Bitter principles Lec.2 (2017)
- 1. Bitter Principles Lecture-2 By Dr. Ahmed Metwaly
- 2. Objectives: Non–Terpenoid Bitters • Phenolic bitters I. Humulone(α-Lupulinic acid) 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses5. Uses 6. Chemical test II. Lupulone (β- Lupulinic acid) 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
- 3. • Chromone bitters I. Khellin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Mechanism of action 7. Chemical test7. Chemical test II. Visnagin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
- 4. • Coumarin bitters I. Xanthotoxin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Mechanism of action 7. Chemical test II. Imperatorin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
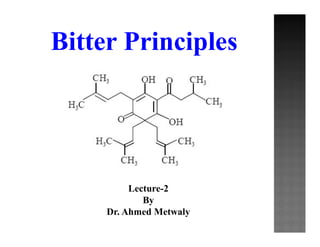
- 5. Phenolic bitters Humulone (α-Lupulinic acid) and Lupulone (β- Lupulinic acid) Structure These are crystalline phloroglucinol derivatives, the α-acids e.g. humulone and the β-acids e.g. lupulone Humulone; (6S)-3,5,6-Trihydroxy-2-(3-methylbutanoyl)-4,6-bis(3-methylbut-2-en-1- yl)cyclohexa-2,4-dien-1-one Lupulone; 3,5-dihydroxy-2-(3-methylbutanoyl)-4,6,6-tris(3-methylbut-2- enyl)cyclohexa-2,4-dien-1-one Phloroglucinol Humulone Lupulone
- 6. O R OHHO O H C C CH3 CH3 C H2 Humulone R = OH Lupulone R =
- 7. Biological Source; Humulone and lupulone are the chief active constituents obtained from the dried flowers (strobiles) of Humulus lupulus (hops) family Cannabinaceae Moraceae. The bitterness of hops is mainly due to acidic bitter principles humulone, lupulone and about 10 % of resins10 % of resins Hops bear glandular hairs which are known as lupulin, which contain most of the bitter principles that account for the use of the product in brewing and in medicine. Humulus lupulus
- 8. Isolation of humulone Hops strobiles are macerated with alcohol and filtered. The filtrate is heated with animal charcoal to adsorb the bitter principle, the suspension cooled and filtered. The charcoal is boiled with alcohol to extract the bitter principles. The alcohol extract is evaporated and the residue treated with boiling water that extracts humulone. The aqueous solution is cooled and humulone extracted by shaking with ether. The ether extract is evaporated to yield humulone. The ether extract is evaporated to yield humulone.
- 9. Characteristic Features 1. The crystals obtained from ether have mp 65-66.5°C 2. It has a distinct bitter taste especially in alcoholic solution. 3. Humulon is observed to be more stable to air than lupulon. 4. It is a monobasic acid. 5. It has a specific optical rotation [α]20 D – 212° (1.0 g in 15.5 g 96% v/v ethanol) 6. It has uvmax (ethanol): 237, 282 nm (ε 13, 760; 8330). 7. It is found to be soluble in usual organic solvent. 8. It is slightly soluble in boiling water from which it normally separates out as a milky precipitate on cooling.on cooling. 9. It readily forms a sodium salt which is rapidly soluble in water. 10. Bacteriostatic Potency: Humulon suffers no loss of bacteriostatic potency against Staphylococcus aureus. However, the addition of ascorbic acid in low concentrations extends the duration of bacteriostatic action. Test for identification; An alcoholic solution of humulone gives a reddish-violet color with alcoholic ferric chloride (phenolic).
- 10. Uses; Humulone and lupulone contribute to the bitterness of hops. They are used as aromatic bitters and exert a mild sedative action. Hops are used in the manufacture of beer for taste improvement (flavoring agent). Humulone was found to exert a bacteriostatic activity
- 11. Chromone bitters
- 12. O O OCH3 R O CH2R1 R R1 Khellin OCH3 H Visnagin H H Khellol glucoside H O-Glucose Khellin and Visnagin Khellin ; 4,9-Dimethoxy-7-methylfuro[3,2-g]chromen-5-one; (Khellin is a Furanochromone compound. Visnagin; 4-methoxy-7-methylfuro[3,2-g]chromen-5-one; Khellol glucoside; 7-[(β-D-glucopyrano)-methyl 4-methoxy-7- methylfuro[3,2-g]chromen-5-one
- 13. Biological source; Fruits of Ammi visnaga family Umbelliferae. The fruit contains about 1 % of khellin beside two other crystalline compounds, visnagin (about 0.1 %) and khellol glycoside (about 0.3 %). Ammi visnaga
- 14. Powdered Ammi visnaga fruits Extract with ether. Ether extract - Concentrate - Cool (refrigerator) Green Oily Cream colored Fatty Green Crystalline Discard by filtration (suction) Discard by dissolving in Petroleum ether Repeated crystallization from metahnol Mother liquor Crystals Upper layer Lower layerMiddle layer Isolation; Mother liquor Crystals Khellin- Evaporate to dryness - Benzene Visnagin & traces of Khellin Benzene solution - Petroleum ether - Cool - Filter Crystals Khellin Filtrate Visnagin Dry & recrystallize from methanol
- 15. Properties; Khellin: 1.Khellin occurs as bitter needle crystals, m.p. 153ºC. 2.It is freely soluble in CHCl3 and alcohol, less soluble in ether, sparingly soluble in petroleum ether and cold H2O, but more soluble in hot water and hot CH3OH. Visnagin: 1.Visnagin occurs as colorless thread-like needles.1.Visnagin occurs as colorless thread-like needles. 2.It is freely soluble in CHCl3 and ether, sparingly soluble in alcohol, insoluble in cold water, and petroleum ether Khellol glucoside; 1. It is obtained as crystals from ethanol having mp 179°C. 2. It has uvmax (ethanol): 250, 325 nm. 3. It is found to be soluble in acetic acid, hot ethanol slightly soluble in hot methanol; and almost insoluble in acetone, ethyl acetate, ether, chloroform, cold alkali.
- 16. Tests for identification; Khellin: 1.Mix few crystals of khellin with solid KOH or NaOH a rose red color is produced (not given with K2CO3 or Na2CO3 or bicarbonate). 2.Mix few crystals of khellin in a porcelain dish with a drop of H2SO4 or of phosphoric acid, a reddish-orange color is produced, that turn to bright yellow on addition of 2-3 drops of H2O. 3.Add about 1-ml nitric acid (1:1) to few mg of khellin in a porcelain dish, then3.Add about 1-ml nitric acid (1:1) to few mg of khellin in a porcelain dish, then add 5 ml NaOH solution, violet color is produced. 4.To few crystals of khellin, add an equal quantity of ninhydrin followed by 2 – 3 drops of concentrated H2SO4, an emerald green color is produced on stirring. Visnagin: Triturate few crystals of visnagin with solid KOH or NaOH, a faint rose red color is produced (paler than that produced with khellin)
- 17. Identification Test Khellol glucoside may be identified by making its following tetraacetate derivative due to the presence of four OH moieties Khellol Glucoside Tetracetate It is obtained as flakes from ethanol having mp 153°C. It is freely soluble in acetone, ethyl acetate; and almost insoluble in petroleum ether.
- 18. Uses ; • Khellin can be used for Vitiligo
- 20. Vitiligo is a disease which causes loss of pigmentation in different portions of skin. Mechanism of Khellin action is; When khellin is applied topically in combination with UVA light, it is able to stimulate melanocytes (cells that produce melanin) in hair follicles for successful treatment of vitiligo
- 21. Khellin can be used for Kidney Stones and Renal colic Khellin has also been used to treat renal colic, which is due mostly to stone formation. It were seen to relieve renal colic by relaxing the ureter and acting as a diuretic.
- 22. As khellin act as vasodilator, It is used in treatment of coronary insufficiency, angina pectoris and in bronchial asthma. Vinagin and kellol glucosides also have a vasodilator effects. Side effects; Unwanted side effects include dizziness, reversible cholestatic jaundice, pseudoallergic reaction, and elevated levels of liver enzymes
- 23. Coumarin bitters Xanthotoxin, Imperatorin and Bergapten OO O R R1R1 R R1 Psolaren H H Xanthotoxin H OCH3 Imperatorin H OCH2- CH=C(CH3)2 Bergapten OCH3 H Xanthotoxin 9-methoxy-7H-furo[3,2-g]chromen-7-one 9-(3-Methylbut-2-enoxy)-7-furo[3,2-g]chromenone
- 24. Biological source; Xanthotoxin (Ammoidin or Methoxsalen) is obtained from the fruits of Ammi majus (Umbelliferae). Imperatorin (Ammidin) occurs in the fruits of Ammi majus (Umbelliferae). Bergapten was first isolated from the oil of bergamot from Citrus bergamia (Rutaceae) and fruits of Ammi majus and some other plants belonging to family Umbelliferae . Ammi majus
- 25. Isolation of Xanthotoxin and Imperatorin; Xanthotoxin and Imperatorin are isolated from the petroleum ether extract of the fruits of Ammi majus according to the following scheme: Powdered Ammi majus fruits - Extract with petroleum ether - Concentrate - Decantation while hot Crystalline mass Dark green , semi-solid Crude xanthotoxin - Dissolve in boiling alcohol - Cool Pale green crystals Mother liquor - Concentrate - Cool Crude imperatorin Xanthotoxin - Dissolve in ether - Concentrate and cool Imperatorin
- 26. Properties; Xanthotoxin occurs as a crystalline solid substance with bitter taste. It is insoluble in cold water, more soluble in boiling H2O, hot petroleum ether, ether, benzene, CHCl3, ethyl acetate and glacial acetic acid. Imperatorin occurs as prisms or needle crystals with the same characters as xanthotoxin.
- 28. Test for Identification; Test Xanthotoxin Imperatorin Triturated few crystals in a porcelain dish with concentrated H2SO4. Test with Marquis reagent. Reduction of ammoniacal Orange yellow color that turns to light green. Orange color that turns to grass green. No reduction Deep orange color that turns to brown. Orange color that turns to brown. ReductionReduction of ammoniacal silver nitrate and Fehling's solution. Test with Wagner's reagent. Boil with dilute HNO3. No reduction Precipitate Yellow color turns to crimson on treatment with alkali. Reduction No precipitate Yellow color turns to purple on treatment with alkali.
- 29. Psoriasis is a chronic autoimmune condition that causes hyper proliferation of outer skin layers
- 30. Xanthotoxin used to treat psoriasis psoriasis
- 34. Uses Xanthotoxin used to treat psoriasis, eczema and vitiligo in conjunction with exposing the skin to UVA light from lamps or sunlight. The dosage comes in 10 mg tablets, which are taken in the amount of 30 mg 75 minutes before a PUVA (psoralen + UVA) light treatment. Mechanism of action; The exact mechanism of action of methoxsalen with the epidermal melanocytes and keratinocytes is not known.
- 35. The exact Topical mechanics in treatment of vitiligo are unknown but it has been suggested that melanocytes in the hair follicles are stimulated to move up the follicle and to repopulate the epidermis. Methoxsalen modifies the way skin cells receive the UVA radiation, allegedly clearing up the disease. Psoralens given orally are preferentially taken up by epidermal cells. The best known biochemical reaction of methoxsalen is with DNA.The best known biochemical reaction of methoxsalen is with DNA. Methoxsalen, upon photoactivation, conjugates and forms covalent bonds with DNA which leads to the formation of both monofunctional (addition to a single strand of DNA) and bifunctional adducts (crosslinking of psoralen to both strands of DNA). Reactions with proteins have also been described.
- 36. Contraindications; Patients with high blood pressure or a history of liver problems are at risk for inflammation and irreparable damage to both liver and skin. The eyes must be protected from UVA radiation. Side effects include nausea, headaches, dizziness, and in rare cases insomnia. Skin burning and increase in risks of squamous cell carcinoma (9 folds more) A prospective study of 1380 patients over 5 years revealed an approximately nine-fold increase in risks of squamous cell carcinoma among PUVA treated patients
- 37. Summary: Non–Terpenoid Bitters • Phenolic bitters I. Humulone(α-Lupulinic acid) 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses5. Uses 6. Chemical test II. Lupulone (β- Lupulinic acid) 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
- 38. • Chromone bitters I. Khellin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Mechanism of action 7. Chemical test7. Chemical test II. Visnagin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
- 39. • Coumarin bitters I. Xanthotoxin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Mechanism of action 7. Chemical test II. Imperatorin 1. Biological Sources 2. Chemical structure 3. Characteristic features 4. Isolation 5. Uses 6. Chemical test
