unit.html
Water Unit http://sciencepowerpoint.com/Water_Molecule_Unit.html
Rivers Unit http://sciencepowerpoint.com/River_and_Water_Quality_Unit.html
= Easier = More Difficult = Most Difficult
5th – 7th grade 6th – 8th grade 8th – 10th grade
636. Physical Science Units Extended Tour Link and Curriculum Guide
Science Skills Unit http://sciencepowerpoint.com/Science_Introduction_Lab_Safety_Metric_Methods.
html
Motion and Machines Unit http://sciencepowerpoint.com/Newtons_Laws_Motion_Machines_Unit.html
Matter, Energy, Envs. Unit http://sciencepowerpoint.com/Energy_Topics_Unit.html
Atoms and Periodic Table Unit http://sciencepowerpoint.com/Atoms_Periodic_Table_of_Elements_Unit.html
Life Science Units Extended Tour Link and Curriculum Guide
Human Body / Health Topics
http://sciencepowerpoint.com/Human_Body_Systems_and_Health_Topics_Unit.html
DNA and Genetics Unit http://sciencepowerpoint.com/DNA_Genetics_Unit.html
Cell Biology Unit http://sciencepowerpoint.com/Cellular_Biology_Unit.html
Infectious Diseases Unit http://sciencepowerpoint.com/Infectious_Diseases_Unit.html
Taxonomy and Classification Unit http://sciencepowerpoint.com/Taxonomy_Classification_Unit.html
Evolution / Natural Selection Unit http://sciencepowerpoint.com/Evolution_Natural_Selection_Unit.html
Botany Topics Unit http://sciencepowerpoint.com/Plant_Botany_Unit.html
Ecology Feeding Levels Unit http://sciencepowerpoint.com/Ecology_Feeding_Levels_Unit.htm
Ecology Interactions Unit http://sciencepowerpoint.com/Ecology_Interactions_Unit.html
Ecology Abiotic Factors Unit http://sciencepowerpoint.com/Ecology_Abiotic_Factors_Unit.html
638. • The entire four year curriculum can be found at...
http://sciencepowerpoint.com/ Please feel free to
contact me with any questions you may have.
Thank you for your interest in this curriculum.
Sincerely,
Ryan Murphy M.Ed
www.sciencepowerpoint@gmail.com