Drug antagonism
- 1. DRUG ANTAGONISM Prof. Amol B. Deore Department of Pharmacology MVP’s Institute of Pharmaceutical Sciences, Nashik
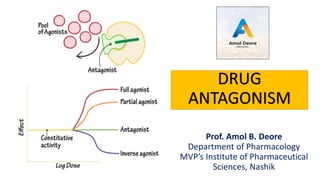
- 2. The effect of one drug blocked (or inhibited) due to another drug is said to be antagonism. In other word, an interaction between two or more drugs that have opposite effects on the body. Drug antagonism may block or reduce effectiveness of one or more of the drugs. Drug Antagonism
- 3. Types of antagonism Pharmacological antagonism: Competitive and Non-Competitive Physiological antagonism Chemical antagonism
- 5. Competitive Antagonism If both the agonist and the antagonist compete for the same receptor in a reversible manner, they are said to be “competitive.” The antagonist drug interacts with the receptor, and blocks it. Therefore it does not produce pharmacological action. The extent of antagonism depends on number of receptors occupied by the both drugs (agonist and antagonist), their affinity for receptors and their concentration.
- 6. Competitive Antagonism Drug A: An agonist Drug C: An antagonist of Drug A that binds to the same receptor sites as Drug A and prevents Drug A from binding Molecule of Drug C bound to receptor site
- 7. Competitive Antagonism The increase in concentration of either one of these drugs can displace the other from receptor binding sites. Drugs interact with their receptors by weak bonds i.e. ionic bond or Hydrogen bond or Vander wal force. Hence duration of action of drug is short. Both agonist and antagonist have chemical resemblance (structural similarity).
- 8. Ex. Competitive antagonist and their receptors Agonist Competitive Antagonist Receptors Acetylcholine Adrenaline Histamine Morphine Atropine Phentolamine Cetrizine Naloxone Cholinergic receptors Adrenergic receptors Histamine receptors Opioid receptors
- 9. Non-Competitive Antagonism In this situation, the antagonist forms a more stable bond (covalent bond) with its receptors. This drug-receptor interaction is irreversible and long lasting. One drug bind to the receptor in a manner which makes it is impossible to reverse the binding (e.g., a strong covalent bond).
- 10. Non-competitive Antagonism Drug A: An agonist Drug D: A non-competitive antagonist of Drug A that binds to different receptor sites from Drug A but still prevents Drug A from binding Molecule of Drug D bound to receptor site different from receptor site for Drug A
- 11. The antagonist may bind with different receptor in such a way that agonist cannot able to produce its action. The antagonist has no chemical resemblance with agonist. A fundamental difference between competitive and non- competitive antagonists is that competitive agonists reduce agonist potency and non-competitive antagonists reduce agonist efficacy.
- 12. Ex. Non-competitive antagonist and their receptors Agonist Non-competitive Antagonist Receptors Nor-Adrenaline Acetylcholine Diazepam Organophosphorous compounds Phenoxybenzamine Bungarotoxin Bicuculine Atropine sulphate alpha receptors Cholinergic receptors GABA receptors Cholinergic receptors
- 13. Physiological Antagonism In this situation, both an agonist and antagonist interact with different receptors and have different site of action, but still their actions are opposite to each other. Ex. glucocorticoids increase blood sugar level, and insulin lowers it, but the two drugs act by completely different pathways.
- 14. Chemical Antagonism In this situation, a chemical reaction take place between two or more drugs within the body and hence biological activity become blocked or diminished. Ex. protamine acts as antidote of heparin molecules • Antacids neutralize the gastric acid • Dimercaprol forms chelate with mercury.
- 15. Prof. Amol B. Deore Department of Pharmacology MVP’s Institute of Pharmaceutical Sciences, Nashik
