Amines fy b. pharmacy pci
- 1. A M I N E
- 2. At the end of session students should able to- 1 2. Explain basic nature of amines 3. Compare basicity of substituted amines 4. Explain functional group test for amine 5. Draw structure and list out various uses of amines
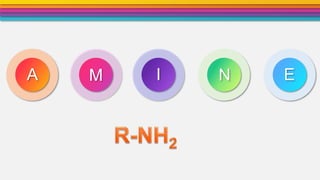
- 3. Definition2 Amines are the derivative of ammonia (NH3) in which one or more hydrogen atom are replaced by alkyl groups. •General formula R-NH2 or Ar-NH2 •(Where R= Alkyl group and Ar= Aromatic ring) NH2 NH diphenylamineAniline H CH3 H3C NH3C N H3C N CH3 trimethyl amine H Methyl amine H dimethyl amine CH3
- 4. Classification3 Amines are classified as primary (1⁰), secondary (2⁰) and tertiary (3⁰) according to the number of alkyl group attached to the nitrogen CH3H CH3 H3C NH3C N H3C N CH3 tertiary H primary H secondary
- 5. Basicity of amines Amines are basic in nature. • The presence of lone pair of electron on nitrogen atom makes it basic. This lone pair of electron is radially available for the formation new N-H bond. • The substance which donate electron in reaction is a base according to Lewis concept. 1 HH + + - + R N H ClR N HCl H Salt H Amine Base acid
- 6. Relative basicity of alkyl amines2 • Here as the substitution of alkyl group increases basicity increases • More alkyl group more rapid the electron donation process. • More stable new N-H bond and positive charge on nitrogen.
- 7. Electron donating group e.g. alkyl group increases the basicity Electron withdrawing group e.g. Phenyl ring decreases the basicity 3
- 8. Aniline4 Aniline is stabilized by Resonance that is delocalization of electron as shown below: NH2
- 9. Basicity of aniline with substituents5 • This is because, in 2,4,6- trinitro aniline internal hydrogen bonding permits delocalization of lone pair of electron on N hence less available for new N-H bond (Less basic) • In 2,4,6- trinitro-N,N-dimethyl aniline due to strong steric inhibition of resonance, lone pair of electron on N is radially available for new N- H bond (More basic) H HH3C CH3 O N ON -++ ONN- O NO2O2N + NNO2 - OO 2,4,6-trinitro aniline 2,4,6-trinitro N,N-dimethyl aniline 2,4,6- trinitro-N,N-dimethyl aniline is more basic than 2,4,6- trinitro aniline
- 10. Qualitative test: Amine Nitrous Acid test: 1 Test Observation Inference 0.2 g or 0.2 ml substance + 2 ml conc. HCl + 2 ml water and boil. Cool in ice bath to 5⁰C. Add 2 ml NaNO2 solution and mix. 1 Evolution of N2 gas as bubble 2. Yellow oily layer 3. No visible sign of reaction i.e. observation 1 & 2 absent 1. Primary amine 2. Secondary amine 3. Tertiary amine ++ Mixture of productHNO2 N2R NH2 Bubble primary amine
- 11. Nitrous acid test reaction2
- 12. Hinsberg test 3 2. Secondary 3. Tertiary amine Test Observation Inference 0.2 g or 0.2 ml substance + 2 ml Pyridine + 2 ml Fresh 2% NaOH. Shake well and add 2 drops of benzene sulphonyl chloride. 1. ppt soluble in NaOH 2. ppt insoluble in NaOH 3. No ppt 1. Primary amine amine
- 14. Qualitative test: Amine5 Carbyl amine test: Test Observation Inference 0.2 g or 0.2 ml substance + 2 ml alcoholic KOH + 2-3 drop chloroform and warm carefully. Evil smell Primary amine Secondary & Tertiary amine do not give this reaction This test is used to distinguish primary amine from secondary and tertiary amine ++ ++ 3H O + 3KClCHCHCl3 R N3KOH 2 R NH2 Isocynate (Evil Smell) primary amine
- 15. Qualitative test: Amine Azo dye test 6 present Test Observation Inference Test tube 1- 0.2 g or 0.2 ml substance + 2 ml dil. HCl & cool in ice bath to 5⁰C. Test tube 2- 2 ml NaNO2 solution & cool in ice bath to 5⁰C and mix test tube 1 & 2. To this add ice cooled solution of β-napthol in NaOH. Formation of orange red dye stuff Primary aromatic amine is
- 16. Azo dye test reaction7
- 17. IUPAC: 2-Aminoethanol 1 For the preparation of buffer solution In production of detergent As emulsifier As corrosion inhibitor In making of hair dye/colour H H H2N C C OH H H
- 18. Structure and Uses : Ethylenediamine2 IUPAC: Ethane-1,2-diamine To produce chelating agent EDTA (Ethylenediamine tetra acetic acid) Used for making intermediate drug / chemical In manufacturing of surfactant and dyes Widely used precursor to various polymer Used in binder, adhesive H H H2N C C NH2 H H
- 19. Structure and Uses: Amphetamine3 IUPAC: 1-phenylpropan-2-amine To treat nacrolepsy i.e. sleep disorder To treat obesity To improve physical (athletic) performance i.e. to increase muscle strength For enhancing cognitive performance i.e. memory Side effect- insomnia, fatigue, mood swing H H C C NH2 H CH3