Organic chemistry (Amines), Sharda Public School, Almora U.K.
- 1. AMINES { Organic Chemistry Class XII Dr. Tanuja Nautiyal
- 2. Amines Amines are similar to ammonia (base) with one or more alkyl groups bonded to the nitrogen atom. Amines occurs widely in living organisms. Many amines are naturally occurring compounds that are very active physiologically. Numerous drugs used for the treatment of mental illness, hay fever, heart problems, and other physical disorders are amines.
- 3. Bonding Characteristics of Nitrogen Atoms in Organic Compounds: Carbon forms four bonds. (4 valence electron, 4 covalent bond, No nonbonding electron pair). Nitrogen forms three bonds (5 valence electron, 3 covalent bond, 1 nonbonding electron pair).
- 4. An Amine is an organic derivative of ammonia (NH4) in which one or more alkyl, cycloalkyl, or aryl groups are attached to the nitrogen atom. Alkyl amine Aryl amine
- 5. Classification of Amines Amines are classified on the basis of how many hydrocarbon groups are bonded to the ammonia nitrogen atom as: Primary amines (10) Secondary amines (20) Tertiary amines (30)
- 6. Classification of Amines Primary Amine Primary Amine is an amine in which the nitrogen atom is bonded to one hydrocarbon group and two hydrogen atoms. The generalized formula for a primary amine is RNH2. The functional group present in a primary amine, the NH2 group is called an Amino group. An Amino group is the -NH2 functional group. CH3 – NH2
- 7. Classification of Amines Secondary Amine Secondary Amine Is an amine in which the nitrogen atom is bonded to two hydrocarbon groups and one hydrogen atom. The generalized formula for a secondary amine is R2NH. CH3 – NH – CH3
- 8. Classification of Amines Tertiary Amine Tertiary Amine is an amine in which the nitrogen atom is bonded to three hydrocarbon groups and no hydrogen atom. The generalized formula for a tertiary amine is R3N
- 10. Physical Properties of Amines: The methylamines (mono-, di-, tri-) and ethylamine are gases at room temperature , other amines are liquid at room temperature. Amines are irritating to the skin, eyes, and mucous membranes and are toxic by ingestion. Aromatic amines are generally toxic. Amines are readily absorbed through the skin and affect both the blood and the nervous system.
- 11. Boiling points of amines The boiling points of amines are intermediate between those of alkanes and alcohols of similar molecular mass. Amines are higher than alkane boiling point, because hydrogen bonding is possible between amine molecules but not between alkane molecules. Intermolecular hydrogen bonding of amines involves the hydrogen atoms and nitrogen atoms of the amino group.
- 12. Boiling points of amines Tertiary Amines Primary Amines Secondary Amines Boiling point Increases Tertiary amines have lower boiling point than primary and secondary amines because intermolecular hydrogen bonding is not possible in tertiary amines. Secondary and tertiary amines have no hydrogen atoms directly bonded to the nitrogen atom.
- 13. Why the boiling point of amines are lower than those of corresponding alcohols? The boiling point of amines are lower than those of corresponding alcohols, because N – H bonds are weaker than O – H bonds, as nitrogen is less electronegative than oxygen. The difference in hydrogen-bond strength : O– H electronegativity differences (3.5) -(2.1) = 1.4 N– H electronegativity differences(3.0) - (2.1) = 0.9
- 14. Solubility of Amines Amines with less than six carbon atoms are infinitely soluble in water. This solubility results from hydrogen bonding between the amines and water as amine nitrogen atom has a nonbonding electron pair that can form a hydrogen bond with a hydrogen atom of water.
- 15. Basicity of Amines Both amines and ammonia produce basic aqueous solution
- 16. Basicity of Amines Amines and Ammonia are weak bases. The weak-base behaviour of amines and ammonia are due to its acceptance of a proton (H+) from water to produce ammonium ion and hydroxyl ion. NH3 + H2O → NH4 + + OH-Ammonia Ammonium ion CH3NH2 + H2O → CH3NH3 + + OH-Methylamine Methyl ammonium ion Both amines and ammonia produce basic aqueous solution
- 19. Nomenclature of Amines Naming the positive ion that results from the interaction of an amine with water is based on the following two rules: Rule 1: For alkylamine, the ending of the name of the amine is changed from amine to ammonium ion Examples: Ethylammonium ion Rule 2: For aromatic amines, the final –e of the name of the amine is replaced by –iumion Examples: Anilinium ion
- 21. Reduction of nitro compounds Aromatic amines are normally prepared by reduction of the corresponding aromatic nitro compound. Aniline is prepared from nitrobenzene. Reducing agents: Fe/H+, Sn/H+ or catalytic hydrogenation (example, H2/Pd or Pt or Ni).
- 22. Reduction of amides Amides yield primary amines on reduction by lithium aluminum hydride, LiAlH4. The oxygen atom is replaced by two hydrogen atoms, 2o and 3o amides produce 2o and 3o amines, respectively.
- 23. Reduction of Nitriles Nitriles can be reduced by strong reducing agent like H2 with catalyst (Ni) or LiAlH4 to yield primary amines.
- 24. Hoffmann degradation of amides Hoffmann degradation of amides Heating the amides with a mixture of bromine and KOH or NaOH. amides will change to amines. is used to shorten the chain by one carbon.
- 25. Reduction of halides with ammonia The reaction of ammonia with an alkyl halide leads to the formation of a primary amine. The primary amine that is formed can also react with the alkyl halide, which leads to a di-substituted amine.(2o amine)
- 26. Chemical Reactions of Amines
- 27. Alkylation of amines Reaction of amines with an acid gives a quarternary salts (an ammonium ion)
- 28. Reaction of Amines Formation of amides From acid chloride From acid anhydride From esters
- 29. Reaction with nitrous acid Primary aliphatic and aromatic amines form alkyldiazonium salts with nitrous acid.
- 30. Reaction with nitrous acid We get many useful products from alkyldiazonium salts, as it is an unstable salt.
- 31. Reaction of nitrous acid with amines Reaction of nitrous acid with aliphatic amines in cold acidic solution can be used to distinguish between primary, secondary and tertiary amines. Primary amines Secondary amines Ternary amines
- 32. Reaction with nitrous acid Secondary aliphatic and aromatic amines form nitrosoamine (yellow oils) with nitrous acid.
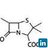
- 33. Carbyl amine Reaction Primary amines heated with tri-chloromethane and alcoholic KOH solution will produce a foul odour of isocyanide RNC
- 34. Conversion of Primary, Secondary & Tertiary Amines
- 35. Presentation by Dr. Tanuja Nautiyal Sharda Public School Almora