Case of Hemimegalencephaly with MRI Images
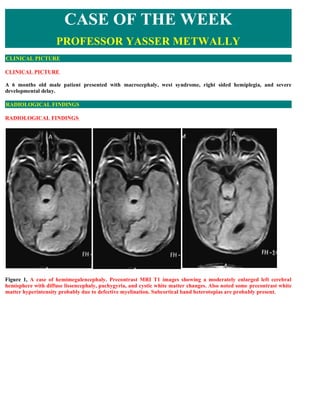
- 1. CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE A 6 months old male patient presented with macrocephaly, west syndrome, right sided hemiplegia, and severe developmental delay. RADIOLOGICAL FINDINGS RADIOLOGICAL FINDINGS Figure 1, A case of hemimegalencephaly. Precontrast MRI T1 images showing a moderately enlarged left cerebral hemisphere with diffuse lissencephaly, pachygyria, and cystic white matter changes. Also noted some precontrast white matter hyperintensity probably due to defective myelination. Subcortical band heterotopias are probably present.
- 2. Figure 2. A case of hemimegalencephaly. Precontrast MRI T1 images showing a moderately enlarged left cerebral hemisphere with diffuse lissencephaly, pachygyria, and cystic white matter changes. Also noted some precontrast white matter hyperintensity probably due to defective myelination. The ventricular system on the left side are enlarged. The genu of the Corpus callosum is poorly seen in these images. Notice abnormal shape of the brain with bulging on the left side. Figure 3. A case of hemimegalencephaly. Precontrast MRI T1 images showing a moderately enlarged left cerebral hemisphere with diffuse lissencephaly, pachygyria, and cystic white matter changes. Also noted some precontrast white matter hyperintensity probably due to defective myelination. The ventricular system on the left side are enlarged. The genu of the Corpus callosum is poorly seen in these images. Notice abnormal shape of the brain with bulging on the left
- 3. side. Figure 4. MRI T2 image (A) and MRI FLAIR images (B,C) showing diffuse white matter hyperintensity (which correlates with histopathologic findings of poor myelination and early cystic changes) and subependymal nodular heterotopias. Notice abnormal shape of the brain with bulging on the left side. Figure 5. MRI T2 image (A) and postcontrast MRI sagittal T1 image (B) showing diffuse white matter hyperintensity (which correlates with histopathologic findings of poor myelination and early cystic changes). Notice agenesis of the corpus callosum with hypoplasia of the cerebellum and the brain stem (B). The basal ganglia are poorly visualized. Notice abnormal shape of the brain with bulging on the left side.
- 4. Summary of radiological findings Abnormal shape of the skull Diffuse unilateral hypertrophy of the brain Migratory disorders (lissencephaly, pachygyria and heterotopias) White matter signal changes which correlates with histopathologic findings of poor myelination, early cystic changes and gliosis Agenesis of the corpus callosum, hypoplasia of the cerebellum and brain stem Enlargement of the ventricular system DIAGNOSIS: DIAGNOSIS: A CASE OF HEMIMEGALENCEPHALY DISCUSSION DISCUSSION Hemimegalencephaly (HME) consists of diffuse unilateral hypertrophy of the brain. This malformation has been the subject of several reports over the past few years (1). The malformation is strictly unilateral. Computed tomography (CT) and magnetic resonance imaging (MRI) show a characteristic appearance of prominent and diffuse enlargement of one hemisphere with a shift of the midline to the normal side (Fig. 6). In most cases the ventricle on the hypertrophic side is enlarged. T2-weighted MRI sequences usually show an intense signal in the subcortical white matter (2). The gyral pattern is slightly modified, typically with widening of the gyri (Fig. 7) and thickening of the cortical ribbon. In a some cases , the brain surface appeared polymicrogyric. Hemimegalencephaly or unilateral megalencephaly is a congenital disorder in which there is hamartomatous overgrowth of all or part of a cerebral hemisphere (20,21). The affected hemisphere may have focal or diffuse neuronal migration defects, with areas of polymicrogyria, pachygyria, and heterotopia. Hemimegalencephaly is a rare disorder and was first described by Sims in 1835 after reviewing 253 autopsies (22). Although the cause is unknown, it is postulated that it occurs due to insults during the second trimester of pregnancy, or as early as the 3rd week of gestation, as a genetically programmed developmental disorder related to cellular lineage and establishment of symmetry (20). Hemimegalencephaly may also be considered a primary disorder of proliferation wherein the neurons that are unable to form synaptic connections are not eliminated and are thus accumulated. Hemimegalencephaly differs from other cerebral dysgeneses because of its extreme asymmetry not corresponding to any normal stage of human brain development. No chromosomal abnormalities have been associated with hemimegalencephaly. There are three types of hemimegalencephaly (20). The isolated form occurs as a sporadic disorder without hemicorporal hypertrophy or cutaneous or systemic involvement. The syndromic form is associated with other diseases and may occur as hemihypertrophy of part or all of the ipsilateral body. It has been described in patients with epidermal nevus syndrome, Proteus syndrome, neurofibromatosis type 1, hypermelanosis of Ito, Klippel- Weber-Trenaunay syndrome, and tuberous sclerosis (21,23). Therefore, the syndromic type may follow a mendelian pattern of inheritance. The third and least common type is total hemimegalencephaly, in which there is also enlargement of the ipsilateral half of the brainstem and cerebellum. Affected patients may have macrocephaly at birth and in early infancy and often present with an intractable seizure disorder, hemiplegia, and severe developmental delay (21). Males and females are equally affected. Pregnancy is usually uncomplicated, but cesarean section may be required owing to cephalopelvic disproportion. Therefore, macrocephaly is often the first presentation at birth (21). Hemimegalencephaly has a high mortality in infancy unrelated to surgery (24,25). A brain tumor may be suspected when there is rapid enlargement of the head in the first months of life. Patients have been misdiagnosed as having obstructive hydrocephalus and undergone unnecessary ventriculoperitoneal shunting (20). In hemimegalencephaly, the clinical signs of intracranial hypertension such as separation of sutures, bulging fontanels, and the quot;setting-sunquot; sign of the eyes are absent. The latter is characteristic of increased intracranial pressure in infants, with both ocular globes deviated downward, the upper lids retracted, and the white sclerae visible
- 5. above the iris. Epilepsy is the most frequent neurologic manifestation, occurring in greater than 90% of patients (20). Although progressive hemiplegia and hemianopia are common, some patients do not have focal motor deficits (20). Hemimegalencephaly in association with neurofibromatosis type 1 may be associated with a more favorable clinical course (21,23). Figure. 6. CT scan showing the midline shift to the normal side and loss of normal gyral pattern of the hypertrophic hemisphere. Figure 7. Gross appearance of hemimegalencephaly: diffuse hypertrophy of the left hemisphere. The temporal lobe appears macrogyric. Cortical disorganization characterized as hemilissencephaly has also been reported (3). HME includes two different histologic abnormalities, neuronal and glial. In the cortex there is lack of alignment in the horizontal layers and an indistinct demarcation between gray and white matter (Fig. 8). Giant neurons scattered throughout the cortex and the subcortical white matter are clearly seen in Fig. 9 a,b. The main morphologic changes in these giant neurons are the abnormal distribution of Nissi bodies and a conspicuous proliferation of the dendritic tree visible after Golgi impregnation (Fig. 10). Glial abnormalities are present in half of the cases. The glial cytoplasm is positive for the periodic acid-Schiff, glial fibrillary acidic protein (GFAP), and vimentin reactions, and contains glial filaments on electron microscopic examination. This type of cell, sometimes called a quot;balloon cell,quot; belongs to the glial line and can be extensively distributed throughout the cortex and subcortical white matter. Invasion of the molecular layer by multinucleated glial cells is observed in many cases. Bundles of glial fibers, sometimes merging with typical Rosenthal fibers, may also be present (4). Striking demyelination of the centrum semiovale is seen in some cases, giving an appearance similar to that of Alexander's disease (5). This involvement of white matter appears to be associated with many Rosenthal fibers throughout the brain (Fig. 11), and may explain the diffuse, hyperintense signal observed in white matter on T2-weighted MRI images. It also indicates that glial cells are involved in the lesion, a clear difference between agyriapachygyria, and focal dysplasia and HME.
- 6. Figure 8. Hemimegalencephaly Figure 9a. Disappearance of horizontal layering, radial organization of neurons, and large hyperchromatic neurons scattered throughout the cortex. It is difficult to decide if HME should be considered a true malformation. Glial and nerve cell abnormalities are so widespread that this disease has sometimes been regarded as a tumor or hamartoma (6,7). It has also been suggested that such cases represent an unusually massive unilateral variant of tuberous sclerosis (8,9). The lack of skin or visceral lesions and the diffuse distribution of neuropathologic changes are the main arguments for distinguishing HME from tuberous sclerosis. This nosologic issue must be considered speculative without precise genetic data on HME: a gene for tuberous sclerosis has been localized to chromosome 9 in some tuberous sclerosis families (10). Figure 9b. Giant neurons and small pyramidal cells The neuronal pattern after Golgi impregnation shows that the increased size of the giant neurons is associated with increased size of their dendritic tree and increased number of dendritic branches. Similar findings have recently been reported in neurons of a tetraploid strain of the frog Xenopus laevis (I 1). This finding lends support to the view that the
- 7. hypertrophic neurons are polyploid, as suggested by Bignami et al. (12) and by Manz et al. (13), who found an increased amount of DNA in the cells of the hypertrophic hemisphere. The localization of neuronal anomalies to one hemisphere and to one part of the neuronal population suggests mosaicism. Interestingly, hypomelanosis of Ito, a neuroectodermal disease with known mosaicism, can be associated with HME (14,15). Figure 10. Typical Rosenthal fiber with a dense osmiophilic core surrounded by many glial filaments, * l3,000. The classical association between Jadas- sohn's linear naevus sebaceus syndrome (sebaceous adenoma) and HME is noteworthy (16-19). These cases are different from isolated HME, and the pathologic similarities to tuberous sclerosis are closer. In our experience, periventricular tumors, including giant cell astrocytomas, can be encountered in Jadassohn syndrome, as well as in tuberous sclerosis, but not in isolated HME, strongly implying that HME is a heterogeneous condition. Figure 11. A case with hemimegalencephaly. (A) Low-power photomicrograph (hematoxylin-eosin stain) of the cerebral cortex shows a thickened cortex with poor neuronal lamination (between brackets). An increased number of neurons are present in the subcortical white matter (arrow). Large abnormal blood vessels with prominent perivascular spaces are also present in the white matter (arrowheads). (B) High-power photomicrograph (hematoxylin-eosin stain) shows poorly myelinated white matter containing scattered ectopic neurons (solid straight arrow), gliosis with hypertrophic changes (curved arrow), numerous Rosenthal fibers (arrowheads), and vacuolar changes in the white matter. Focally scattered calcifications are also present in the white matter (open arrow). NEUROIMAGING OF HEMIMEGALENCEPHALY The diagnosis of hemimegalencephaly can usually be made at cross-sectional imaging. At CT, asymmetry of the cranium may be evident with enlargement of all or part of a cerebral hemisphere and ipsilateral ventricle. There is often focal, small, or extensive calcification in the white and gray matter, and the white matter may have abnormally low attenuation representing heterotopia and dysplasia of neurons. MR is the imaging modality of choice. A characteristic
- 8. finding is straightening of the ipsilateral frontal horn of the enlarged ventricle (21). However, the ipsilateral ventricle may be small in some patients. In some cases, ventricular enlargement is less severe compared with that of the involved hemisphere. At MR imaging, the white matter shows heterogeneous but frequently high signal intensity and there is often distinction of areas of agyria, pachygyria, and/or polymicrogyria. The white matter of the affected hemisphere may show advanced myelination for age (26). There is a roughly inverse relationship between the severity of the cortical and white matter abnormalities and the size of the cerebral hemisphere. Patients with agyria tend to have mild to moderate hemispheric enlargement, while those with polymicrogyria have more severe hemispheric enlargement (21,23). Prenatal and postnatal cranial sonography may reveal ventricular asymmetry and unilateral ventricular dilatation. Functional imaging with positron emission tomography has had good correlation with CT and MR imaging findings and has disclosed functionally abnormal brain regions in the noninvolved hemisphere that appeared structurally normal at CT and MR imaging (27). The gross pathologic appearance correlates with the imaging findings of enlargement of the affected cerebral hemisphere. The brain surface may show pachygyria and polymicrogyria. Microscopically, nerve cells are larger and less densely packed than in the normal side of the brain, and the number of glial cells is increased. Areas of polymicrogyria, neuronal heterotopia, and pachygyria occur. Histologically, there is no difference between focal cortical dysplasia and hemimegalencephaly. However, macroscopically, hemimegalencephaly involves the whole hemisphere, whereas focal cortical dysplasia is more limited (28). White matter may show areas of poor myelination, cystic change, and gliosis, which correspond to increased signal intensity on T2-weighted MR images. Some patients have extensive gliosis, microcystic changes, and Rosenthal fibers in the white matter resembling leukodystrophy. Such extensive white matter involvement is unusual in hemimegalencephaly. Delayed myelination was the extent of involvement described by Woo et al (28) in three patients with hemimegalencephaly. Figure 12. A case with hemimegalencephaly. (A), Axial unenhanced (a) and contrast material-enhanced (B) T1-weighted MR images show enlargement of the right cerebral hemisphere, cavitation in the region of the centrum semiovale (arrowhead), and diffuse gyral thickening (arrows) with diminished sulcation, a finding consistent with pachygyria. There are patchy, linear regions of increased signal intensity in the white matter of the right hemisphere. No pathologic enhancement is seen on the contrast-enhanced image (B).
- 9. Figure 13. A case with hemimegalencephaly. (A) Axial unenhanced T1-weighted MR image obtained at the level of the basal ganglia shows an enlarged and dysmorphic right cerebral hemisphere. The right basal ganglia are poorly demonstrated. There is moderate mass effect anteriorly (arrow). (B) On a sagittal T1-weighted MR image obtained at the midline, the corpus callosum is poorly seen (arrowhead). Figure 14. A case with hemimegalencephaly. Axial (A) and coronal (b) unenhanced T2-weighted MR images show enlargement of the right cerebral hemisphere. There is diffuse high signal intensity in the white matter, which correlates with histopathologic findings of poor myelination and early cystic changes. The right lateral ventricle is compressed (arrow in b). The cerebellum is symmetrical and appears normal (arrowhead in A).
- 10. Figure 15. A case with hemimegalencephaly. Coronal section through the right frontal hemisphere shows broad gyri and a thick cortex, particularly in the frontal lobe (solid straight arrow). The occipital lobe has a more normal gyral pattern (arrowhead). The white matter is gliotic and shows areas of mucinous and cystic degeneration (curved arrow). The gray matter-white matter junction is indistinct. The basal ganglia and thalami are small and poorly demarcated. Subventricular gray matter heterotopia is also noted (open arrow). MANAGEMENT OF HEMIMEGALENCEPHALY Syndromic hemimegalencephaly has a worse prognosis than the isolated type, and there is generally poor neurologic function in cases of hemimegalencephaly. Seizure control is the principal goal of therapy, and patients often require multiple antiepileptic medications that have adverse side effects. Hemispherectomy was first performed for treatment of refractory epilepsy in 1978 and is considered the best therapeutic choice for patients with intractable seizures (23,24,25). Anatomic or functional hemispherectomy has also been performed with improvement in quality of life (29). Nevertheless, there is a high mortality and morbidity rate associated with hemispherectomy (24,25,20). Complications include subdural hematomas and hydrocephalus, often requiring surgical intervention and ventriculoperitoneal shunting. The age of the patient at the time of surgical intervention is an important factor in development of secondary hydrocephalus, with patients younger than 9 months being more at risk (30). The intracranial space left by resection of a large portion of the brain may be intraoperatively filled with Ringer lactate or may eventually become filled with cerebrospinal fluid, but it remains vulnerable to infection and hemorrhage. Table 2. Definition of developmental disorders. Type Comment schizencephaly Schizencephaly (cleft in brain) has been regarded by many as a migration abnormality; however, it is best understood as a disorder of segmentation because one of the genes that is (disorder of abnormal in the more severe and familial forms is EMX2 [6,7]. Thus, this developmental segmentation) disorder, at least in the more severe cases, appears to be the result of failure of regional specification of a clone of cells that are destined to be part of the cortex. Megalencephaly The terms megalencephaly and hemimegalencephaly refer to disorders in which the brain volume is greater than normal (not owing to the abnormal storage of material); usually, the (Non-neoplastic disorder enlarged brain is accompanied by macrocephaly, or a large head. of neuronal proliferation) Microcephaly The term microcephaly refers to disorders in which the brain volume is smaller than normal
- 11. (Non-neoplastic disorder of neuronal proliferation) Dysembryoplastic Neoplastic proliferative disorders neuroepithelial tumor and ganglioglioma Lissencephaly Lissencephaly (smooth brain) refers to the external appearance of the cerebral cortex in those disorders in which a neuronal migration aberration leads to a relatively smooth (disorder of neuronal cortical surface. One should not consider only agyria in making this diagnosis, rather, the migration) full spectrum includes agyria and pachygyria. Agyria Extreme end of lissencephaly (sever lissencephaly) spectrum in which gyri are completely absent and the brain surface is completely smooth. (disorder of neuronal migration) Pachygyria The other end of lissencephaly spectrum (mild lissencephaly), the brain have a few broad, flat gyri separated by shallow sulci (pachygyria). The cortex is thick in pachygyria. (disorder of neuronal migration) Polymicrogyria Polymicrogyria (many small gyri) is a disorder often considered to be a neuronal migration disorder, but alternate theories exist regarding its pathogenesis, The microscopic (disorder of neuronal appearance of the lesion is that of too many small abnormal gyri. The gyri may be shallow migration) and separated by shallow sulci, which may be associated with an apparent increased cortical thickness on neuroimaging. The multiple small convolutions may not have intervening sulci, or the sulci may be bridged by fusion of overlying molecular layer, which may give a smooth appearance to the brain's surface. Heterotopias Heterotopias are collections of normal-appearing neurons in an abnormal location, presumably secondary to a disturbance in migration. Heterotopias may be classified by (disorder of neuronal their location: subpial, within the cerebral white matter, and in the subependymal region. migration) Tuberous sclerosis Disorders such as tuberous sclerosis, in which both tumor development and areas of cortical dysplasia are seen, might be a differentiation disorder. The brain manifestations of (differentiation disorder) this disorder include hamartomas of the subependymal layer, areas of cortical migration abnormalities (tubers, cortical dysgenesis), and the development of giant-cell astrocytomas in upwards of 5% of affected patients. SUMMARY The terms megalencephaly and hemimegalencephaly refer to disorders in which the brain volume is greater than normal (not owing to the abnormal storage of material); usually, the enlarged brain is accompanied by macrocephaly, or a large head. Although considered by some to be a migration disorder, the increase in brain size in these disorders appears to be attributable to errors in neuroepithelial proliferation, as the microscopic appearance of the brain is that of an increase in number of cells (both neurons and glia) and in cell size. Typically, patients are noted to have large heads at birth, and may manifest an accelerated head growth in the first few months of life. Children with megalencephaly or hemimegalencephaly may come to medical attention when presenting with seizures, a developmental disorder (mental retardation), hemihypertrophy, or a hemiparesis (opposite the affected hemisphere). Seizures vary both in onset and in type, and usually are the most problematic symptom. sometimes necessitating hemispherectomy or callosotomy.
- 12. Addendum A new version of this PDF file (with a new case) is uploaded in my web site every week (every Saturday and remains available till Friday.) To download the current version follow the link quot;http://pdf.yassermetwally.com/case.pdfquot;. You can also download the current version from my web site at quot;http://yassermetwally.comquot;. Screen resolution is better set at 1024*768 pixel screen area for optimum display REFERENCES References I .Robain 0, Floquet J, Heidt N, Rozemberg F. Hemimegalencephaly: a clinicopathological study of four cases. Neuropathol Appl Neurobiol 1988; 14:125-35. 2. Katifa GL, Chiron C, Sellier N, et al. Hemimegalencephaly MR imaging in five children. Radiology 1987;165:29-33. 3. De Rosa MJ, Secor DL, Barsom M, Fisher RS, Vinters HV. Neuropathologic findings in surgically treated hemimegalencephaly: immunohistochemical, morphometric, and ultrastructural study. Acta Neuropathol (Berl) 1992;84:250-60. 4. Robain 0, Chiron C, Dulac 0. Electron, microscopic and Golgi study in a case of hemimegalencephaly. Acta Neuropathol (Berl) 1989;77:664-6. 5. Squier M. White matter change in hemimegalencephaly. Rev Neurol 1993;149:370. 6. Dom R, Brucher JM. Hamartoblastome (gangliocytome diffus) unilateral de encore cerebrale, Rev Neurol 1969;120:317-8. 7. Townsend JJ, Nielsen SL, Malamud N. Unilateral megalencephaly hamartoma or neoplasm. Neurology 1975;25:448- 53. 8. Davis RLM, Nelson E. Unilateral ganglioglioma in a tuberosclerotic brain. J Neuropathol Exp Neurol 1961;21:571-81. 9. Jervis GA. Spongioneuroblastoma and tuberous sclerosis. J Neuropathol Exp Neurol 1954;13:105- 16. 10. Fryer AE, Connor JM, Povey S, et al. Evidence that the gene for tuberous sclerosis is on chromosome 9. Lancet 1987;1:659-61. II. Szaro BG, Tompkins R. Effects of tetraploidy on dendritic branching in neurons and glial cells of the frog Xenopus laevis. J Comp Neurol 1987;258: 304--16. 12. Bignami A, Palladini G, Zappella M. Unilateral megalencephaly with cell hypertrophy. An anatomical and quantitative histochemical study. Brain Res 1968;9:103-14. 13. Manz H, Phillips T, Rowden G, McCullough DC. Unilateral megalencephaly, cerebral cortical dysplasia, neuronal hypertrophy and heterotopia: cytophotometric fluorometric cytochemical and biochemical analysis. Acta Neuropathol (Berl) 1979; 45:97-103. 14. David TJ. Hypomelanosis of Ito: a neurocutaneous syndrome. Arch Dis Child 1981;56:798-800. 15. Turleau C, Taillard F. Hypomelanosis of Ito (incontinentia pigmenti achromians) and mosaicism for a microdeletion of 15 ql. Hum Genet 1986;74: 185-7.
- 13. 16. Choi BH, Kudo M. Abnormal neuronal migration and gliomatosis cerebri in epidermal naevus syndrome. Acta Neuropathol (Berl) 1981;53:319-25. 17. Vigevano F, Aicardi J, Lini M, Pasquinelli A. La sindrome del nevo sebaceo lineare presentazione di una casuistica multicentra. Boll Lega It Epil 1984;45-46:59-63. 18. Vles JSH, Degraeuwe P, De Cock P, Casaer P. Neuroradiological findings in Jadassohn's naevus phakomatosis: a report of two cases. Eur J Pediatr 1985;144:290-4. 19. Zaremba J, Wislawski J, Bidzinski J, Kansky J, Bogna S. ladassohn's naevus phakomatosis: a report of two cases. J Ment Deric Res 1978;22:91- 102. 20. Flores-Sarnat L. Hemimegalencephaly. I. Genetic, clinical, and imaging aspects. J Child Neurol 2002; 17:373-384. 21. Barkovich AJ, Chuang SH. Unilateral megalencephaly: correlation of MR imaging and pathologic characteristics. AJNR Am J Neuroradiol 1990; 11:523-531. 22. Sims J. On hypertrophy and atrophy of the brain. Med Quir Trans 1835; 19:315-380. 22. Wolpert SM, Cohen A, Libenson MH. Hemimegalencephaly: a longitudinal MR study. AJNR Am J Neuroradiol 1994; 15:1479-1482. 23. Vigevano F, Bertini E, Boldrini R, et al. Hemimegalencephaly and intractable epilepsy: benefits of hemispherectomy. Epilepsia 1989; 30:833-843. 24. Mathis JM, Barr JD, Albright AL, Horton JA. Hemimegalencephaly and intractable epilepsy treated with embolic hemispherectomy. AJNR Am J Neuroradiol 1995; 16:1076-1079. 25. Yagishita A, Arai N, Tamagawa K, Oda M. Hemimegalencephaly: signal changes suggesting abnormal myelination in MRI. Neuroradiology 1998; 40:734-738. 26. Rintahaka PJ, Chugani HT, Messa C, Phelps ME. Hemimegalencephaly: evaluation with positron emission tomography. Pediatr Neurol 1993; 9:21-28. 27. Adamsbaum C, Robain O, Cohen P, Delalande O, Fohlen M, Kalifa G. Focal cortical dysplasia and hemimegalencephaly: histological and neuroimaging correlations. Pediatr Radiol 1998; 28:583-590. 28. Woo C, Chuang S, Becker L, et al. Radiologic-pathologic correlation in focal cortical dysplasia and hemimegalencephaly in 18 children. Pediatr Neurol 2001; 25:295-303. 29. Carreno M, Wyllie E, Bingaman W, Kotagal P, Comair Y, Ruggieri P. Seizure outcome after functional hemispherectomy for malformations of cortical development. Neurology 2001; 57:331-333. 30. Di Rocco C, Iannelli A. Hemimegalencephaly and intractable epilepsy: complications of hemispherectomy and their correlation with the surgical technique—a report on 15 cases. Pediatr Neurosurg 2000; 33:198-207. 31. Metwally, MYM: Textbook of neurimaging, A CD-ROM publication, (Metwally, MYM editor) WEB-CD agency for electronic publishing, version 9.1a January 2008
