Heat of Reaction
•
1 recomendación•1,623 vistas
M
M BRExperiment to determine heat of reaction
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
IB Chemistry Kinetics Design IA and uncertainty calculation for Order and Rate

IB Chemistry Kinetics Design IA and uncertainty calculation for Order and Rate
Preparation of tetraaminecopper(ii) sulphate complex

Preparation of tetraaminecopper(ii) sulphate complex
Preparation of potassium trioxalatoaluminate(III) trihydrate

Preparation of potassium trioxalatoaluminate(III) trihydrate
Preparation of tris(ethylenediamine)nickel(ii) chloride complex.

Preparation of tris(ethylenediamine)nickel(ii) chloride complex.
Destacado
Destacado (20)
2010 05 02 10 Dr Daniel C Tsui Physics Nobel Prize King Carl Xvi Gustaf Of S...

2010 05 02 10 Dr Daniel C Tsui Physics Nobel Prize King Carl Xvi Gustaf Of S...
Similar a Heat of Reaction
Similar a Heat of Reaction (20)
To Determine the Enthalpy of H2O2 Decomposition in Aqueous Solution

To Determine the Enthalpy of H2O2 Decomposition in Aqueous Solution
To estimate the amount of zinc as zn2 p2o7 in the given solution of zinc sulp...

To estimate the amount of zinc as zn2 p2o7 in the given solution of zinc sulp...
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
List out the chemical reactions you observe in your surroundings 

List out the chemical reactions you observe in your surroundings
LabQuest 18 Chemistry with Vernier 18 - 1 Additi.docx

LabQuest 18 Chemistry with Vernier 18 - 1 Additi.docx
Chem 162 Lab 7 Freezing Point Depression Lab- .docx

Chem 162 Lab 7 Freezing Point Depression Lab- .docx
Más de M BR
Más de M BR (13)
Último
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Último (20)
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
9548086042 for call girls in Indira Nagar with room service

9548086042 for call girls in Indira Nagar with room service
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Heat of Reaction
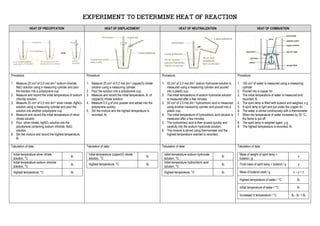
- 1. EXPERIMENT TO DETERMINE HEAT OF REACTION HEAT OF PRECIPITATION HEAT OF DISPLACEMENT HEAT OF NEUTRALIZATION HEAT OF COMBUSTION Procedure: 1. Measure 25 cm3 of 0.5 mol dm-3 sodium chloride, NaCl solution using a measuring cylinder and pour the solution into a polystyrene cup. 2. Measure and record the initial temperature of sodium chloride solution. 3. Measure 25 cm3 of 0.5 mol dm-3 silver nitrate, AgNO3 solution using a measuring cylinder and pour the solution into another polystyrene cup. 4. Measure and record the initial temperature of silver nitrate solution. 5. Pour silver nitrate, AgNO3 solution into the polystyrene containing sodium chloride, NaCl solution. 6. Stir the mixture and record the highest temperature, θ3 Procedure: 1. Measure 25 cm3 of 0.2 mol dm-3 copper(II) nitrate solution using a measuring cylinder. 2. Pour the solution into a polystyrene cup. 3. Measure and record the initial temperature, θ1 of copper(II) nitrate solution. 4. Measure 0.5 g of zinc powder and added into the polystyrene quickly. 5. Stir the mixture and the highest temperature is recorded, θ2 Procedure: 1. 50 cm3 of 2.0 mol dm-3 sodium hydroxide solution is measured using a measuring cylinder and poured into a plastic cup. 2. The initial temperature of sodium hydroxide solution is measured after a few minutes. 3. 50 cm3 of 2.0 mol dm-3 hydrochloric acid is measured using another measuring cylinder and poured into a plastic cup. 4. The initial temperature of hydrochloric acid solution is measured after a few minutes. 5. The hydrochloric acid is then poured quickly and carefully into the sodium hydroxide solution. 6. The mixture is stirred using thermometer and the highest temperature reached is recorded. Procedure: 1. 100 cm3 of water is measured using a measuring cylinder. 2. Poured into a copper tin. 3. The initial temperature of water is measured and recorded, θ1 . 4. The spirit lamp is filled with butanol and weighed, x g 5. A spirit lamp is light and put under the copper tin. 6. The water is stirred continuously with a thermometer 7. When the temperature of water increased by 30 °C, the flame is put off 8. The spirit lamp is weighed again, y g 9. The highest temperature is recorded, θ2 Tabulation of data: Initial temperature silver nitrate solution, °C θ1 Initial temperature sodium chloride solution, °C θ2 Highest temperature, °C θ3 Tabulation of data: Initial temperature copper(II) nitrate solution, °C θ1 Highest temperature, °C θ3 Tabulation of data: Initial temperature sodium hydroxide solution, °C θ1 Initial temperature hydrochloric acid solution, °C θ2 Highest temperature, °C θ3 Tabulation of data: Mass of weight of spirit lamp + butanol / g x Final mass of spirit lamp + butanol / g y Mass of butanol used / g x – y = z Highest temperature of water / °C θ2 Initial temperature of water / °C θ1 Increased in temperature / °C θ2 - θ1 = θ3