2018 BDSRA Mitchell CLN5 CLN6
•
1 recomendación•28 vistas
Gene Therapy in Sheep CLN5 CLN6
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Recomendados
Recomendados
Sheep as models for Batten disease - update on current Australian research2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7

2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7Batten Disease Support and Research Association
Rustic global and widespread local white matter abnormalities in juvenile neuronal ceroid lipofuscinosis2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...

2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...Batten Disease Support and Research Association
Developing New Therapies for Batten Disease: Patient Involvement2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7

2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7Batten Disease Support and Research Association
Testing therapies for CLN5 and CLN62017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6

2017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6Batten Disease Support and Research Association
Investigating the molecular basis of NCL: a genetic research path towards drug development2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...

2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...Batten Disease Support and Research Association
Más contenido relacionado
Más de Batten Disease Support and Research Association
Sheep as models for Batten disease - update on current Australian research2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7

2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7Batten Disease Support and Research Association
Rustic global and widespread local white matter abnormalities in juvenile neuronal ceroid lipofuscinosis2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...

2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...Batten Disease Support and Research Association
Developing New Therapies for Batten Disease: Patient Involvement2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7

2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7Batten Disease Support and Research Association
Testing therapies for CLN5 and CLN62017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6

2017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6Batten Disease Support and Research Association
Investigating the molecular basis of NCL: a genetic research path towards drug development2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...

2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...Batten Disease Support and Research Association
Más de Batten Disease Support and Research Association (20)
2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7

2017 BDSRA Tammen, Grupen, James and Delerue CLN6 CLN7
2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...

2017 BDSRA Autti, U. Roine, T. Roine, Aberg, Tokola, Balk, Hakkarainen, Manne...
2017 BDSRA Trometer, Potier, Cournoyer, and Schermer

2017 BDSRA Trometer, Potier, Cournoyer, and Schermer
2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7

2017 BDSRA Mole, Band, Codd and West CLN3, CLN6, CLN7
2017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6

2017 BDSRA Hughes, Best, Wicky, Clare, Cheong, Yip, Biggs and Mace CLN5 CLN6
2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...

2017 BDSRA Cotman, Chandrachud, Hillje, Ilo, Sci, Nowell and Klein CLN2, CLN3...
Último
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Escort ServiceModels Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...GENUINE ESCORT AGENCY
Model Call Girl Services in Delhi reach out to us at 🔝 9953056974 🔝✔️✔️
Our agency presents a selection of young, charming call girls available for bookings at Oyo Hotels. Experience high-class escort services at pocket-friendly rates, with our female escorts exuding both beauty and a delightful personality, ready to meet your desires. Whether it's Housewives, College girls, Russian girls, Muslim girls, or any other preference, we offer a diverse range of options to cater to your tastes.
We provide both in-call and out-call services for your convenience. Our in-call location in Delhi ensures cleanliness, hygiene, and 100% safety, while our out-call services offer doorstep delivery for added ease.
We value your time and money, hence we kindly request pic collectors, time-passers, and bargain hunters to refrain from contacting us.
Our services feature various packages at competitive rates:
One shot: ₹2000/in-call, ₹5000/out-call
Two shots with one girl: ₹3500/in-call, ₹6000/out-call
Body to body massage with sex: ₹3000/in-call
Full night for one person: ₹7000/in-call, ₹10000/out-call
Full night for more than 1 person: Contact us at 🔝 9953056974 🔝. for details
Operating 24/7, we serve various locations in Delhi, including Green Park, Lajpat Nagar, Saket, and Hauz Khas near metro stations.
For premium call girl services in Delhi 🔝 9953056974 🔝. Thank you for considering us!Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X79953056974 Low Rate Call Girls In Saket, Delhi NCR
Último (20)
Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...

Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...
Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...

Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...
Coimbatore Call Girls in Thudiyalur : 7427069034 High Profile Model Escorts |...

Coimbatore Call Girls in Thudiyalur : 7427069034 High Profile Model Escorts |...
Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available

Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available
Call Girls Coimbatore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Coimbatore Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Most Beautiful Call Girl in Bangalore Contact on Whatsapp

Most Beautiful Call Girl in Bangalore Contact on Whatsapp
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Call Girls in Gagan Vihar (delhi) call me [🔝 9953056974 🔝] escort service 24X7
Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...

Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...
Call Girls in Lucknow Just Call 👉👉7877925207 Top Class Call Girl Service Avai...

Call Girls in Lucknow Just Call 👉👉7877925207 Top Class Call Girl Service Avai...
Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...

Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...
Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Mumbai Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Mumbai Just Call 8250077686 Top Class Call Girl Service Available
Coimbatore Call Girls in Coimbatore 7427069034 genuine Escort Service Girl 10...

Coimbatore Call Girls in Coimbatore 7427069034 genuine Escort Service Girl 10...
Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...

Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...
💚Call Girls In Amritsar 💯Anvi 📲🔝8725944379🔝Amritsar Call Girl No💰Advance Cash...

💚Call Girls In Amritsar 💯Anvi 📲🔝8725944379🔝Amritsar Call Girl No💰Advance Cash...
Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...

Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...
2018 BDSRA Mitchell CLN5 CLN6
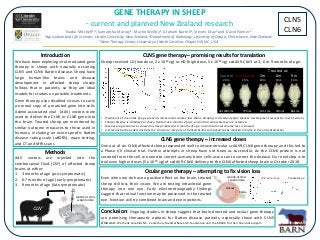
- 1. CLN5 CLN6 GENE THERAPY IN SHEEP - current and planned New Zealand research Introduction We have been exploring viral-mediated gene therapy in sheep with naturally occurring CLN5 and CLN6 Batten disease. Sheep have large human-like brains and disease development in affected sheep closely follows that in patients, so they are ideal models for studies on possible treatments. Gene therapy uses disabled viruses to carry a normal copy of a mutated gene into cells. Adeno-associated viral (AAV) vectors were used to deliver the CLN5 or CLN6 gene into the brain. Treated sheep are monitored by similar outcome measures to those used in humans, including an ovine-specific Batten disease rating scale (oBDRS), maze testing, and CT and MRI scans. Conclusion: Ongoing studies in sheep suggest that brain-directed and ocular gene therapy are promising therapeutic options for Batten disease patients, especially those with CLN5 disease. We thank CureKids NZ, Canterbury Medical Research Foundation and the BDSRA for their financial support. Nadia Mitchell1,2, Samantha Murray2, Martin Wellby1, Graham Barrell1, Steven Gray3 and David Palmer1 1Agriculture and Life Sciences, Lincoln University, New Zealand; 2Department of Radiology, University of Otago, Christchurch, New Zealand; 3Gene Therapy Center, University of North Carolina, Chapel Hill, NC, USA Methods AAV vectors are injected into the cerebrospinal fluid (CSF) of affected sheep brains at either: 1. 3 months of age (pre-symptomatic) 2. 6-7 months of age (early symptomatic) 3. 9 months of age (late symptomatic) CLN-/- scAAV9.CLN5 or scAAV9.CLN6 • • • • • • Vitreous Retina CLN5 gene therapy – promising results for translation • Treatment at 3 months of age prevents intracranial volume loss (brain atrophy) and stereotypical disease development (except for loss of vision). • Clinical disease is stabilised in sheep treated at 6 months of age, and intracranial volumes are retained. • Disease progression was slowed in sheep treated at 9 months of age, and intracranial volume loss is slowed. • 3-D reconstructions demonstrate the structural integrity of treated brains compared to the marked atrophy in the untreated brain. Sheep received LD (low dose, 2 x 1012 vg) or HD (high dose, 5 x 1012 vg) scAAV9.CLN5 at 3, 6 or 9 months of age. CLN6 gene therapy – increased doses One out of six CLN6 affected sheep responded well to intraventricular scAAV9.CLN6 gene therapy and this led to a Phase I/II clinical trial. Further attempts in sheep have not been as successful. As the CLN6 protein is not secreted from the cell, we need to correct as many brain cells as we can to correct the disease. Our next step is to trial even higher doses (5 x 1013 vg) of scAAV9.CLN6 delivery to the CLN6 affected sheep brain in October 2018. Even when we do have a positive effect on the brain, treated sheep still loss their vision. We are testing intravitreal gene therapy into one eye. Early electroretinography findings suggest that retinal function may be preserved in the treated eye. Next we will try combined brain and eye injections. Ocular gene therapy – attempting to fix vision loss Control 20m Untreated 19m Treated at: 3m 6m 9m 19m 19m 19m Vol: 103.5 mL 77.9 mL 101.1 mL 90.3 mL 88.1 mL Untreated eye Treated eyescAAV9.CLN5 or scAAV9.CLN6
