Weightloss2-1-1
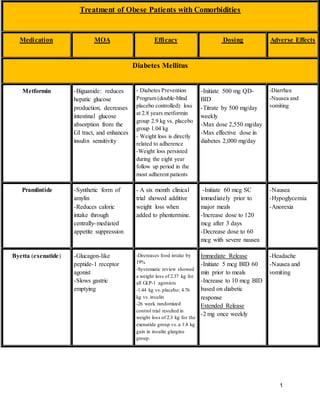
- 1. 1 Treatment of Obese Patients with Comorbidities Medication MOA Efficacy Dosing Adverse Effects Diabetes Mellitus Metformin -Biguanide: reduces hepatic glucose production, decreases intestinal glucose absorption from the GI tract, and enhances insulin sensitivity - Diabetes Prevention Program (double-blind placebo controlled): loss at 2.8 years metformin group 2.9 kg vs. placebo group 1.04 kg - Weight loss is directly related to adherence -Weight loss persisted during the eight year follow up period in the most adherent patients -Initiate 500 mg QD- BID -Titrate by 500 mg/day weekly -Max dose 2,550 mg/day -Max effective dose in diabetes 2,000 mg/day -Diarrhea -Nausea and vomiting Pramlintide -Synthetic form of amylin -Reduces caloric intake through centrally-mediated appetite suppression - A six month clinical trial showed additive weight loss when added to phentermine. -Initiate 60 mcg SC immediately prior to major meals -Increase dose to 120 mcg after 3 days -Decrease dose to 60 mcg with severe nausea -Nausea -Hypoglycemia -Anorexia Byetta (exenatide) -Glucagon-like peptide-1 receptor agonist -Slows gastric emptying -Decreases food intake by 19% -Systematic review showed a weight loss of 2.37 kg for all GLP-1 agonists -1.44 kg vs.placebo; 4.76 kg vs.insulin -26 week randomized control trial resulted in weight loss of 2.3 kg for the exenatide group vs.a 1.8 kg gain in insulin glargine group. Immediate Release -Initiate 5 mcg BID 60 min prior to meals -Increase to 10 mcg BID based on diabetic response Extended Release -2 mg once weekly -Headache -Nausea and vomiting
- 2. 2 Victoza (liraglutide) Saxenda (liraglutide) -Glucagon-like peptide-1 receptor agonist -Saxenda is FDA approved for weight loss -20 week European trial evaluated various daily doses and weight loss effect: 1.2 mg (4.8 kg), 1.8 mg (5.5 kg), 2.4 mg (6.3 kg), 3.0 mg (7.2 kg) vs. 2.8 kg in the placebo group vs. 4.1 kg in the orlistat group -76% of 3.0 mg treatment group achieved >5% weight loss Chronic Weight Management: -Initiate 0.6 mg SC QD for one week -Increase dose by 0.6 mg weekly to a target 3.0 mg Diabetes Treatment: -Initiate 0.6 mg SC QD for one week -Increase to 1.2 mg QD -Max 1.8 mg QD -Headache -Nausea and vomiting Depression, Epilepsy, Migraine Bupropion -Inhibitor of norepinephrine and dopamine reuptake -Reduces food intake by acting on adrenergic and dopaminergic receptors in the hypothalamus -Reduced body weight by 6.2% (300 mg/day) and 7.2% (400 mg/day) -Dose varies based on comorbidity and salt form -Tachycardia -Headache -Agitation Topiramate -Reduces appetite by enhancing GABA activity -n=385 patients randomized to five groups (64 mg/d, 96 mg/d, 192 mg/d, 384 mg/day, or placebo) resulted in weight loss of 7.3% of initial weight at six months -Weight loss is dose related -Dose varies based on comorbidity -Used in combination with phentermine for weight loss: Phentermine/Topiramate 3.75/23 mg (initial), 7.5/46 mg (recommended), 15/92 mg (max) -Paresthesias -Somnolence -Difficulty with concentration, memory, and attention
- 3. 3 Current Drugs Approved for Treatment of Obesity Medication MOA Efficacy Dosing/Monitoring Adverse Effects Phentermine -Phentermine is a sympathomimetic amine producing CNS stimulation. -The mechanism by which it produces weight loss is primarily by appetite suppression. -First approved by the FDA in 1959. -Weight loss at 6 months with Phentermine 15 mg was 4.6% of total body weight (n=756), and 8.1 +/- 3.9 kg at 12 weeks (n=37). Short-term: -15-30 mg PO QD -18.75 mg PO BID -37.5 mg PO BID -Increased blood pressure -Increased heart rate -Appetite suppression -Xerostomia -Insomia -Irritability -Nervousness Belviq (lorcaserin) -Selectively targets and binds to the serotonin 2c receptors (agonist), thereby promoting satiety and decreased food intake. BLOOM, BLOSSOM, BLOOM-DM enrolled patients with a BMI of >/= 27. At 48 weeks, weight loss was ~10 kg in the lorcaserin group, and weight maintenance was demonstrated versus placebo. -HbA1c decreased by 0.9 +/- 0.06, FBG decreased by 27.4 +/- 2.5 mg/dL. -10 mg PO BID -Discontinue at 12 weeks of therapy if only </= 5% weight loss -Contraindicated in pregnancy. -Monitoring for potential development of valvulopathy. -Nausea -Dizziness -Fatigue -Heart valve disorder -Hypoglycemia -Suicidal ideation -Serotonin syndrome when used with SSRIs or MAOIs
- 4. 4 Qsymia (phentermine/ topiramate ER) -Dual mechanism of appetite suppression/decreased food intake, and increased satiety via enhanced catecholamine release. -EQUIP and CONQUER trial reported weight losses approaching 10%, and maintained weight loss of 9.3% below baseline at the end of two years at 7.5/46 mg. Patients taking 15/92 mg maintained 10.7% weight loss after two years. -Improvements in cardiovascular risk factors, such as blood pressure, triglycerides, and blood glucose. - 3.75/23 mg QAM for 14 days, then increase to 7.5/46 mg QD. -Evaluate weight loss at 12 weeks of therapy, and discontinue or escalate the dose if <3% weight loss. -To escalate the dose increase phentermine dose to 11.25/69 mg for 14 days, then 15/92 mg for 12 weeks. -If <5% weight loss, gradually discontinue. -Paresthesias -Dizziness -Dysgeusia -Insomnia -Constipation -Xerostomia -Negative pregnancy test before and during treatment, as topiramate is associated with a risk of oral cleft in the developing fetus. Orlistat (tetrahydrolipstatin) -Orlistat is a selective inhibitor of lipases in the stomach and intestines that ultimately reduces the intestinal digestion of fat. -539 adolescents received 120 mg TID with a mean net weight loss -2.87 kg (-3.21 to - 2.53) 95% CI. -120 mg PO TID (prescription) -60 mg PO TID (OTC) -Recommended to eat a balanced diet containing at least 30% calories from fat. -Fecalfat loss and increased urge to defecate -Small decreases in fat soluble vitamins -Rare cases of idiosyncratic liver injury. Contrave (naltrexone/bupropion) -Reduction of food intake by inhibiting the reuptake of dopamine and norepinephrine, increasing firing in the appetite control center. -Weight loss at one year with this combination was intermediate between Qsymia and Belviq. -No increase in cardiovascular events to date. -Initial dose escalation, naltrexone 8 mg/bupropion 90 mg (1 tablet) PO QAM for week 1; then 1 tablet twice daily, QAM and QPM,for week 2; then 2 tablets QAM and 1 tablet QPM for week 3; maintenance dose, week 4 and thereafter,2 tablets PO BID, for a total daily dosage of naltrexone 32 mg/bupropion 360 mg - Daily doses greater than naltrexone 32 mg/bupropion 360 mg not recommended. -Assess weight loss at 12 -Increased blood pressure -Increased pulse