Periodic classification of elements gr10, 2020-21
•Descargar como PPTX, PDF•
9 recomendaciones•549 vistas
For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell you about the history of periodic classification of elements. HOPE YOU ENJOY IT. NEXT POST ON: WHY DO WE WEIGH LESS ON THE MOON ?
Denunciar
Compartir
Denunciar
Compartir
Recomendados
Chemistry notes class 11 chapter 3 classification of elements and periodicity...

This document provides an overview of the history of classifying elements and the development of the periodic table. It discusses early classification systems proposed by Prout, Dobereiner, Newlands and others. Meyer proposed classifying elements based on an atomic volume curve. Mendeleev created the first recognizable periodic table in 1869, which was improved upon over time. Key periodic properties like atomic radius, ionization energy, electronegativity and valency are explained in relation to an element's position on the periodic table. The modern periodic table is based on atomic number and has 18 groups and 7 periods.
School of elements

The document discusses the history and development of the periodic table from Dobereiner's law of triads to Mendeleev's periodic table to the modern periodic table. It summarizes key features of the modern periodic table including its organization into periods and groups based on atomic number, trends in properties from period to period and group to group, and classification of elements into s, p, d and f blocks based on electron configuration.
Chapter - 5, Periodic Classification of Elements, Science, Class 10

I have expertise in making educational and other PPTs. Email me for more PPTs at a very reasonable price that perfectly fits in your budget.
Email: parmarshivam105@gmail.com
Chapter-5, Periodic Classification of Elements, Science Class10
CLASSIFICATION
DOBEREINER’S TRIAD
LIMITATIONS
NEWLAND’S LAW OF OCTAVES
CHARACTERISTICS OF LAW OF OCTAVES
LIMITATIONS OF NEWLANDS LAW OF OCTAVES
MENDELEEV’S PERIODIC TABLE
PROPERTIES OF GROUPS STUDIED BY MENDELEEV
LIMITATIONS OF MENDELEEV’S
PERIODIC TABLE
MERITS OF MENDELEEV’S PERIODIC CLASSIFICATION
MODERN PERIODIC LAW
PROPERTIES OF MODERN PERIODIC TABLE
NOBLE GASES
POSITION OF ELEMENTS IN THE MODERN PERIODIC -TABLE
METALS
NON-METALS
METALLOIDS
TRENDS IN MODERN PERIODIC TABLE
Every topic of this chapter is well written concisely and visuals will help you in understanding and imagining the practicality of all the topics.
By Shivam Parmar (Entrepreneur & Teacher)
The Periodic Table

The document summarizes key aspects of the periodic table. It describes how the periodic table is organized into rows called periods and columns called families or groups. Elements in the same group have similar properties. Metals are on the left and center of the table, while non-metals are on the right. Metalloids separate metals and non-metals. Chemical families have elements with similar properties, such as alkali metals in Group 1 reacting easily with water and air. The number of valence electrons increases from left to right across a period and decreases down a group.
Carbon and its compounds

Carbon is a nonmetallic element that forms covalent bonds and exists in multiple allotropes depending on how its atoms are arranged. It can form linear, branched, or cyclic hydrocarbons ranging from methane with one carbon to complex molecules like cellulose. Key properties depend on factors like the number of carbon atoms, their arrangement into straight chains, branches, or rings, and whether bonds are single or double. Functional groups determine characteristic reactions by specific atoms within organic molecules.
The periodic table

The document summarizes key aspects of the periodic table, including its discovery by Dmitri Mendeleev who predicted undiscovered elements, and the periodic law stating elements' properties repeat periodically with their atomic number. It describes the main categories of elements as metals, nonmetals, and metalloids, and explains parts of the periodic table including periods and groups. It provides details on each group's properties including electron configuration, reactivity, and shared physical traits.
1.Periodic Properties & Variations of Properties Class 10 ,ICSE.pptx

(i) Periodic properties and their variations in groups and periods.
(ii) Periodicity on the basis of atomic number for elements.
1.atomic size
2. ionisation POTENTIAL/
3,electron affinity
4. metals
non metals
5. density
Periodic Trends
Periods
groups
Mendeleev periodic table
Moseley periodic table
long form of periodic table
Atoms and molecules

Atoms are the smallest particles that make up all matter. John Dalton's atomic theory states that all matter is made of tiny indivisible particles called atoms. Atoms of different elements have different masses and chemical properties. Two or more atoms can combine to form molecules, which are the smallest units that retain the properties of a substance. Molecules are formed when atoms bond together via chemical bonds and are the smallest particles that can exist independently. Common examples of molecules include water (H2O) and oxygen (O2).
Recomendados
Chemistry notes class 11 chapter 3 classification of elements and periodicity...

This document provides an overview of the history of classifying elements and the development of the periodic table. It discusses early classification systems proposed by Prout, Dobereiner, Newlands and others. Meyer proposed classifying elements based on an atomic volume curve. Mendeleev created the first recognizable periodic table in 1869, which was improved upon over time. Key periodic properties like atomic radius, ionization energy, electronegativity and valency are explained in relation to an element's position on the periodic table. The modern periodic table is based on atomic number and has 18 groups and 7 periods.
School of elements

The document discusses the history and development of the periodic table from Dobereiner's law of triads to Mendeleev's periodic table to the modern periodic table. It summarizes key features of the modern periodic table including its organization into periods and groups based on atomic number, trends in properties from period to period and group to group, and classification of elements into s, p, d and f blocks based on electron configuration.
Chapter - 5, Periodic Classification of Elements, Science, Class 10

I have expertise in making educational and other PPTs. Email me for more PPTs at a very reasonable price that perfectly fits in your budget.
Email: parmarshivam105@gmail.com
Chapter-5, Periodic Classification of Elements, Science Class10
CLASSIFICATION
DOBEREINER’S TRIAD
LIMITATIONS
NEWLAND’S LAW OF OCTAVES
CHARACTERISTICS OF LAW OF OCTAVES
LIMITATIONS OF NEWLANDS LAW OF OCTAVES
MENDELEEV’S PERIODIC TABLE
PROPERTIES OF GROUPS STUDIED BY MENDELEEV
LIMITATIONS OF MENDELEEV’S
PERIODIC TABLE
MERITS OF MENDELEEV’S PERIODIC CLASSIFICATION
MODERN PERIODIC LAW
PROPERTIES OF MODERN PERIODIC TABLE
NOBLE GASES
POSITION OF ELEMENTS IN THE MODERN PERIODIC -TABLE
METALS
NON-METALS
METALLOIDS
TRENDS IN MODERN PERIODIC TABLE
Every topic of this chapter is well written concisely and visuals will help you in understanding and imagining the practicality of all the topics.
By Shivam Parmar (Entrepreneur & Teacher)
The Periodic Table

The document summarizes key aspects of the periodic table. It describes how the periodic table is organized into rows called periods and columns called families or groups. Elements in the same group have similar properties. Metals are on the left and center of the table, while non-metals are on the right. Metalloids separate metals and non-metals. Chemical families have elements with similar properties, such as alkali metals in Group 1 reacting easily with water and air. The number of valence electrons increases from left to right across a period and decreases down a group.
Carbon and its compounds

Carbon is a nonmetallic element that forms covalent bonds and exists in multiple allotropes depending on how its atoms are arranged. It can form linear, branched, or cyclic hydrocarbons ranging from methane with one carbon to complex molecules like cellulose. Key properties depend on factors like the number of carbon atoms, their arrangement into straight chains, branches, or rings, and whether bonds are single or double. Functional groups determine characteristic reactions by specific atoms within organic molecules.
The periodic table

The document summarizes key aspects of the periodic table, including its discovery by Dmitri Mendeleev who predicted undiscovered elements, and the periodic law stating elements' properties repeat periodically with their atomic number. It describes the main categories of elements as metals, nonmetals, and metalloids, and explains parts of the periodic table including periods and groups. It provides details on each group's properties including electron configuration, reactivity, and shared physical traits.
1.Periodic Properties & Variations of Properties Class 10 ,ICSE.pptx

(i) Periodic properties and their variations in groups and periods.
(ii) Periodicity on the basis of atomic number for elements.
1.atomic size
2. ionisation POTENTIAL/
3,electron affinity
4. metals
non metals
5. density
Periodic Trends
Periods
groups
Mendeleev periodic table
Moseley periodic table
long form of periodic table
Atoms and molecules

Atoms are the smallest particles that make up all matter. John Dalton's atomic theory states that all matter is made of tiny indivisible particles called atoms. Atoms of different elements have different masses and chemical properties. Two or more atoms can combine to form molecules, which are the smallest units that retain the properties of a substance. Molecules are formed when atoms bond together via chemical bonds and are the smallest particles that can exist independently. Common examples of molecules include water (H2O) and oxygen (O2).
Metals and Non Metals

Topics Included
• Introduction
• Metals
→ Physical properties of metals
→ Chemical Properties of metals
• Non-metals
→ Physical properties of non-metals
→ Chemical Properties of metals
• Difference between metals and non-metals
• Reaction with Acids
• Reaction with Bases
Periodic table ppt cscope

The periodic table arranges the elements based on atomic number and chemical properties. It is divided into metals, nonmetals, and metalloids. The periodic table predicts chemical behavior, trends, and element properties. It organizes elements by atomic structure, number, and physical/chemical traits.
Periodic Properties of Elements (Class X CBSE & ICSE)

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands attempted to classify elements but their systems had limitations. Mendeleev created the first successful periodic table in 1869 by arranging elements by atomic mass and leaving gaps for undiscovered elements. The modern periodic table is arranged by atomic number based on the repeating properties of elements, and classifies elements into blocks and groups with similar properties. The periodic table provides an organized framework for studying and predicting the properties of all known elements.
The modern periodic table

The document discusses the structure and organization of the modern periodic table. Elements are arranged horizontally in order of increasing atomic number and grouped vertically into columns based on their electron configurations. The periodic table is divided into blocks including s-, p-, d- and f-blocks. An element's position in the table is determined by its atomic number and electron configuration, especially the outermost electrons.
Presentation on periodic table

Atomic radii decrease across a period as nuclear charge increases. Cations are smaller than their parent atoms. Among isoelectronic species, the one with the larger positive nuclear charge will have the smallest radius. Ionization energy generally increases across a period as it is more difficult to remove electrons, and decreases down a group as shielding increases. Electronegativity follows similar trends as ionization energy.
The periodic table

The document discusses the organization and classification of elements in the periodic table. It describes how Mendeleev organized the elements based on increasing atomic mass and similar properties. The modern periodic table is organized by increasing atomic number. Elements are classified into metals, nonmetals, and metalloids, which are grouped based on their location on the periodic table. Each group of elements has a characteristic number of valence electrons that determine their chemical properties.
Periodic classification of elements Grade-11

The document discusses the periodic classification of elements. It provides the atomic numbers, symbols, and atomic masses of the first 30 elements in the periodic table. It then discusses the key features and components of modern periodic tables, including that elements are arranged by atomic number rather than atomic mass. The modern periodic table contains 7 periods and 18 groups, with elements in the same group having the same number of electrons in their outer shell. It also notes the advantages of the modern periodic table over previous classifications.
Atoms, Molecules, And Matter

An atom is the smallest particle of an element that contains protons, neutrons, and electrons. Elements are substances made of a single type of atom that combine to form molecules, which are made of two or more atoms joined together. Molecules such as water, written as H2O with two hydrogen atoms and one oxygen atom, are the smallest particles that combine without chemical change to form all matter in the world.
Atoms, elements, compounds and mixtures.pptx

1) The document discusses atoms, elements, compounds, and mixtures. It aims to explain what an atom is, differentiate between elements, compounds and mixtures, and give examples of each.
2) Atoms are the basic building blocks of all matter and are very small. Elements are substances made of only one type of atom that cannot be broken down further.
3) Compounds are formed when two or more elements are chemically bonded together and have different properties than the original elements. Mixtures contain two or more substances that are not chemically bonded and can be separated.
Tissue class 9

Meristematic tissue is a plant tissue that is undifferentiated and can divide to form other tissues. It is found in three main regions: the apical meristem at the tips of stems and roots, the intercalary meristem at the bases of leaves and stems, and the lateral meristem or cambium responsible for increasing the girth of stems and roots. Permanent tissues are differentiated tissues that do not divide, such as parenchyma, collenchyma and sclerenchyma. Epithelial tissue forms protective layers in animals, such as the skin and lining of organs, while connective tissue binds and supports other tissues.
Elements

Elements are pure substances that cannot be broken down further through chemical or physical means. There are currently 118 known elements, with new ones occasionally being discovered through artificial nuclear reactions. Elements are identified by their atomic number, which represents the number of protons in the nucleus. Elements are the basic building blocks of all matter and are used to create thousands of materials, though some like plutonium are also used in nuclear weapons due to their radioactive properties.
Carbon and its compounds

This document is a PowerPoint presentation on carbon and its compounds submitted by a student group. It provides information on carbon including that it is a nonmetallic element with an atomic number of 6 that forms covalent bonds. It discusses the origin of carbon in nature, the reasons for its versatility including catenation and tetracovalency. The presentation also covers different forms of carbon including graphite, diamond and fullerenes as well as saturated and unsaturated compounds. It provides examples of functional groups and homologous series and discusses chemical properties and uses of carbon compounds.
The periodic table class 10

The document discusses the development of the periodic table. It describes early classification systems by Dobereiner and Newlands that grouped elements based on properties but had limitations as new elements were discovered. Mendeleev organized the elements into the first recognizable periodic table based on atomic mass and predicted properties of undiscovered elements. Moseley later modified this to be based on atomic number. The modern periodic table is arranged into periods and groups with trends in properties like atomic radius, ionization potential, and electronegativity explained by variation in nuclear charge and number of electron shells across and down the table.
Periodic table

The periodic table organizes 118 known elements and displays their symbols and names. It arranges elements in rows called periods and columns called groups based on their atomic structure and properties. Elements in the same group have the same number of outer electrons and similar chemical properties, while properties vary greatly across each period from very reactive solids to inactive gases. The periodic table provides a way to classify and compare elements according to their atomic number and mass.
Periodic Law

The document summarizes the history and development of the periodic table. It describes how early chemists like Cannizzaro, Mendeleev, and Moseley contributed to organizing the elements based on their atomic masses and properties. The modern periodic table is arranged by increasing atomic number and groups elements with similar properties together in periods and families. Key periodic properties like atomic radius, ionization energy, and electronegativity are influenced by an element's position on the table.
Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

The document discusses the history and development of the periodic table. It explains that:
1) Elements have been organized into the periodic table based on their atomic weights and properties in order to systematically study and predict chemical behavior.
2) Early periodic tables were proposed by scientists like Dobereiner, Newlands, and Mendeleev based on trends in atomic weights, though Mendeleev's 1869 table was the most successful in predicting new elements.
3) Modern periodic tables are arranged by atomic number according to the periodic law proposed by Moseley, with elements grouped into blocks based on their electron configurations. The table better explains trends in physical and chemical properties.
Inside An Atom

The document discusses the structure of atoms and their composition. It explains that atoms are composed of a tiny, dense nucleus containing protons and neutrons, surrounded by electrons in defined orbits. Niels Bohr revised earlier atomic models by suggesting that electrons occupy specific orbits and can jump between these orbits by absorbing or emitting energy. The arrangement of electrons in an atom is called its electronic configuration. Atoms of the same element can have different numbers of neutrons, forming isotopes with the same chemical properties but different physical properties. Isotopes have applications including use in nuclear power and medical treatments.
Periodic Classification of Elements and Periodicity

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands began classifying elements based on their properties, but that Dmitri Mendeleev created the first recognizable periodic table in 1871. His table arranged elements in order of atomic mass and predicted properties of undiscovered elements. Later, Moseley's discovery of atomic number allowed the modern periodic table to arrange elements by atomic number instead of mass. The document also discusses trends in properties within the periodic table.
Class 10. Chapter 5. Periodic Classification of Elements

This document summarizes key concepts from Chapter 5 on the periodic classification of elements:
1. Early scientists like Dobereiner and Newlands attempted to classify elements based on their properties but their systems did not comprehensively explain the trends.
2. Mendeleev organized the known elements into the first periodic table based on their atomic masses and properties, noting periodic trends. This helped predict new elements.
3. Modern periodic tables are organized by atomic number not mass, reflecting Moseley's discovery of each element's unique atomic number. Periodic trends exist in properties like size, valence, and metallicity across periods and groups.
Chemical Bonding

This document discusses chemical bonding and macromolecular structures. It begins by explaining the different types of bonds - ionic bonds formed between metals and non-metals by electron transfer, and covalent bonds formed between non-metals by electron sharing. It describes the properties of ionic and covalent compounds. It then discusses macromolecular structures found in substances like diamond, graphite and metals. It explains metallic bonding and compares the structures and properties of diamond and graphite. In the end, it discusses the different uses of diamond and graphite based on their properties.
Periodic calssification of elements ncert shashikumar b s

The document provides an overview of the periodic classification of elements. It discusses early attempts at classification including Dobereiner's triads and Newlands' law of octaves. It then covers Mendeleev's periodic table, including its merits and defects. Finally, it describes the modern periodic table based on atomic number, including periodic trends in properties like atomic size and electronegativity across periods and down groups. Key topics covered include the groups and periods in the modern table, how to read and understand its layout, and common questions about periodic trends and properties.
THE-PERIODICAL-TABLE-OF-ELEMENTS GROUP 2.pptx

The history of the periodic table's development, the arrangement of the elements, and the reactivity of each group of elements will all be covered in this lecture.
Más contenido relacionado
La actualidad más candente
Metals and Non Metals

Topics Included
• Introduction
• Metals
→ Physical properties of metals
→ Chemical Properties of metals
• Non-metals
→ Physical properties of non-metals
→ Chemical Properties of metals
• Difference between metals and non-metals
• Reaction with Acids
• Reaction with Bases
Periodic table ppt cscope

The periodic table arranges the elements based on atomic number and chemical properties. It is divided into metals, nonmetals, and metalloids. The periodic table predicts chemical behavior, trends, and element properties. It organizes elements by atomic structure, number, and physical/chemical traits.
Periodic Properties of Elements (Class X CBSE & ICSE)

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands attempted to classify elements but their systems had limitations. Mendeleev created the first successful periodic table in 1869 by arranging elements by atomic mass and leaving gaps for undiscovered elements. The modern periodic table is arranged by atomic number based on the repeating properties of elements, and classifies elements into blocks and groups with similar properties. The periodic table provides an organized framework for studying and predicting the properties of all known elements.
The modern periodic table

The document discusses the structure and organization of the modern periodic table. Elements are arranged horizontally in order of increasing atomic number and grouped vertically into columns based on their electron configurations. The periodic table is divided into blocks including s-, p-, d- and f-blocks. An element's position in the table is determined by its atomic number and electron configuration, especially the outermost electrons.
Presentation on periodic table

Atomic radii decrease across a period as nuclear charge increases. Cations are smaller than their parent atoms. Among isoelectronic species, the one with the larger positive nuclear charge will have the smallest radius. Ionization energy generally increases across a period as it is more difficult to remove electrons, and decreases down a group as shielding increases. Electronegativity follows similar trends as ionization energy.
The periodic table

The document discusses the organization and classification of elements in the periodic table. It describes how Mendeleev organized the elements based on increasing atomic mass and similar properties. The modern periodic table is organized by increasing atomic number. Elements are classified into metals, nonmetals, and metalloids, which are grouped based on their location on the periodic table. Each group of elements has a characteristic number of valence electrons that determine their chemical properties.
Periodic classification of elements Grade-11

The document discusses the periodic classification of elements. It provides the atomic numbers, symbols, and atomic masses of the first 30 elements in the periodic table. It then discusses the key features and components of modern periodic tables, including that elements are arranged by atomic number rather than atomic mass. The modern periodic table contains 7 periods and 18 groups, with elements in the same group having the same number of electrons in their outer shell. It also notes the advantages of the modern periodic table over previous classifications.
Atoms, Molecules, And Matter

An atom is the smallest particle of an element that contains protons, neutrons, and electrons. Elements are substances made of a single type of atom that combine to form molecules, which are made of two or more atoms joined together. Molecules such as water, written as H2O with two hydrogen atoms and one oxygen atom, are the smallest particles that combine without chemical change to form all matter in the world.
Atoms, elements, compounds and mixtures.pptx

1) The document discusses atoms, elements, compounds, and mixtures. It aims to explain what an atom is, differentiate between elements, compounds and mixtures, and give examples of each.
2) Atoms are the basic building blocks of all matter and are very small. Elements are substances made of only one type of atom that cannot be broken down further.
3) Compounds are formed when two or more elements are chemically bonded together and have different properties than the original elements. Mixtures contain two or more substances that are not chemically bonded and can be separated.
Tissue class 9

Meristematic tissue is a plant tissue that is undifferentiated and can divide to form other tissues. It is found in three main regions: the apical meristem at the tips of stems and roots, the intercalary meristem at the bases of leaves and stems, and the lateral meristem or cambium responsible for increasing the girth of stems and roots. Permanent tissues are differentiated tissues that do not divide, such as parenchyma, collenchyma and sclerenchyma. Epithelial tissue forms protective layers in animals, such as the skin and lining of organs, while connective tissue binds and supports other tissues.
Elements

Elements are pure substances that cannot be broken down further through chemical or physical means. There are currently 118 known elements, with new ones occasionally being discovered through artificial nuclear reactions. Elements are identified by their atomic number, which represents the number of protons in the nucleus. Elements are the basic building blocks of all matter and are used to create thousands of materials, though some like plutonium are also used in nuclear weapons due to their radioactive properties.
Carbon and its compounds

This document is a PowerPoint presentation on carbon and its compounds submitted by a student group. It provides information on carbon including that it is a nonmetallic element with an atomic number of 6 that forms covalent bonds. It discusses the origin of carbon in nature, the reasons for its versatility including catenation and tetracovalency. The presentation also covers different forms of carbon including graphite, diamond and fullerenes as well as saturated and unsaturated compounds. It provides examples of functional groups and homologous series and discusses chemical properties and uses of carbon compounds.
The periodic table class 10

The document discusses the development of the periodic table. It describes early classification systems by Dobereiner and Newlands that grouped elements based on properties but had limitations as new elements were discovered. Mendeleev organized the elements into the first recognizable periodic table based on atomic mass and predicted properties of undiscovered elements. Moseley later modified this to be based on atomic number. The modern periodic table is arranged into periods and groups with trends in properties like atomic radius, ionization potential, and electronegativity explained by variation in nuclear charge and number of electron shells across and down the table.
Periodic table

The periodic table organizes 118 known elements and displays their symbols and names. It arranges elements in rows called periods and columns called groups based on their atomic structure and properties. Elements in the same group have the same number of outer electrons and similar chemical properties, while properties vary greatly across each period from very reactive solids to inactive gases. The periodic table provides a way to classify and compare elements according to their atomic number and mass.
Periodic Law

The document summarizes the history and development of the periodic table. It describes how early chemists like Cannizzaro, Mendeleev, and Moseley contributed to organizing the elements based on their atomic masses and properties. The modern periodic table is arranged by increasing atomic number and groups elements with similar properties together in periods and families. Key periodic properties like atomic radius, ionization energy, and electronegativity are influenced by an element's position on the table.
Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

The document discusses the history and development of the periodic table. It explains that:
1) Elements have been organized into the periodic table based on their atomic weights and properties in order to systematically study and predict chemical behavior.
2) Early periodic tables were proposed by scientists like Dobereiner, Newlands, and Mendeleev based on trends in atomic weights, though Mendeleev's 1869 table was the most successful in predicting new elements.
3) Modern periodic tables are arranged by atomic number according to the periodic law proposed by Moseley, with elements grouped into blocks based on their electron configurations. The table better explains trends in physical and chemical properties.
Inside An Atom

The document discusses the structure of atoms and their composition. It explains that atoms are composed of a tiny, dense nucleus containing protons and neutrons, surrounded by electrons in defined orbits. Niels Bohr revised earlier atomic models by suggesting that electrons occupy specific orbits and can jump between these orbits by absorbing or emitting energy. The arrangement of electrons in an atom is called its electronic configuration. Atoms of the same element can have different numbers of neutrons, forming isotopes with the same chemical properties but different physical properties. Isotopes have applications including use in nuclear power and medical treatments.
Periodic Classification of Elements and Periodicity

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands began classifying elements based on their properties, but that Dmitri Mendeleev created the first recognizable periodic table in 1871. His table arranged elements in order of atomic mass and predicted properties of undiscovered elements. Later, Moseley's discovery of atomic number allowed the modern periodic table to arrange elements by atomic number instead of mass. The document also discusses trends in properties within the periodic table.
Class 10. Chapter 5. Periodic Classification of Elements

This document summarizes key concepts from Chapter 5 on the periodic classification of elements:
1. Early scientists like Dobereiner and Newlands attempted to classify elements based on their properties but their systems did not comprehensively explain the trends.
2. Mendeleev organized the known elements into the first periodic table based on their atomic masses and properties, noting periodic trends. This helped predict new elements.
3. Modern periodic tables are organized by atomic number not mass, reflecting Moseley's discovery of each element's unique atomic number. Periodic trends exist in properties like size, valence, and metallicity across periods and groups.
Chemical Bonding

This document discusses chemical bonding and macromolecular structures. It begins by explaining the different types of bonds - ionic bonds formed between metals and non-metals by electron transfer, and covalent bonds formed between non-metals by electron sharing. It describes the properties of ionic and covalent compounds. It then discusses macromolecular structures found in substances like diamond, graphite and metals. It explains metallic bonding and compares the structures and properties of diamond and graphite. In the end, it discusses the different uses of diamond and graphite based on their properties.
La actualidad más candente (20)
Periodic Properties of Elements (Class X CBSE & ICSE)

Periodic Properties of Elements (Class X CBSE & ICSE)
Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
Periodic Classification of Elements and Periodicity

Periodic Classification of Elements and Periodicity
Class 10. Chapter 5. Periodic Classification of Elements

Class 10. Chapter 5. Periodic Classification of Elements
Similar a Periodic classification of elements gr10, 2020-21
Periodic calssification of elements ncert shashikumar b s

The document provides an overview of the periodic classification of elements. It discusses early attempts at classification including Dobereiner's triads and Newlands' law of octaves. It then covers Mendeleev's periodic table, including its merits and defects. Finally, it describes the modern periodic table based on atomic number, including periodic trends in properties like atomic size and electronegativity across periods and down groups. Key topics covered include the groups and periods in the modern table, how to read and understand its layout, and common questions about periodic trends and properties.
THE-PERIODICAL-TABLE-OF-ELEMENTS GROUP 2.pptx

The history of the periodic table's development, the arrangement of the elements, and the reactivity of each group of elements will all be covered in this lecture.
Periodic Classification Of Elements

1. The document discusses the historical development of the periodic table from Dobereiner's classification of elements into triads to Moseley's ordering of all elements by their atomic number.
2. Mendeleev's periodic table was a significant improvement as it predicted future elements and organized elements into periods and groups according to chemical properties.
3. The modern periodic table is based on Moseley's work classifying elements by atomic number, with elements in the same group having the same number of valence electrons and similar chemical properties.
PERIODIC CLASSIFICATION OF ELEMENTS

This document discusses the periodic table and classification of elements. It explains that elements are arranged in the periodic table in order of increasing atomic number. The table is divided into blocks based on the subshell where electrons are located (s, p, d, f blocks). Elements are also classified into groups based on their physical and chemical properties. The modern periodic table improved upon earlier versions by being based on the fundamental property of atomic number, better correlating an element's position with its electronic configuration.
10-Periodic-Classification-of-Elements-converted.pdf

Mendeleev arranged elements in increasing order of atomic mass and placed those with similar properties in the same group, forming the first periodic table. This allowed for accurate prediction of undiscovered elements and correction of atomic masses. The modern periodic table is arranged by atomic number, with elements in periods by electron shell and groups by valence electrons. Trends across periods include decreasing atomic size and metallic character from left to right as nuclear charge increases, while trends down groups include increasing size and metallic character as shells are added.
3940538.ppt

This document discusses the development of the periodic table from early classification attempts to the modern periodic table. It describes Dobereiner's discovery of triads of elements with similar properties in the 1810s. It then discusses Newlands' law of octaves from the 1860s which noted patterns every eighth element. Mendeleev developed the first periodic table in 1869, arranging elements by atomic mass and leaving gaps for undiscovered elements. The modern periodic table is arranged by atomic number instead of mass and helped resolve limitations of earlier classifications. It describes trends in properties like valency and size across periods and groups in the modern periodic table.
Chapter 9 periodic classification of elements

This document provides information about the periodic classification of elements. It discusses early classification schemes proposed by Dobereiner and Newlands, and their limitations. It then describes Mendeleev's periodic table from 1869, which arranged elements in order of increasing atomic mass and predicted new elements. The document outlines properties of groups studied by Mendeleev and limitations of his table. Finally, it discusses the modern periodic law and properties of the modern periodic table, including trends in atomic size, ionization energy, and metallic character across periods and down groups.
CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

The document discusses the classification of elements and the development of the periodic table over time. It describes early attempts at classification by Dobereiner, Newlands, and Mendeleev. Mendeleev organized the elements into the first periodic table based on increasing atomic mass. Later, Moseley established the modern periodic table based on increasing atomic number. The document outlines the structural features and characteristics of the main blocks (s, p, d, f) of the periodic table. It also defines atomic properties like atomic radius, covalent radius, and metallic radius, and how they vary within the periodic table.
Periodic classification of elements

The periodic table organizes the elements according to increasing atomic number. Early scientists like Dobereiner, Newlands, and Mendeleev made attempts to classify elements, with Mendeleev devising the first successful periodic table. Mendeleev arranged elements with similar properties into columns and left gaps for elements not yet discovered. The periodic table provides information on elemental properties like valency, atomic size, metallic character, and electronegativity based on an element's position.
Periodic Classification Of Elements Class 10th

Johann Wolfgang Döbereiner studied chemistry and became a professor of chemistry and pharmacy. He discovered similar triads of elements with increasing atomic masses, which led to the development of the periodic table. John Newlands arranged elements in order of increasing atomic mass and found they repeated properties every eighth element, calling this the Law of Octaves. However, this law did not apply to all elements as more were discovered. Dmitri Mendeleev arranged elements based on atomic mass and chemical properties, leaving gaps for undiscovered elements, which contributed greatly to the early periodic table. The modern periodic table is based on atomic number according to Henry Moseley's discovery that it is a more fundamental property than atomic mass.
classification-of-elements-periodicity-in-properties-for-class-xi.ppt

This document discusses the classification and periodic trends of chemical elements. It begins by outlining early classification systems developed by Dobereiner, Newlands, Meyer and Mendeleev that organized elements by their increasing atomic masses and recognized periodic trends in their properties. Mendeleev organized the elements into the first periodic table. Later, Moseley determined that atomic number, not atomic mass, was fundamental in organizing elements. This led to the modern periodic table arranged by increasing atomic number. The document then discusses the organization and features of the long form periodic table, including grouping elements by their electronic configurations and periodic trends in physical and chemical properties.
Chapter 6 Periodic table

The document discusses the organization and structure of the periodic table. It describes how elements are arranged based on their atomic structure and properties. Elements are organized by increasing atomic number and grouped according to similar characteristics. The periodic table displays the symbols, names, atomic numbers and masses of each element.
Chapter 6 Periodic Table

The document discusses the organization and structure of the periodic table. It describes how elements are arranged based on their atomic structure and properties. Elements are organized by increasing atomic number and grouped according to similar characteristics. The periodic table displays the symbols, names, atomic numbers and masses of each element.
Periodic_classification_of_elements edited.pptx

The document discusses the history of the periodic classification of elements from early systems proposed by Lavoisier, Dalton, Dobereiner, and Newlands to Mendeleev's periodic table. It outlines some of the key developments and properties used in their classification schemes, including atomic weight and electronic configuration. The modern periodic table is based on atomic number according to Moseley's discovery that properties are periodic functions of atomic number rather than atomic weight. The document also discusses periodic trends in properties like atomic radius, metallic character, electronegativity, and their relationship to position in the periodic table.
chapter-6-Science 8-1223664031830946-8.pdf

For educational purposes only. No copyright intended to use the material. Credited the owner of the ppt. This is only for reference for the topic of Grade 8 Science in TNHS 2023-2024
Chapter 6-1223664031830946-8

The document summarizes key aspects of the periodic table, including its structure, historical development, and trends in elemental properties. It explains that the periodic table arranges elements in rows and columns based on atomic number and properties. Mendeleev and Moseley helped develop the modern periodic table by arranging elements based on atomic mass and charge. Elements are classified as metals, nonmetals, and metalloids based on their physical and chemical properties, which vary predictably within and between groups and periods.
Classification of elements periodicity properties IUPAC names 100 to 118

This document discusses the classification of elements and periodic trends in their physical and chemical properties. It begins with an overview of how elements have historically been classified from Dobereiner's triads to Mendeleev's periodic table. Key periodic properties discussed include atomic radius, ionization energy, and electronegativity. Periodic trends in these properties are explained in terms of the periodic variation in the effective nuclear charge experienced by outer shell electrons due to shielding effects. Chemical properties also vary periodically, including oxidation states and reactivity. The document aims to explain these periodic trends using concepts of electronic configuration.
Grade 8 SCIENCE

The document provides information about the periodic table of elements and related concepts:
1) The periodic table arranges the chemical elements by atomic number and electron configuration, showing periodic trends in properties. Elements are grouped into blocks by their electron configurations and periods by the filling of electron shells.
2) Key concepts discussed include atomic number, electron configuration, chemical properties, periodic trends, metals, nonmetals, groups such as halogens and noble gases, periods, and shells and subshells.
3) Dmitri Mendeleev is credited with publishing the first recognizable periodic table in 1869, organizing known elements and predicting properties of undiscovered elements. His work established the basis for the modern periodic table
THE PERIODIC TABLE

Welcome to the project dedicated to the remarkable Periodic Table of Elements! Prepare to embark on an exciting exploration of the building blocks of our universe through our comprehensive presentation.
Our presentation offers a captivating visual representation of the Periodic Table, showcasing the elements and their essential properties. Each element is presented with its atomic number, symbol, and atomic mass, providing a clear overview of the entire table.
Delve deeper into the presentation and unlock a wealth of knowledge about each element. Discover their unique characteristics, such as their physical and chemical properties, common uses, and even their historical significance. Engaging narratives and captivating illustrations bring the elements to life, making the learning experience both enjoyable and informative.
Whether you're a student, a science enthusiast, or simply curious about the elements that shape our world, our presentation on the Periodic Table will enlighten and inspire. Join us on this educational journey as we unravel the mysteries and marvels of the elements that surround us.
Periodic Table (Classification of elements) MANIK

Electronic structure of atoms, modern periodic table and periodic law, variation of periodic properties within periods and groups, ionization potential, electron affinity, electronegativity, usefulness and limitation of periodic table.
Similar a Periodic classification of elements gr10, 2020-21 (20)
Periodic calssification of elements ncert shashikumar b s

Periodic calssification of elements ncert shashikumar b s
10-Periodic-Classification-of-Elements-converted.pdf

10-Periodic-Classification-of-Elements-converted.pdf
CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
classification-of-elements-periodicity-in-properties-for-class-xi.ppt

classification-of-elements-periodicity-in-properties-for-class-xi.ppt
Classification of elements periodicity properties IUPAC names 100 to 118

Classification of elements periodicity properties IUPAC names 100 to 118
Más de MhdAfz
Surface Chemistry - Class 12

For more such informative content, go to https://scifitechify.blogspot.com/. Surface chemistry presentation will provide lots of valuable information about its day-to-day applications in real life. It explains concepts of adsorption, absorption, activation energy, Arrhenius equation, colloids, solutions, chemical processes etc.
It also elaborates on specific chemical reactions like peptisation reaction.
Skeletal System and the Nervous System CBSE

For more such informative content, go to https://scifitechify.blogspot.com/.
This presentation is about a biology topic. It talks about the skeletal and nervous system. It tasks about their structures and different parts involved in the structure. It is a really informative presentation.
Coal and Petroleum Grade 8 CBSE 

For more such informative content, go to https://scifitechify.blogspot.com/.
This presentation has all topics of coal and petroleum covered. It talks about their real-world uses and application in electricity generation, automobile, fuel etc. It also talks about the disadvantages of using fossil fuels. It promotes sustainable and renewable energy sources over conventional fossil fuels. It also talks about hydrogen being the cleanest fuel and mixing with petrol.
Upcoming science topics

For more such informative content, go to https://scifitechify.blogspot.com/.
This presentation explains the characteristic features of Falcon 9 and what sets it apart from other conventional rockets. It explains how it would eventually help humans become a space-faring civilization. it is about recent advancements in space technology and space research and breakthroughs.
Falcon 9 - The Revolution of Space Industry

For more such informative content, go to https://scifitechify.blogspot.com/.
This presentation explains the characteristic features of Falcon 9 and what sets it apart from other conventional rockets. It explains how it would eventually help humans become a space-faring civilisation.
Thermodynamics class 11 physics

For more such informative content, go to https://scifitechify.blogspot.com/.
This PPT is about thermodynamics. It explains isothermal, isobaric, adiabatic, and isochoric processes. In-depth explanation of formulae and derivation is provided.
CHAP 3 ATOMS AND MOLECULES GR 9 2020-21

For more such informative content, go to https://scifitechify.blogspot.com/. This video will introduce you to the world of ATOMS & MOLECULES. HOPE YOU ENJOY IT. NEXT POST ON: WHY DO WE WEIGH LESS ON THE MOON ?
GRADE 7 CBSE CHAPTER 10 RESPIRATION IN ORGANISMS

For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell you about the respiration in different kinds of organisms and the process and mechanism behind it. HOPE YOU ENJOY IT. NEXT POST ON: WHY DO WE WEIGH LESS ON THE MOON ? DON'T FORGET TO CHECK OUT : THE CORONAVIRUS SERIES
L.05 carbon and its compounds gr 10, 2019-20

1. Carbon exists in Earth's crust and atmosphere and forms millions of compounds due to its ability to catenate and be tetravalent. Organic chemistry studies carbon compounds except oxides, carbonates, and bicarbonates.
2. Berzelius proposed that organic compounds could only be synthesized in living organisms, but Wohler synthesized urea in the laboratory in 1828, disproving this vital force theory.
3. Carbon forms covalent bonds by sharing electrons in stable octet configurations, allowing it to form diverse chains, rings, and functional groups that exhibit properties ranging from nonpolar to ionic.
TOP 10 FUN FACTS OF VENUS

For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell you some of the UNKNOWN facts of venus. HOPE YOU ENJOY IT. NEXT POST ON: WHY DO WE WEIGH LESS ON THE MOON
WHAT IS A MIRAGE ?

For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell you about the explanation behind the formation of mirage. CATCH UP ON: IS MARS REALLY RED ?
TOP 20 FUN FACTS OF STARS
TOP 10 FUN FACTS OF GOLD
NEXT POST ON: WHY DO WE WEIGH LESS ON THE MOON ?
THE 8 PLANETS OF THE SOLAR SYSTEM

The document discusses the solar system but provides no details. It mentions what a mirage is and that the next post will be on that topic, but gives no other information. The summary is only 3 sentences because the original document contains no substantive content to summarize.
THE CORONAVIRUS STORY: COVID - 19 PANDEMIC OUTBREAK SERIES I Episode 2 I Str...

For more such informative content, go to https://scifitechify.blogspot.com/. THE CORONAVIRUS STORY: COVID - 19 PANDEMIC OUTBREAK SERIES I Episode 2 I Structure of the Virus. NEXT POST ON: THE CORONAVIRUS STORY:
COVID - 19 PANDEMIC OUTBREAK SERIES I Episode 3 I Life Cycle of the Virus
TOP 10 FUN FACTS OF GOLD

For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell you some of the UNKNOWN facts about precious metal gold. HOPE YOU ENJOY IT. NEXT POST ON: WHAT IS A MIRAGE ?
Top 20 fun facts of stars

There are at least 10 billion trillion stars in the universe. Stars are formed from dense knots of dust and gas called nebulae, with some nebulae containing enough matter to produce over 10,000 stars. When looking at the night sky with the naked eye, every visible star is part of the Milky Way Galaxy, which contains stars that are on average around 13.6 billion years old.
IS MARS REALLY RED ?

For more such informative content, go to https://scifitechify.blogspot.com/. This video will give you a brief introduction to the REAL COLOUR of mars and why does it appear to be red. HOPE YOU ENJOY IT. NEXT POST ON: WHAT IS A MIRAGE ?
TOP 10 FUN FACTS OF MARS 

For more such informative content, go to https://scifitechify.blogspot.com/. This video will tell some of the UNKNOWN facts of Mars. ENJOY IT. NEXT POST ON: IS MARS REALLY RED ?
WHY ARE AURORAS FORMED ? 

For more such informative content, go to https://scifitechify.blogspot.com/. This video will give you the brief expanation as to why these colourful and wonderful auroras are formed in northern places. NEXT POST ON: WHAT IS A MIRAGE ?
WHY COMETS HAVE TAILS ?

For more such informative content, go to https://scifitechify.blogspot.com/. This video will give you brief information as to why comets have tials and how is it formed. NEXT POST ON: WHY ARE AURORAS FORMED ?
TOP 10 FUN FACTS OF SOLAR SYSTEM

The document discusses 10 fun facts about the solar system but provides no details. It mentions comets having tails and a next post topic but gives no other information from the document.
Más de MhdAfz (20)
THE CORONAVIRUS STORY: COVID - 19 PANDEMIC OUTBREAK SERIES I Episode 2 I Str...

THE CORONAVIRUS STORY: COVID - 19 PANDEMIC OUTBREAK SERIES I Episode 2 I Str...
Último
Phenomics assisted breeding in crop improvement

As the population is increasing and will reach about 9 billion upto 2050. Also due to climate change, it is difficult to meet the food requirement of such a large population. Facing the challenges presented by resource shortages, climate
change, and increasing global population, crop yield and quality need to be improved in a sustainable way over the coming decades. Genetic improvement by breeding is the best way to increase crop productivity. With the rapid progression of functional
genomics, an increasing number of crop genomes have been sequenced and dozens of genes influencing key agronomic traits have been identified. However, current genome sequence information has not been adequately exploited for understanding
the complex characteristics of multiple gene, owing to a lack of crop phenotypic data. Efficient, automatic, and accurate technologies and platforms that can capture phenotypic data that can
be linked to genomics information for crop improvement at all growth stages have become as important as genotyping. Thus,
high-throughput phenotyping has become the major bottleneck restricting crop breeding. Plant phenomics has been defined as the high-throughput, accurate acquisition and analysis of multi-dimensional phenotypes
during crop growing stages at the organism level, including the cell, tissue, organ, individual plant, plot, and field levels. With the rapid development of novel sensors, imaging technology,
and analysis methods, numerous infrastructure platforms have been developed for phenotyping.
Compexometric titration/Chelatorphy titration/chelating titration

Classification
Metal ion ion indicators
Masking and demasking reagents
Estimation of Magnisium sulphate
Calcium gluconate
Complexometric Titration/ chelatometry titration/chelating titration, introduction, Types-
1.Direct Titration
2.Back Titration
3.Replacement Titration
4.Indirect Titration
Masking agent, Demasking agents
formation of complex
comparition between masking and demasking agents,
Indicators/Metal ion indicators/ Metallochromic indicators/pM indicators,
Visual Technique,PM indicators (metallochromic), Indicators of pH, Redox Indicators
Instrumental Techniques-Photometry
Potentiometry
Miscellaneous methods.
Complex titration with EDTA.
The binding of cosmological structures by massless topological defects

Assuming spherical symmetry and weak field, it is shown that if one solves the Poisson equation or the Einstein field
equations sourced by a topological defect, i.e. a singularity of a very specific form, the result is a localized gravitational
field capable of driving flat rotation (i.e. Keplerian circular orbits at a constant speed for all radii) of test masses on a thin
spherical shell without any underlying mass. Moreover, a large-scale structure which exploits this solution by assembling
concentrically a number of such topological defects can establish a flat stellar or galactic rotation curve, and can also deflect
light in the same manner as an equipotential (isothermal) sphere. Thus, the need for dark matter or modified gravity theory is
mitigated, at least in part.
Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...

The pygmy halfbeak Dermogenys colletei, is known for its viviparous nature, this presents an intriguing case of relatively low fecundity, raising questions about potential compensatory reproductive strategies employed by this species. Our study delves into the examination of fecundity and the Gonadosomatic Index (GSI) in the Pygmy Halfbeak, D. colletei (Meisner, 2001), an intriguing viviparous fish indigenous to Sarawak, Borneo. We hypothesize that the Pygmy halfbeak, D. colletei, may exhibit unique reproductive adaptations to offset its low fecundity, thus enhancing its survival and fitness. To address this, we conducted a comprehensive study utilizing 28 mature female specimens of D. colletei, carefully measuring fecundity and GSI to shed light on the reproductive adaptations of this species. Our findings reveal that D. colletei indeed exhibits low fecundity, with a mean of 16.76 ± 2.01, and a mean GSI of 12.83 ± 1.27, providing crucial insights into the reproductive mechanisms at play in this species. These results underscore the existence of unique reproductive strategies in D. colletei, enabling its adaptation and persistence in Borneo's diverse aquatic ecosystems, and call for further ecological research to elucidate these mechanisms. This study lends to a better understanding of viviparous fish in Borneo and contributes to the broader field of aquatic ecology, enhancing our knowledge of species adaptations to unique ecological challenges.
Applied Science: Thermodynamics, Laws & Methodology.pdf

When I was asked to give a companion lecture in support of ‘The Philosophy of Science’ (https://shorturl.at/4pUXz) I decided not to walk through the detail of the many methodologies in order of use. Instead, I chose to employ a long standing, and ongoing, scientific development as an exemplar. And so, I chose the ever evolving story of Thermodynamics as a scientific investigation at its best.
Conducted over a period of >200 years, Thermodynamics R&D, and application, benefitted from the highest levels of professionalism, collaboration, and technical thoroughness. New layers of application, methodology, and practice were made possible by the progressive advance of technology. In turn, this has seen measurement and modelling accuracy continually improved at a micro and macro level.
Perhaps most importantly, Thermodynamics rapidly became a primary tool in the advance of applied science/engineering/technology, spanning micro-tech, to aerospace and cosmology. I can think of no better a story to illustrate the breadth of scientific methodologies and applications at their best.
ESR spectroscopy in liquid food and beverages.pptx

With increasing population, people need to rely on packaged food stuffs. Packaging of food materials requires the preservation of food. There are various methods for the treatment of food to preserve them and irradiation treatment of food is one of them. It is the most common and the most harmless method for the food preservation as it does not alter the necessary micronutrients of food materials. Although irradiated food doesn’t cause any harm to the human health but still the quality assessment of food is required to provide consumers with necessary information about the food. ESR spectroscopy is the most sophisticated way to investigate the quality of the food and the free radicals induced during the processing of the food. ESR spin trapping technique is useful for the detection of highly unstable radicals in the food. The antioxidant capability of liquid food and beverages in mainly performed by spin trapping technique.
Randomised Optimisation Algorithms in DAPHNE

Slides from talk:
Aleš Zamuda: Randomised Optimisation Algorithms in DAPHNE .
Austrian-Slovenian HPC Meeting 2024 – ASHPC24, Seeblickhotel Grundlsee in Austria, 10–13 June 2024
https://ashpc.eu/
Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water

Sharlene Leurig - Enabling Onsite Water Use with Net Zero WaterTexas Alliance of Groundwater Districts
Presented at June 6-7 Texas Alliance of Groundwater Districts Business MeetingESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...Advanced-Concepts-Team
Presentation in the Science Coffee of the Advanced Concepts Team of the European Space Agency on the 07.06.2024.
Speaker: Diego Blas (IFAE/ICREA)
Title: Gravitational wave detection with orbital motion of Moon and artificial
Abstract:
In this talk I will describe some recent ideas to find gravitational waves from supermassive black holes or of primordial origin by studying their secular effect on the orbital motion of the Moon or satellites that are laser ranged.Thornton ESPP slides UK WW Network 4_6_24.pdf

ESPP presentation to EU Waste Water Network, 4th June 2024 “EU policies driving nutrient removal and recycling
and the revised UWWTD (Urban Waste Water Treatment Directive)”
8.Isolation of pure cultures and preservation of cultures.pdf

Isolation of pure culture, its various method.
Micronuclei test.M.sc.zoology.fisheries.

Current Ms word generated power point presentation covers major details about the micronuclei test. It's significance and assays to conduct it. It is used to detect the micronuclei formation inside the cells of nearly every multicellular organism. It's formation takes place during chromosomal sepration at metaphase.
The debris of the ‘last major merger’ is dynamically young

The Milky Way’s (MW) inner stellar halo contains an [Fe/H]-rich component with highly eccentric orbits, often referred to as the
‘last major merger.’ Hypotheses for the origin of this component include Gaia-Sausage/Enceladus (GSE), where the progenitor
collided with the MW proto-disc 8–11 Gyr ago, and the Virgo Radial Merger (VRM), where the progenitor collided with the
MW disc within the last 3 Gyr. These two scenarios make different predictions about observable structure in local phase space,
because the morphology of debris depends on how long it has had to phase mix. The recently identified phase-space folds in Gaia
DR3 have positive caustic velocities, making them fundamentally different than the phase-mixed chevrons found in simulations
at late times. Roughly 20 per cent of the stars in the prograde local stellar halo are associated with the observed caustics. Based
on a simple phase-mixing model, the observed number of caustics are consistent with a merger that occurred 1–2 Gyr ago.
We also compare the observed phase-space distribution to FIRE-2 Latte simulations of GSE-like mergers, using a quantitative
measurement of phase mixing (2D causticality). The observed local phase-space distribution best matches the simulated data
1–2 Gyr after collision, and certainly not later than 3 Gyr. This is further evidence that the progenitor of the ‘last major merger’
did not collide with the MW proto-disc at early times, as is thought for the GSE, but instead collided with the MW disc within
the last few Gyr, consistent with the body of work surrounding the VRM.
Equivariant neural networks and representation theory

Or: Beyond linear.
Abstract: Equivariant neural networks are neural networks that incorporate symmetries. The nonlinear activation functions in these networks result in interesting nonlinear equivariant maps between simple representations, and motivate the key player of this talk: piecewise linear representation theory.
Disclaimer: No one is perfect, so please mind that there might be mistakes and typos.
dtubbenhauer@gmail.com
Corrected slides: dtubbenhauer.com/talks.html
Último (20)
Compexometric titration/Chelatorphy titration/chelating titration

Compexometric titration/Chelatorphy titration/chelating titration
The binding of cosmological structures by massless topological defects

The binding of cosmological structures by massless topological defects
Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...

Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...
mô tả các thí nghiệm về đánh giá tác động dòng khí hóa sau đốt

mô tả các thí nghiệm về đánh giá tác động dòng khí hóa sau đốt
Applied Science: Thermodynamics, Laws & Methodology.pdf

Applied Science: Thermodynamics, Laws & Methodology.pdf
ESR spectroscopy in liquid food and beverages.pptx

ESR spectroscopy in liquid food and beverages.pptx
Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water

Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water
ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...
8.Isolation of pure cultures and preservation of cultures.pdf

8.Isolation of pure cultures and preservation of cultures.pdf
The debris of the ‘last major merger’ is dynamically young

The debris of the ‘last major merger’ is dynamically young
Equivariant neural networks and representation theory

Equivariant neural networks and representation theory
Periodic classification of elements gr10, 2020-21
- 1. Periodic classification of elements
- 2. Classification of elements ● The earlier attempt to classify elements involve dividing them into metals and nonmetals. ● The classification was done by Antoine Laurent Lavoisier (Father of chemistry). ● This method failed because most of the elements were metals and a very few were non metals. ● Some elements resembled metals and nonmetals and hence they could not be classified.
- 3. Dobereiner's triads ● In the year 1817, Johann Wolfgang Dobereiner, a German chemist classified elements having similar properties into groups called triads. ● When the elements were arranged in groups of 3, in the increasing order of atomic masses, the middle element has the atomic mass and properties roughly the average of the other two elements.
- 4. Alkali metal group Alkaline earth metal group Element At: mass Element At: mass Li 7 Ca 40 Na 23 Sr 88 K 39 Ba 137 Halogen group Element At: mass Cl 35.5 Br 80 I 127
- 5. Newlands law of Octaves - John Newland When elements are arranged in the increasing order of their atomic masses (from H to Th), the properties of the 8th element resembled with the properties of the first element. This was compared to the octaves found in music. Hence called Newlands law of octaves.
- 6. Demerits of Newland’s Law of Octaves ● It was found that the Law of Octaves was applicable only upto calcium, as after calcium every eighth element did not possess properties similar to that of the first. ● It was assumed by Newlands that only 56 elements existed in nature and no more elements would be discovered in the future. But, later on, several new elements were discovered, whose properties did not fit into the Law of Octaves. ● In order to fit elements into his Table, Newlands adjusted two elements in the same slot, but also put some unlike elements under the same note. Cobalt and nickel are in the same slot and these are placed in the same column as fluorine, chlorine and bromine which have very different properties than these elements. Iron, which resembles cobalt and nickel in properties, has been placed far away from these elements. Thus, Newlands’ Law of Octaves worked well with lighter elements only.
- 7. Mendeleev’s Periodic Law - Dmitri Ivanovich Mendeleev When Mendeleev started his work , 63 elements were known. He examined the relationship between the atomic masses of the elements and their physical and chemical properties. “ The properties of elements are a periodic function of their atomic masses”
- 9. Features of Mendeleev’s Periodic table. ● The vertical columns were called groups and horizontal rows were called periods. There were 6 periods and 8 groups in the table. ● The elements in each group resembles with each other in many properties. ● All the elements in the group show the same valency. ● The properties like atomic size, MP and BP changed gradually in any period while going from left to right.
- 10. Advantages of Mendeleev’s Periodic table. ● Mendeleev could classify all the 63 elements discovered at that time on the basis of similarity in properties. ● He left gaps for elements yet to be discovered. He named them by prefixing the sanskrit numeral, Eka (One) to the name of the preceding element in the same group. Eg: Scandium, gallium and germanium have properties similar to Eka-Boron, Eka-Aluminium and Eka-Silicon respectively. ● He predicted the properties of the undiscovered elements and helped in the discovery of these elements. ● His periodic table could accommodate noble gases when they were discovered. ● He could correct the masses of certain elements with respect to their position in the table.
- 11. Limitations of Mendeleev’s Periodic table ● A correct position for hydrogen could not be given in the periodic table. Hydrogen resembles alkali metals and halogens. ● The position of isotopes could not be explained. ● Wrong order of atomic mass of some elements could not be explained. Eg: Cobalt (Atomic mass 58.9) appeared before nickel (Atomic mass 58.7). ● Atomic mass do not increase in a regular manner. So it was not possible to predict how many elements could be discovered between two elements. ● He does not make any attempt to separate metals from nonmetals.
- 12. Position of Hydrogen ● Hydrogen resembles alkali metals - They combine with halogens, oxygen and sulphur to form compounds having similar formula. ● Like halogens, hydrogen is diatomic and it combines with metals and nonmetals to form covalent compounds.
- 13. Modern Periodic Law Henry Moseley showed that atomic number is a more fundamental property than atomic mass. “ Properties of elements are periodic function of their atomic number”
- 14. ● Atomic number (Z) is the number of protons present in an atom. ● Z increases by one in going from one element to the next. ● Prediction of properties of elements was more precise with the arrangement of elements in the increasing order of atomic number.
- 20. New names added to the periodic table. ● 113 - Nh -Nihonium - Japan ● 115 - Mc - Moscovium - Moscow region ● 117 - Ts - Tennessine - Tennessee ● 118 - Og - Oganesson - Yuri Oganessian
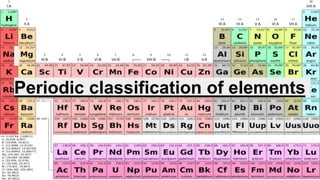
- 22. General features of long form periodic table ● The elements are arranged in the increasing order of atomic number. ● There are 7 horizontal rows called periods. ● There are 18 vertical columns called groups. ● Elements having similar valence electrons are placed in the same group. ● Each period starts with the filling of a new shell.
- 23. ● The 14 elements after Lanthanum are called lanthanoids. ● The 14 elements after Actinium are called actinoids ● To limit the size of the periodic table and to keep the elements with similar properties together, Lanthanoids and Actinoids are placed at the bottom of the periodic table. ● The elements of group1, 2 and 13-17 (s-block and p- block) are called representative elements or main group elements. They have completely filled inner shells but incomplete valence shell. ● The elements of group 3-12(d-block) are called transition elements. They have incomplete outer shells
- 24. ● The elements placed at the bottom of the periodic table (f- block) are called inner transition elements. They are also called rare earth metals because they occur rarely in the earth's crust. ● Group 18 elements are called noble gases or inert gases. Their outer orbits are completely filled and are unreactive. ● A zig-zag line separates metals from nonmetals by some elements called metalloids or semimetals (B, Si, Ge, As, Sb, Te and Po) ● Metals are found on the left side of the periodic table and nonmetals on the right side.
- 25. Important points to note ● Elements in any group have same number of valence electrons. ● Size of the atom decreases across a period and increases down a group. ● Each period starts with a new shell. ● Maximum number of electrons in each shell is given by the formula 2n2, where n is the number of the given shell. (K=1, L=2, M=3, ….)
- 27. Characteristics / trends of Modern periodic table 1. Valency - It is the combining capacity of an element. It denotes the number of electrons lost or gained during a chemical reaction. ● On moving from left to right in a period, valency of the elements first increases from 1 to 4 and then decreases to zero. ● All the elements in a group has same valency, as they have same number of valence shell electrons.
- 28. 2. Atomic size: ● On moving from left to right in a period, the atomic size (atomic radius) of the atom decreases. ● As we move from left to right in a period, the atomic number increases, which means the number of protons also increases. Due to the large positive charge on the nucleus, the electrons are pulled in more closer to the nucleus and size of the atom decreases. ● Down a group size of atom increases, as new shells are added up.
- 29. 3. Metallic character: ● Metals are found on the left hand side of the table and nonmetals on the right hand side. ● Metals has a tendency to lose electrons - electropositive. ● Nonmetals has a tendency to gain electrons - electronegative. ● On moving from left to right in a period, the electropositive character decreases and electro negative character increases. ● On moving from left to right in a period, the tendency of atoms to lose electrons decreases and tendency of atoms to gain electron increases.
- 30. ● On moving down a group the metallic character of the elements increases or the electropositive character of the element increases. ● As we go down in group 17, the electronegative character (nonmetallic character) of elements decreases. ● Fluorine is the most electronegative element.
- 31. 4. Chemical reactivity. ● Chemical reactivity of metals increases on going down a group. ● Chemical reactivity of nonmetals decreases on going down a group ● On moving from left to right in a period, the chemical reactivity of elements first decreases and then increases.
- 32. 5. Nature of oxides. ● On moving from left to right in a period the basic nature of oxides decreases and the acidic nature of oxides increases. ● On moving down a group there is no change in the nature of oxides of elements ● All elements of group 1 form basic oxides ● All the elements of group 17 form acidic oxides
- 33. 6. Ionisation Energy ● The energy required to remove the most loosely bound electron from a gaseous neutral atom in the ground state is known as Ionisation energy. ● It is expressed in Kj/mol. ● Ionisation energy increases with the increase in atomic number across a period, due to the increase in nuclear charge and decrease in atomic size. ● Ionisation energy decreases down a group due to the increase in atomic size and increase in the shielding effect.
- 35. Reactivity of metals and nonmetals