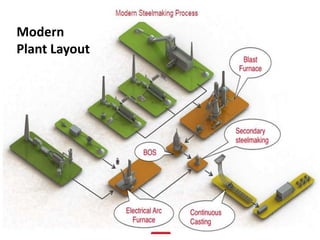
Modern Steelmaking Layouts and Processes
- 2. 27/09/2010 Materials Engineering NEDUET Karachi 2 Basic Oxygen Steelmaking Converter Plant Layout
- 3. 27/09/2010 Materials Engineering NEDUET Karachi 3 BOS process Basic Oxygen Steelmaking Converter • The Basic Oxygen process is the major method of making steel. • Modern furnaces, or converters, will take a charge of up to 350 tonnes at a time and convert it into steel in little more than 15 minutes • The molten iron accounts for about 80% of the total charge, the rest consisting of steel scrap.
- 4. 27/09/2010 Materials Engineering NEDUET Karachi 4
- 5. 27/09/2010 Materials Engineering NEDUET Karachi 5 A water-cooled oxygen lance is lowered into the converter, and high-purity oxygen is blown on to the metal at very high pressure. The oxygen forms a chemical bond with the unwanted elements and removes them as a slag which floats on top of the liquid steel. These oxidation reactions – the chemical boil – produce heat, and the temperature of the metal is controlled by the quality of the scrap and iron ore coolant added.
- 6. Functions of the Basic Oxygen • The main functions of the Basic Oxygen Furnace (BOF) are to decarburize and remove phosphorus from the hot metal, and to optimize the steel temperature so that any further treatments prior to casting can be performed with minimal reheating or cooling of the steel 27/09/2010 Materials Engineering NEDUET Karachi 6
- 7. Critical Operations of BOS • According to the changes in metal characteristics during refining, which of these operations are the most critical? (choose three answers) C removal Mn removal Si removal P removal S removal Temperature adjustment 27/09/2010 Materials Engineering NEDUET Karachi 7
- 8. Critical Operations of BOS • According to the changes in metal characteristics during refining, which of these operations are the most critical? (choose three answers) C removal Mn removal Si removal P removal S removal Temperature adjustment 27/09/2010 Materials Engineering NEDUET Karachi 8 • Yes. A large amount of carbon has to be extracted ( about 45 kg per ton of steel) and a narrow bracket of final is required, e.g. 0.05/- 0.01 %. • No. In general Mn oxidation proceeds without problem. There is no strict constraint on final Mn in the BOF. Mn alloying is performed in the ladle. • No. Si oxidation is very easy and takes place during the first 1/3 of the blow. Si alloying is performed in the ladle. • Yes. P removal can in practice be made only in the converter (or during hot metal pretreatment in Japan). All P entering the steel ladle (in particular from slag carryover) will remain in the steel. • No. Hot metal desulfurisation is a common practice, and ladle steel desulfurisation is performed for ultra low S contents, e.g. S less than 0.0010 %. • Yes. Final temperature has to be reached within a narrow bracket (e.g. / -10% 0 C) to allow a harmonious ladle metallurgy process. All temperature adjustments in the ladle (through chemical reaction- aluminothermy- or use of a ladle furnace when available) are very costly in time and for metal quality.
- 9. 27/09/2010 Materials Engineering NEDUET Karachi 9 The raw materials charged into the converter are: • Liquid 'hot metal' from the blast furnace after specific pre-treatments, i.e. desulfurization or dephosphorization • Other iron-containing additions, essentially scrap and ore, calculated so as to adjust the thermal balance and obtain the required steel temperature • The additions necessary to form a slag of appropriate composition, involving mainly lime (CaO) and dolomitic lime (CaO-MgO), usually in the form of 20 to 40 mm lumps • Pure oxygen injected either through a multi-hole lance or through bottom tuyeres After a completed blowing operation the materials produced are: • Liquid steel • Exhaust gas rich in CO (about 80-90%), recovered through the closed or suppressed combustion hood, and is often used in the burners of the reheating furnaces • Slag, poured out of the vessel after the steel • Both gas and slag are valuable by-products, provided they are properly recovered and stored. Inputs, Additions and Outputs
- 10. Chemical Reactions • The hot metal charge is refined by rapid oxidation reactions on contact with the injected oxygen, under conditions far removed from thermodynamic equilibrium with the other elements present: • C + ½ O2 → CO • CO is partially oxidized into CO2 above the melt (post-combustion). These gaseous reaction products are evacuated through the exhaust hood. The ratio CO2/(CO+CO2) is called post combustion ratio (PCR). • Other oxidation reactions occurring during refining include: • Si + O2 → SiO2 • 2P + (5/2)O2 → P2O5 • Mn + ½O2 → MnO • Fe + ½O2 → FeO • 2Fe + (3/2)O2 → Fe2O3 • These oxides combine with the other oxides charged (lime, dolomitic lime) to form a liquid slag which floats on the surface of the metal bath 27/09/2010 Materials Engineering NEDUET Karachi 10
- 11. Slag/Metal Reactions Slag/metal reactions concern: Si removal • This reaction is very fast and the total amount of silicon is transferred to the slag in the first one third of the blow P and S removal • These reactions require a very accurate control of slag formation and of the final slag composition in order to guarantee the low contents sought for the liquid steel. • Of the two, dephosphorization is the most crucial and is analyzed here. To be successful and cost efficient, the steelmaker has to consider both thermodynamic and kinetic aspects. 27/09/2010 Materials Engineering NEDUET Karachi 11
- 12. Making steel: EAF process 27/09/2010 Materials Engineering NEDUET Karachi 12
- 13. 27/09/2010 Materials Engineering NEDUET Karachi 13
- 14. 27/09/2010 Materials Engineering NEDUET Karachi 14 In the Electric Arc Furnace (EAF), recycled steel scrap is melted and converted into high quality steel by using high-power electric arcs. The main task of most modern EAFs is to convert the solid raw materials to liquid crude steel as fast as possible and then refine further in subsequent secondary steelmaking processes. Nevertheless, if time is available, almost any metallurgical operation may be performed during flat bath operation period (after melting), which is usually performed as a pre-treatment to the secondary steelmaking operations. This module introduces the equipment, raw materials and processes used to produce steel in the EAF, finishing with a simulation, which allows you to put all this in practice and melt your own cast
- 15. 27/09/2010 Materials Engineering NEDUET Karachi 15
- 16. 27/09/2010 Materials Engineering NEDUET Karachi 16
- 17. Electrode Breakage • Electrode breakage occurs occasionally in the EAF, predominantly during the melt-down operation. This should be avoided due to the high costs associated with a breakage Electrode breakage is usually a consequence of mechanical overload on the electrode from the surrounding scrap: • Scrap is caving in from the side as the electrode penetrates into the scrap pile inside the furnace. If heavy pieces hit directly on the electrode side breakage may occur. • The electrodes are moving downward without detecting that a non-conducting material is present at the tip of the electrode. As the electrode continues to push downwards, breakage may occur. • Animation: • http://www.steeluniversity.org/content/html/eng/default.asp?catid=25&pageid=2081271938 27/09/2010 Materials Engineering NEDUET Karachi 17
- 18. 27/09/2010 Materials Engineering NEDUET Karachi 18
- 19. Slag Foaming • Foaming slag is used to increase the thermal efficiency of the furnace during the refining period, when the side walls are fully exposed to the arc radiation. A foaming slag will rise and cover the electric arcs, thus permitting the use of a high tap setting without increasing the thermal load on the furnace walls. In addition, an electrical arc covered by a foaming slag will have a higher efficiency in transferring the energy into the steel phase. • Slag foaming is obtained by injecting oxygen into the liquid steel, where mainly iron is oxidized according to the reaction: • O2 + 2 Fe = 2 (FeO) • Carbon powder is then injected simultaneously into the slag phase where iron oxide is reduced. • (FeO) + C = Fe + CO (g) • The resulting CO gas is a critical component in order to obtain a foaming slag. 27/09/2010 Materials Engineering NEDUET Karachi 19
- 20. 27/09/2010 Materials Engineering NEDUET Karachi 20
- 21. 27/09/2010 Materials Engineering NEDUET Karachi 21
- 22. Slag Foamability • Slag foamability is as critical a parameter as CO gas generation in order to obtain foam. Foamability is controlled by the slag phase physical properties viscosity, surface tension and density. These properties vary with slag composition. • Due to the nature of the EAF – with large variations in temperature and composition in different locations of the furnace – the slag is likely to be partly solid at some positions during some stages of the operation. A slag that is not fully liquid, but contains some solid material such as undissolved lime, also influences the foamability, since there is a change in the apparent viscosity: • where: • η = apparent viscosity of the solid-containing melt • η0 = viscosity of the solid-free melt • f = volume fraction of solid particles in the melt • a and n are constants. 27/09/2010 Materials Engineering NEDUET Karachi 22
- 23. 27/09/2010 Materials Engineering NEDUET Karachi 23
- 24. 27/09/2010 Materials Engineering NEDUET Karachi 24
- 25. 27/09/2010 Materials Engineering NEDUET Karachi 25
- 26. Slag Reduction • Although the oxidation of alloys is prevented as far as possible, furnace operations will result in some valuable elements such as Cr being present in the slag phase at the end of the operation. For economic reasons, these need to be recovered by the addition of reductants such as FeSi onto the slag which melt and react with the slag oxides, according to: 27/09/2010 Materials Engineering NEDUET Karachi 26
- 27. Deslagging • A slag door is positioned in the side wall at the back of the furnace. This opening is used by the operator for visual inspection of the inside of the furnace, injection of oxygen and carbon using consumable lances, and for deslagging. • During slag foaming, slag is continuously pouring out through the slag door and a limited deslagging is achieved. • A more deliberate deslagging is made whenever a new slag is needed in order to perform the next metallurgical operation. This is always the case when a shift in oxygen potential is needed, e.g. shifting from highly oxidizing operating conditions to reducing conditions. 27/09/2010 Materials Engineering NEDUET Karachi 27
- 28. 27/09/2010 Materials Engineering NEDUET Karachi 28
- 29. 27/09/2010 Materials Engineering NEDUET Karachi 29
- 30. Tapping • Tapping of the furnace is initiated by the operator when the processing in the furnace is finalized and the target temperature has been reached. Tapping should be performed as fast as possible in order to save time. • There are two common furnace designs that have different tapping configurations. 1. Eccentrically-Bottom Tapping (EBT) furnaces have a taphole positioned off-center in the base of the furnace. Such a configuration enables slag-free tapping. In these cases a "hot heel" (small amount of remaining metal and slag) is retained in the furnace between the heats. 2. Spout furnaces are used for some steel grades. Tapping via a spout causes the slag to be carried over to the ladle, where it is thoroughly mixed with the steel. In these cases all the metal is poured out, without any hot heel remaining in the furnace. 27/09/2010 Materials Engineering NEDUET Karachi 30
- 31. 27/09/2010 Materials Engineering NEDUET Karachi 31
- 32. 27/09/2010 Materials Engineering NEDUET Karachi 32
