Wound healing 2014
- 1. Wound Healing สรีระวิทยาและกลไกการหายของบาดแผล นายแพทย์ ชูชัย ศรชา นิพบ. วว. ศัลยศาสตร์ทั่วไป chuchai.s@nhso.go.th chuchai.sn@gmail.com
- 2. INTRODUCTION Wound healing is a vague term that often confuses and diverts the clinician from focusing on a specific diagnosis. Over the ages, many agents have been placed on wounds to improve healing.
- 3. To date nothing has been identified that can accelerate healing in a normal individual. • Over the ages, many agents have been placed on wounds to improve healing. • Many hinder the healing process. • A surgeon’s goal in wound management is to create an environment where the healing process can proceed optimally.
- 4. Normal Wound-healing Process Phase Cellular and Bio-physiologic Events Hemostasis 1.vascular constriction 2.platelet aggregation, degranulation, and fibrin formation (thrombus) Inflammation 1.neutrophil infiltration 2.monocyte infiltration and differentiation to macrophage 3.lymphocyte infiltration Proliferation 1.re-epithelialization 2.angiogenesis 3.collagen synthesis 4.ECM formation Remodeling 1.collagen remodeling 2.vascular maturation and regression ECM, extracellular matrix. ที่มา J Dent Res. Mar 2010; 89(3): 219–229.
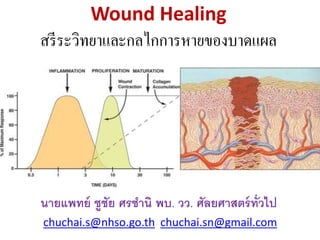
- 5. Wound Healing Events Approximate times of the different phases of wound healing, with faded intervals marking substantial variation, depending mainly on wound size and healing conditions, but image does not include major impairments that cause chronic wounds.
- 6. Wounding • Blood vessels are disrupted, resulting in bleeding. Hemostasis is the first goal achieved in the healing process. • Cellular damage occurs, this initiates an inflammatory response. • The inflammatory response triggers events that have implications for the entire healing process. • Step one then is hemostasis, resulting in Fibrin.
- 7. Wound Healing 1. Vascular and inflammatory phase 2. Re epithelization 3. Granulation tissue formation 4. Fibroplasia and matrix formation 5. Wound contraction 6. Neo vascularization 7. Matrix and collagen remodelling
- 8. PART I NORMAL WOUND HEALING E.G. CLEAN CUT WOUND
- 9. EARLY EVENTS
- 10. Steps i. diapedesis ii. hemostatic clot – formed by plateletes iii. fibrin clot formation – formed by fibroblasts Plateletes – 1st cells to produce essential cytokines which modulates most of the subsequent steps in wound healing
- 11. Early Events The early phase, which begins immediately following skin injury, involves cascading molecular and cellular events leading to hemostasis and formation of an early, makeshift extracellular matrix— providing structural support for cellular attachment and subsequent cellular proliferation.
- 12. Hemostasis : Vascular • Initial vasoconstriction (5-10 min) then vasodilation (persistent) • Exposure of sub endothelial von Willebrand / factor VII, and fibrillar collagen – platelet plug • Hageman factor (XII) – initiation of clotting cascade and fibrin clot formation
- 13. Hemostasis : Fibrin • Fibrin and fibronectin form a lattice that provides scaffold for migration of inflammatory, endothelial, and mesenchymal cells. • Fibronectin is produced by fibroblasts, has a dozen or so binding sites. • Binds cytokines • Its breakdown products stimulate angiogenesis.
- 14. Hemostasis : Clotting Cascade Intrinsic Pathway Surface Contact F XI F XIa F IX F IXa Platelet Factor 3 Factor F X Collagen FXII activator F XII F XIIa Ca2+ Ca2+ Extrinsic Pathway Tissue/Cell Defect F VIIa F VII Ca2+ F III (Tissue Thromboplastin) Ca2+ Factor F X Ca2+ Fibrin Fibrinogen monomers Fibrin polymers Crosslinked Fibrin Meshwork F XIIIa F XIII F Va F V F VIII F VIIIa Prothrombin I Factor F Xa Ca2+ Thrombin
- 15. Inflammation : Signs • Erythema • Edema • Pain • Heat Inflammation – migration of leukocytes into the wound. 1st 24 hours, polymorphonucleocytes followed by macrophages.
- 16. Inflammation : Physiological Changes • Immediately after injury, intense vasoconstriction leads to blanching, a process mediated by epinephrine, NE, and prostaglandins released by injured cells. • Vasoconstriction reversed after 10 min, by vasodilatation. • Now redness and warmth. • Vasodilatation mediated by histamine, linins, prostaglandins.
- 17. • Platelets – derived growth factor (PDGF), proteases and vasoactive substances such as serotonin and histamine • Polymorphonuclear leukocytes • Macrophages (replace PMNs after 5 days) • Fibroblasts (recruited by chemotactic factors released by the above cells)
- 18. Inflammation : Physiological Changes • As microvenules dilate, gaps form between the endothelial cells , resulting in vascular permeability. Plasma leaks out into extravascular space. • Leukocytes now migrate into the wound by diapedesis, adhere to endothelial cells, to wounded tissues. • Alteration in pH from breakdown products of tissue and bacteria, along with swelling causes the pain.
- 19. Inflammation : Physiological Changes • Neutrophils, macrophages and lymphocytes come into wound. • Neutrophils first on scene, engulf and clean up. Macrophages then eat them or they die releasing O2 radicals and destructive enzymes into wound. • Monocytes migrate into extravascular space and turn into macrophages. • Macrophages very important in normal wound healing.
- 20. Inflammation : Physiological Changes • Macrophages eat bacteria, dead tissue, secrete matrix metallo proteinases that break down damaged matrix. • Macrophages source of cytokines that stimulate fibroblast proliferation, collagen production. • Lymphocytes produce factors like FGF, EGF, IL-2. • At 48-72 hrs, macrophages outnumber neuts. • By days 5-7 few remain.
- 22. Intermediate Events As in the other phases of wound healing, steps in the proliferative phase do not occur in a series but rather partially overlap in time. •About two or three days after the wound occurs, fibroblasts begin to enter the wound site, marking the onset of the proliferative phase even before the inflammatory phase has ended.
- 23. Proliferation • Mesenchymal cell chemotaxis • Mesenchymal cell proliferation • Angiogenesis • Epithelialization Fibroplasia – increases wound strength, hence tissue integrity is restored. Within 10 hours after injury, there is increased wound collagen synthesis. Within 5-7 days, collagen has peaked and will decline gradually.
- 24. Proliferation • Fibroblasts are the major mesenchymal cells involved in wound healing, although smooth muscle cells are also involved. • Normally reside in dermis, damaged by wounding. • Macrophage products are chemotactic for fibroblasts. PDGF, EGF, TGF, IL-1, lymphocytes are as well.
- 25. Proliferation Angiogenesis reconstructs vasculature in areas damaged by wounding, stimulated by high lactate levels, acidic pH, decreased O2 tension in tissues. • Cytokines directly stimulate the endothelial cell migration and proliferation required for angiogenesis. Many are produced by Macs. • Fibroblast growth factor : FGF-1 is most potent angiogenic stimulant identified. Heparin important as cofactor, Transforming growth factor : TGF-alpha, beta, prostaglandins also stimulate.
- 26. Epithelialization The formation of granulation tissue in an open wound allows the re epithelialization phase to take place, as epithelial cells migrate across the new tissue to form a barrier between the wound and the environment. They advance in a sheet across the wound site and proliferate at its edges, ceasing movement when they meet in the middle.
- 27. Epithelialization • The process of epithelial renewal after injury. • Particularly important in partial thickness injuries, but plays a role in all healing. • Partial thickness wounds have epidermis and dermis damaged, with some dermis preserved. • Epithelial cells involved in healing come from wound edges and sweat glands, sebaceous glands in the more central portion of wound.
- 28. Re epithelization • Migration (wound edges, hair follicles, adnexa) • Proliferation (48-72 hours) • Sutured wounds have a layer of keratinocytes within 24-48 hours
- 29. Skin Anatomy • Epidermis is composed of multiple layers of epithelial cells superficial to the dermis. • The first layer above the dermis is the basal layer, which is composed of basaloid cells. • The cells become more elongated as you go to superficial stratum corneum. • Stratum corneum is mostly keratin and dead tissue.
- 30. Layers of Skin
- 31. Clean incision wound Epithelialization • In contrast in an incisional wound, cellular migration occurs over a short distance. • Incisional wounds are re-epithelialized in 24- 48h. • The sequence of events here are cellular detachment, migration, proliferation, differentiation.
- 32. Epithelialization • First 24h, basal cell layer thickens, then elongate, detach from basement membrane (BM) and migrate to wound as a monolayer across denuded area. • Generation of a provisional BM which includes fibronectin, collagens type 1 and 5. • Basal cells at edge of wound divide 48-72 h after injury. • Epithelial cells proliferation contributes new cells to the monolayer. Contact inhibition when edges come together.
- 33. LATE WOUND HEALING EVENTS
- 34. Late Wound Healing Events RE MODELING Cytokines –provides communication for cell to cell interaction. Roles include: 1. Regulation of Fibrosis 2. Healing of wounds and skin grafts. 3. Vascularization 4. Bone and Tendon Strengthening 5. Control of Malignancy
- 35. Collagen • Synthesized by fibroblasts beginning 3-5 days after injury. • Rate increases rapidly, and continues at a rapid rate for 2-4 weeks in most wounds. • As more collagen is synthesized, it gradually replaces fibrin as the primary matrix in the wound. • After 4 weeks, synthesis declines, balancing destruction by collagenase.
- 37. Keratinocytes • Fibronectin – Cross links to fibrin – matrix/scaffold for keratinocyte adhesion and migration – Functions as an early component of the extracellular matrix. – Binds to collagen and interacts with matrix glycosaminoglycans. – Has chemotactic properties for macrophages, fibroblasts and endothelial and epidermal cells. – Promotes opsonization and phagocytosis. – Forms a component of the fibronexus. – Forms scaffolding for collagen deposition • Collagenases and neutral proteases – debridement • Plasminogen activator – clot dissolution • Type V collagen • Requires moisture for epithelial migration
- 38. Granulation • Highly vascular network of glycoproteins, collagen and glycosaminoglycans • Fibroblasts – collagen – Elastin – Fibronectin – Sulfated and non-sulfated Glycosaminoglycans – Proteases • Inflammatory cells
- 39. Fibroplasia • Fibroblasts • Mainly Type III collagen first • Replaced by type I and II collagen • Hydroxylation of proline and lysine – Iron, copper, vitamin C – Cross linkage
- 40. Collagen • Synthesized by fibroblasts beginning 3-5 days after injury. • Rate increases rapidly, and continues at a rapid rate for 2-4 weeks in most wounds. • As more collagen is synthesized, it gradually replaces fibrin as the primary matrix in the wound. • After 4 weeks, synthesis declines, balancing destruction by collagenase.
- 42. Collagen • Age, tension, pressure and stress affect rate of collagen synthesis. • TGF-b stimulates it, glucocorticoids inhibit it. • 28 types identified. Type 1(80-90%) most common, found in all tissue. The primary collagen in a healed wound. • Type 3(10-20%) seen in early phases of wound healing. Type V smooth muscle, Types 2,11 cartilage, Type 4 in BM.
- 43. Collagen • Three polypeptide chains, right handed helix. • Most polypeptide chains used in collagen assembly are alpha chains.
- 45. Collagen • Every third AA residue is Glycine. • Another critical component is hydroxylation of lysine and proline within the chains. Hydroxyproline is necessary for this. Requires Vit C, ferrous iron, and alpha ketoglutarate as co-enzymes. • Steroids suppress much of this, resulting in underhydroxylated collagen, which is incapable of making strong cross-links leading to easy breakdown.
- 46. Wound Contraction • Contraction is a key phase of wound healing. If contraction continues for too long, it can lead to disfigurement and loss of function. • Fibroblasts, stimulated by growth factors, differentiate into myofibroblasts. Myofibroblasts, which are similar to smooth muscle cells, are responsible for contraction. Myofibroblasts contain the same kind of actin as that found in smooth muscle cells.
- 47. Contraction • Myofibroblasts • Fibronexus (Singer) – Connections between intracellular actin microfilaments and extracellular collagen, fibronectin, and between myofibroblasts – Transmits force along entire network – Centripetal contraction
- 48. Wound Contraction • Begins approximately 4-5 days after wounding. • Represents centripetal movement of the wound edge towards the center of the wound. • Maximal contraction occurs for 12-15 days, although it will continue longer if wound remains open.
- 49. Wound Contraction • The wound edges move toward each other at an average rate of 0.6 to .75 mm/day. • Wound contraction depends on laxity of tissues, so a buttocks wound will contract faster than a wound on the scalp or pretibial area. • Wound shape also a factor, square is faster than circular.
- 50. Wound Contraction • Contraction of a wound across a joint can cause contracture. • Appears in 2nd degree burns or skin loss
- 51. Wound Contraction • Can be limited by skin grafts, full better than split thickness. • The earlier the graft the less contraction. • Splints temporarily slow contraction.
- 52. TERMINAL WOUND HEALING EVENT
- 53. Neovascularization • Fibronectin • Macrophage derived angiogenic factor • Endothelial migration
- 54. Wound Remodeling • Increased tensile strength • Decreased bulk, and erythema • Replacement of fibronectin by collagen • Dehydration – Promotes further crosslinkage of collagen – Reorientation of collagen to parallel skin collagen.
- 55. Remodeling • After 21 days, net accumulation of collagen becomes stable. Bursting strength is only 15% of normal at this point. Remodeling dramatically increases this. • 3-6 weeks after wounding greatest rate of increase, so at 6 weeks you are at 80% to 90% of eventual strength and at 6mos 90% of skin breaking strength.
- 56. Remodeling • The number of intra and intermolecular cross-links between collagen fibers increases dramatically. • A major contributor to the increase in wound breaking strength. • Quantity of Type 3 collagen decreases replaced by Type 1 collagen • Remodeling continues for 12 mos, so scar revision should not be done prematurely.
- 57. HEALING AT DIFFERENT PART OF BODY
- 58. Healing at Different Part of Body Skin graft • donor site • Split (partial) thickness skin graft • Full thickness skin graft Skin Flap • Local flap • Distance flap Contraction : the process whereby there is spontaneous closure of full thickness skin wounds
- 59. Healing at Different Part of Body • Tendon – composed mainly of type I collagen with significant amounts of proteoglycan. After disruption tendon and sheath have to be sutured. – Connective Tissue Matrix Deposition : the process whereby fibroblasts are recruited to the site of injury and produce a new connective tissue matrix. The cross-linked collagen provides the strength and integrity to all tissue.
- 60. Healing at Different Part of Body Bone • Soft callus formation • Mineralized as cartilage • Replaced by osteoid or bone – beginning of remodeling
- 61. Healing at Different Part of Body Gastrointestinal Tract : Bowel anastomotic strength develops more rapidly than that of the skin. Major complications of intestinal anastomoses are a. leak b. disruption The submucosa provide the major strength in anastmotic closure because it contains the majority of the fibrous connective tissue. Contraction : constriction of tubular organs such as the CBD or esophagus.
- 62. Part II DISTURBANCES IN WOUND HEALING
- 63. Factors Affecting Wound Healing Local Factors Systemic Factors •Oxygenation •Infection •Foreign body •Venous sufficiency •Age and gender •Sex hormones •Stress •Ischemia •Diseases: diabetes, keloids, fibrosis, hereditary healing disorders, jaundice, uremia •Obesity •Medications: glucocorticoid steroids, non-steroidal anti-inflammatory drugs, chemotherapy •Alcoholism and smoking •Immunocompromised conditions: cancer, radiation therapy, AIDS •Nutrition
- 64. รายละเอียดของ Local Factors • Infection versus contamination • Infection is when number or virulence of bacteria exceed the ability of local defenses to control them. • 100,000 organisms per gram of tissue. • Foreign bodies, hematomas promote infection, impaired circulation, radiation.
- 65. รายละเอียดของ Systemic Factors • Smoking stimulates vasoconstriction. • Increases platelet adhesiveness • Limits O2 carrying capacity • Endothelial changes • Diminished amount of collagen deposition.
- 66. รายละเอียดของ Systemic Factors • Radiation damages the DNA of cells in exposed areas. • Fibroblasts that migrate into radiated tissues are abnormal. • Collagen is synthesized to an abnormal degree in irradiated tissue causing fibrosis. • Blood vessels become occluded. • Damage to hair and sweat glands • Vitamin A has been used to counteract this.
- 67. รายละเอียดของ Systemic Factors • Malnutrition • Cancer • Old Age • Diabetes- impaired neutrophil chemotaxis, phagocytosis. • Steroids and immunosuppression suppresses macrophage migration, fibroblast proliferation, collagen accumulation, and angiogenesis. Reversed by Vitamin A 25,000u per day.
- 68. เมื่อแผลไม่หายหรือหายช้า Consider the negative effects of 1. Endocrine diseases (eg, diabetes, hypothyroidism) 2. Hematologic conditions (eg, anemia, polycythemia, myeloproliferative disorders) 3. Cardiopulmonary problems (eg, chronic obstructive pulmonary disease , congestive heart failure)
- 69. เมื่อแผลไม่หายหรือหายช้า Consider the negative effects of 6. GI problems that cause malnutrition and vitamin deficiencies 7. Obesity 8. Peripheral vascular pathology (eg, atherosclerotic disease, chronic venous insufficiency, lymphedema)
- 71. Part III SET BALANCE
- 72. Hypertrophic Scars and Keloids • Excessive healing results in a raised, thickened scar, with both functional and cosmetic complications. • If it stays within margins of wound it is hypertrophic. Keloids extend beyond the confines of the original injury. • Dark skinned, ages of 2-40. Wound in the presternal or deltoid area, wounds that cross langerhans lines.
- 73. Growth Factors • Epidermal growth factor • Macrophage derived growth factor (MDGF) • Platelet derived growth factor (PDGF) • Thrombin • Insulin • Lymphokines
- 74. Plasminogen activator inhibitor • Found to be elevated in Keloid scars • PAI-1 -/- “knockout” mice show accelerated wound healing after cutaneous injury • PAI-1 seems to regulate fibrinolytic and proteolytic activity during the replacement of fibrin by collagen. • PAI-1 is upregulated in cultured fibroblasts in a hypoxic environment
- 75. Metalloproteinases & Tissue Inhibitor of Metalloproteinases • Regulatory role in fibroblasia and scarring – Found in high concentrations in fetal wounds – MMP/TIMP is higher in “scarless” fetal wounds – TGF-beta decreased the MMP/TIMP ratio by increasing TIMP – May promote more rapid epithelization
- 76. TGF Beta-1 • Higher concentrations and exaggerated response in keloid fibroblasts • When added to fetal wounds – thicker scars made.
- 78. Hypertrophic Scars and Keloids • Excessive healing results in a raised, thickened scar, with both functional and cosmetic complications. • If it stays within margins of wound it is hypertrophic. Keloids extend beyond the confines of the original injury. • Dark skinned, ages of 2-40. Wound in the presternal or deltoid area, wounds that cross langerhans lines.
- 79. Hypertrophic Scars and Keloids • Keloids more familial • Hypertrophic scars develop soon after injury, keloids up to a year later. Hypertrophic scars more likely to cause contracture over joint surface.
- 80. Hypertrophic Scars and Keloids Keloids Treatment i. Triamcinolone ii. Excision – high recurrence rate
- 82. Part IV Wound Closure
- 83. Types of Wound Closure Primary intention 1. Primary Closure approximate disrupted tissues by sutures, staples, or tapes. With time there will be a. synthesis b. deposition c. cross-linking of collagen to provide the tissue with strength.
- 84. Types of Wound Closure Secondary intention 2. Delayed Primary Closure – also called tertiary closure. Wound closure is delayed for several days to prevent wound infection where there is: a. bacterial contamination b. foreign bodies c. extensive tissue trauma * Cleaning of the wound is done daily using NSS
- 85. Primary intention Examples: well-repaired lacerations, well reduced bone fractures, healing after flap surgery • involves epidermis and dermis without total penetration of dermis healing by process of epithelialization • When wound edges are brought together so that they are adjacent to each other (re-approximated)
- 86. Primary intention Examples: well-repaired lacerations, well reduced bone fractures, healing after flap surgery • Minimizes scarring • Most surgical wounds heal by primary intention healing • Wound closure is performed with sutures (stitches), staples, or adhesive tape
- 87. Secondary intention Examples: gingivectomy, gingivoplasty, tooth extraction sockets, poorly reduced fractures. • The wound is allowed to granulate • Surgeon may pack the wound with a gauze or use a drainage system • Granulation results in a broader scar
- 88. Secondary intention Examples: hepatectomy, loss skin open wound, burn • Healing process can be slow due to presence of drainage from infection • Wound care must be performed daily to encourage wound debris removal to allow for granulation tissue formation
- 89. Types of Wound Closure Delayed primary closure or secondary suture 3. Spontaneous Closure - wound closes by contraction of the wound edges.
- 90. Tertiary intention (Delayed primary closure or secondary suture) Examples: healing of wounds by use of tissue grafts. • The wound is initially cleaned, debrided and observed, typically 4 or 5 days before closure. • The wound is purposely left open
- 91. Tertiary intention • If the wound edges are not re approximated immediately, delayed primary wound healing transpires. • This type of healing may be desired in the case of contaminated wounds. By the fourth day, phagocytosis of contaminated tissues is well underway, and the processes of epithelization, collagen deposition, and maturation are occurring.
- 92. Tertiary intention • Foreign materials are walled off by macrophages that may metamorphose into epithelioid cells, which are encircled by mononuclear leukocytes, forming granulomas. • Usually the wound is closed surgically at this juncture, and if the "cleansing" of the wound is incomplete, chronic inflammation can ensue, resulting in prominent scarring.
- 93. PART V WOUND CARE
- 94. Basic Elements of Wound Care • Cleanse Debris from the Wound • Possible Debridement • Absorb Excess Exudate • Promote Granulation and Epithelialization When Appropriate • Possibly Treat Infections • Minimize Discomfort
- 95. Prevention • Inspect skin • Moisture control • Proper positioning and transfer techniques • Nutrition • Avoid pressure on heels and bony prominences • Use of positioning devices
- 96. Risk Assessment • Nutritional status • Alteration in sensation • Co-morbid conditions • Medications that delay healing • Decreased blood flow
- 97. Assessment & Monitor • Location • Stage and Size • Periwound • Undermining • Tunneling • Exudate • Color of wound bed • Necrotic Tissue • Granulation Tissue • Effectiveness of Treatment
- 98. Types of Wounds • Surgical Wounds • Pressure Ulcers • Arterial Insufficiency • Diabetic Ulcers • Venous Insufficiency • Tumors
- 99. Wet or Dry Dressings • Causes Injury to New Tissue Growth • Is Painful • Predisposes Wound to Infection • Becomes a Foreign Body • Delays Healing Time
- 100. Frequency • Goal is to minimize the frequency of dressing change • Daily dressing changes increase chances of infection and disrupts the healing of tissue Decrease Frequency of Dressing Changes
- 101. General treatment of non healing wounds • Successful treatment of difficult wounds requires assessment of the entire patient and not just the wound. • Systemic problems often impair wound healing; conversely, non healing wounds may herald systemic pathology.
- 102. Successful treatment of wounds • Characterize the wound – chemotherapeutic drugs inhibit wound healing • Ensure adequate oxygenation • Ensure adequate nutrition – (malnutrition affects wound healing by inhibiting the immune response (opsonization) – Address protein-calorie malnutrition and deficiencies of vitamins and minerals e.g. Vitamin C, E, Zinc)
- 103. Successful treatment of wounds • Treat infection – Bowel anastomotic strength develops more rapidly than that of the skin. The submucosa provide the major strength in anastmotic closure because it contains the majority of the fibrous connective tissue. • Remove foreign bodies • Irrigate, Provide a moist (not wet) wound bed
- 105. Part VI SUTURE MATERIALS AND TECHNIQUES
- 106. The Ideal Suture Material • Can be used in any tissue • Easy to handle • Good knot security • Minimal tissue reaction
- 107. The Ideal Suture Material • Unfriendly to bacteria • Strong yet small • Won’t tear through tissues • Cheap
- 108. What’s It Used for? • To bring tissue edges together and speed wound healing (=tissue apposition) • Orthopedic surgery to help stabilize joints – Repair ligaments • Ligate vessels or tissues
- 109. Types of Needles • Eyed needles – More Traumatic – Only thread through once – Suture on a reel – Tends to unthread itself easily
- 110. Types of Needles • Swaged-on needles – Much less traumatic – More expensive suture material – Sterile
- 111. Points of Needles • Taper – Atraumatic – Internal organs
- 112. Points of Needles Cutting • Cutting edge on inside of circle • Skin • Traumatic
- 113. Points of Needles Reverse Cutting • Cutting edge on outside of circle • Skin • Less traumatic than cutting
- 114. Cutting vs Reverse Cutting • Cutting • Reverse cutting
- 115. Shapes of Needles • 3/8 circle • 1/2 circle • Straight • Specialty
- 116. Characteristics of Suture Material • Absorbable VS. Non - absorbable • Monofilament VS. Multifilament • Natural or Synthetic
- 117. Absorbable Sutures • Internal • Intradermal/ subcuticular • Rarely on skin
- 118. Non-absorbable Suture • Primarily Skin – Needs to be removed later • Stainless steel = exception – Can be used internally • Ligature • Orthopedics – Can be left in place for long periods
- 120. Reading the Suture Label Size Needle • Company Order Code Name Also: LENGTH NEEDLE SYMBOL COLOR Absorbable or Non
- 121. Choosing Absorbable Vs. Nonabsorbable • How long you need it to work • Do you want to see the animal again for suture removal
- 122. Monofilament Vs. Multifilament • memory easy to handle • less tissue drag more tissue drag • doesn’t wick wicks/ bacteria • poor knot security good knot security • - tissue reaction +tissue reaction
- 123. Natural Vs. Synthetic • Natural: – Gut – Chromic Gut – Silk – Collagen • All are absorbable
- 124. Gut/ Chromic Gut • Made of submucosa of small intestines • Multifilament / Mono filament • Breaks down by phagocytosis: inflammatory reaction common
- 125. Gut/ Chromic Gut • Chromic: tanned, lasts longer, less reactive • Easy handling • Plain: 3-5 days • Chromic: 10-15 days • Bacteria love this stuff!
- 126. Collagen and Silk • Natural sutures • VERY reactive, absorbable • Collagen : Ophthalmic surgery only
- 127. Synthetic Vicryl (Polyglactin 910) • Braided, synthetic, absorbable • Stronger than gut: retains strength 3 weeks • Broken down by enzymes, not phagocytosis • Break-down products inhibit bacterial growth – Can use in contaminated wounds, unlike other multifilaments
- 128. Synthetic Dexon and PGA • Polymer of glycolic acids • Braided, synthetic, absorbable • Broken down by enzymes • Both PGA and dexon have increased tissue drag, good knot security • Both are stronger than gut
- 129. Synthetic PDS (polydioxine) • Monofilament (less drag, worse knot security – lots of “memory”) • Synthetic, absorbable • Very good tensile strength (better than gut, vicryl, dexon) which lasts months • Absorbed completely by 182 days
- 130. Synthetic Maxon (polyglyconate) • Monofilament- memory • Synthetic Absorbable • Very little tissue drag • Poor knot security • Very strong
- 131. NONABSORBABLE SUTURES • Natural or Synthetic • Monofilament or multifilament
- 132. NYLON • Synthetic • Mono or Multifilament • Memory • Very little tissue reaction • Poor knot security
- 133. Polypropylene • Prolene, Surgilene • Monofilament, Synthetic • Won’t lose tensile strength over time • Good knot security • Very little tissue reaction
- 134. Stainless Steel • Monofilament • Strongest ! • Great knot security • Difficult handling • Can cut through tissues • Very little tissue reaction, won’t harbor bacteria
- 135. Suture Sizes • Sized #5-4-3-2-1-0-00-000- 0000…30-0 BIGGER >>>>>>>>>>>>>>>>SMALLER • 00 = 2-0, “two ought” • SA : 0 through 3-0 (Optho 5-0 >>7-0) • LA : 0 through 3
- 136. Suture Sizes (cont) • Stainless Steel – In gauges (like needles) • Smaller gauge = bigger, stronger • Larger gauge= smaller, finer – 26 gauge = “ought” – 28 gauge = 2-0
- 137. Skin Staples • Very common in human medicine • Expensive • Very easy • Very secure • Very little tissue reaction • Removal = – Special tool required
- 138. Staples • Rapid closure of wound • Easy to apply • Evert tissue when placed properly
- 139. Tissue Adhesive • Nexaband, Vetbond, and others • Little strength • Should not be placed between skin layers or inside body
- 140. Steri-strips • Sterile adhesive tapes • Available in different widths • Frequently used with subcuticular sutures • Used following staple or suture removal • Can be used for delayed closure
- 142. Suture Patterns
- 143. Knot Strength • Generally 4 “throws” for >90% knot security (nylon may need 5) – Less “throws” = more likely to untie itself • Stainless steel = exception again – 2 “throws” = 99% knot security
- 144. Two-Hand Square Knot • Easiest and most reliable • Used to tie most suture materials
- 145. Instrument Tie • Useful when one or both ends of suture material are short • Commonly used technique for laceration repair
- 146. Wound Closure • Basic suturing techniques: – Simple sutures – Mattress sutures – Subcuticular sutures • Goal: “approximate, not strangulate”
- 149. Simple Sutures Simple interrupted stitch – Single stitches, individually knotted (keep all knots on one side of wound) – Used for uncomplicated laceration repair and wound closure
- 151. Simple Interrupted
- 152. Simple Continuous
- 153. Mattress Sutures • Horizontal mattress stitch – Provides added strength in fascial closure; also used in calloused skin (e.g. palms and soles) – Two-step stitch: • Simple stitch made • Needle reversed and 2nd simple stitch made adjacent to first (same size bite as first stitch)
- 154. Mattress Sutures Vertical mattress stitch – Affords precise approximation of skin edges with eversion – Two-step stitch: • Simple stitch made – “far, far” relative to wound edge (large bite) • Needle reversed and 2nd simple stitch made inside first – “near, near” (small bite)
- 156. Subcuticular Sutures • Usually a running stitch, but can be interrupted • Intradermal horizontal bites • Allow suture to remain for a longer period of time without development of crosshatch scarring
- 157. Subcuticular
- 161. chuchai.s@nhso.go.th chuchai.sn@gmail.com facebook line : Morchuchai
Notas del editor
- PDGF platelet derived growth factor – D induce macrophage recruitment, increase interstitial pressure ,and blood vessel maturation during angiogenesis. EGF epidermal growth factor mediate monocyte chemotaxis and macropage proliferation to sub endothelial space. TGF transforming growth factor β enhance macrophage ability to produce IL 10
- FGF 1 fibroblast growth factor family member possess broad mitogenic and cell survival activities, and are involve in a variety of biological processes including embryogenic development , cell growth, morphogenesis, tissue repair, tumor growth and invasion.
- Collagen is a group of naturally occurring protein found in animals, especially in the fresh and connective tissues of mammals. It is making up about 25% - 35% of the whole body protein content. Collagen , in the form elongated fibril , is mostly found infibrous tissue. The fibroblast is the most common cell which create collagen. Over 90% of collagen is of type 1.So far 28 type of collagen have been identified and described. Type 1 skin , tendon, vascular ligature, organs, bone. 2 cartilage.3 reticulate 4 base of cell basement membrane. 5 cell surface and placenta.
- opsonization and phagocytosis a process call antibodies dependent cellular cytotoxicity
- Poly glycolic acid
