Prodrug as drug career
•
22 recomendaciones•1,589 vistas
PRODRUG AND ITS TYPES, VARIOUS APPLICATIONS IN DRUG TARGETING
Denunciar
Compartir
Denunciar
Compartir
Recomendados
Recomendados
THE PRESENTATION GIVES DETAILED EXPLANATION ON PRODRUGS AND HOW TO DESIGN AN PRODRUG.Rationale of prodrug design and practical consideration of

Rationale of prodrug design and practical consideration ofCollege of Pharmacy,Sri Ramakrishna Institute of Paramedical Sciences,Coimbatore
(a) Drugs Affecting the Central Nervous System: Morphine alkaloids
(b) Anticancer Drugs: Paclitaxel and Docetaxel, Etoposide and Teniposide
(c) Cardiovascular Drugs: Lovastatin, Teprotide, and Dicoumarol
(d) Neuromuscular Blocking Drugs: Curare alkaloids
(e) Anti-malarial drugs and analogs
(f) Chemistry of macrolide antibiotics (Erythromycin, Azithromycin, Roxithromycin, and Clarithromycin) and β - Lactam antibiotics (Cephalosporins and Carbapenem)study of natural products as leads for new pharmaceuticals for the various cl...

study of natural products as leads for new pharmaceuticals for the various cl...Subham Kumar Vishwakarma
Más contenido relacionado
La actualidad más candente
THE PRESENTATION GIVES DETAILED EXPLANATION ON PRODRUGS AND HOW TO DESIGN AN PRODRUG.Rationale of prodrug design and practical consideration of

Rationale of prodrug design and practical consideration ofCollege of Pharmacy,Sri Ramakrishna Institute of Paramedical Sciences,Coimbatore
(a) Drugs Affecting the Central Nervous System: Morphine alkaloids
(b) Anticancer Drugs: Paclitaxel and Docetaxel, Etoposide and Teniposide
(c) Cardiovascular Drugs: Lovastatin, Teprotide, and Dicoumarol
(d) Neuromuscular Blocking Drugs: Curare alkaloids
(e) Anti-malarial drugs and analogs
(f) Chemistry of macrolide antibiotics (Erythromycin, Azithromycin, Roxithromycin, and Clarithromycin) and β - Lactam antibiotics (Cephalosporins and Carbapenem)study of natural products as leads for new pharmaceuticals for the various cl...

study of natural products as leads for new pharmaceuticals for the various cl...Subham Kumar Vishwakarma
La actualidad más candente (20)
Rationale of prodrug design and practical consideration of

Rationale of prodrug design and practical consideration of
Role of chirality in stereoselective and specific theraputic agent

Role of chirality in stereoselective and specific theraputic agent
study of natural products as leads for new pharmaceuticals for the various cl...

study of natural products as leads for new pharmaceuticals for the various cl...
Medicinal Chemistry of Antihypertensive agents pptx

Medicinal Chemistry of Antihypertensive agents pptx
Sythesis of heterocyclic drugs ketoconazole and metronidazole

Sythesis of heterocyclic drugs ketoconazole and metronidazole
Destacado
Destacado (20)
Prodrug approaches for cns delivery ppt finished copy

Prodrug approaches for cns delivery ppt finished copy
Prodrugs an approach to solve problems related to adme

Prodrugs an approach to solve problems related to adme
Parenteral controlled drug delivery system sushmitha

Parenteral controlled drug delivery system sushmitha
Similar a Prodrug as drug career
Similar a Prodrug as drug career (20)
Basic concepts of Prodrug & their application in pharmacy fields

Basic concepts of Prodrug & their application in pharmacy fields
Prodrug basic concepts and application of Prodrug Design.pptx

Prodrug basic concepts and application of Prodrug Design.pptx
Más de abhishek rai
Más de abhishek rai (9)
Último
PEMESANAN OBAT ASLI : +6287776558899
Cara Menggugurkan Kandungan usia 1 , 2 , bulan - obat penggugur janin - cara aborsi kandungan - obat penggugur kandungan 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 bulan - bagaimana cara menggugurkan kandungan - tips Cara aborsi kandungan - trik Cara menggugurkan janin - Cara aman bagi ibu menyusui menggugurkan kandungan - klinik apotek jual obat penggugur kandungan - jamu PENGGUGUR KANDUNGAN - WAJIB TAU CARA ABORSI JANIN - GUGURKAN KANDUNGAN AMAN TANPA KURET - CARA Menggugurkan Kandungan tanpa efek samping - rekomendasi dokter obat herbal penggugur kandungan - ABORSI JANIN - aborsi kandungan - jamu herbal Penggugur kandungan - cara Menggugurkan Kandungan yang cacat - tata cara Menggugurkan Kandungan - obat penggugur kandungan di apotik kimia Farma - obat telat datang bulan - obat penggugur kandungan tuntas - obat penggugur kandungan alami - klinik aborsi janin gugurkan kandungan - ©Cytotec ™misoprostol BPOM - OBAT PENGGUGUR KANDUNGAN ®CYTOTEC - aborsi janin dengan pil ©Cytotec - ®Cytotec misoprostol® BPOM 100% - penjual obat penggugur kandungan asli - klinik jual obat aborsi janin - obat penggugur kandungan di klinik k-24 || obat penggugur ™Cytotec di apotek umum || ®CYTOTEC ASLI || obat ©Cytotec yang asli 200mcg || obat penggugur ASLI || pil Cytotec© tablet || cara gugurin kandungan || jual ®Cytotec 200mcg || dokter gugurkan kandungan || cara menggugurkan kandungan dengan cepat selesai dalam 24 jam secara alami buah buahan || usia kandungan 1_2 3_4 5_6 7_8 bulan masih bisa di gugurkan || obat penggugur kandungan ®cytotec dan gastrul || cara gugurkan pembuahan janin secara alami dan cepat || gugurkan kandungan || gugurin janin || cara Menggugurkan janin di luar nikah || contoh aborsi janin yang benar || contoh obat penggugur kandungan asli || contoh cara Menggugurkan Kandungan yang benar || telat haid || obat telat haid || Cara Alami gugurkan kehamilan || obat telat menstruasi || cara Menggugurkan janin anak haram || cara aborsi menggugurkan janin yang tidak berkembang || gugurkan kandungan dengan obat ©Cytotec || obat penggugur kandungan ™Cytotec 100% original || HARGA obat penggugur kandungan || obat telat haid 1 bulan || obat telat menstruasi 1-2 3-4 5-6 7-8 BULAN || obat telat datang bulan || cara Menggugurkan janin 1 bulan || cara Menggugurkan Kandungan yang masih 2 bulan || cara Menggugurkan Kandungan yang masih hitungan Minggu || cara Menggugurkan Kandungan yang masih usia 3 bulan || cara Menggugurkan usia kandungan 4 bulan || cara Menggugurkan janin usia 5 bulan || cara Menggugurkan kehamilan 6 Bulan
________&&&_________&&&_____________&&&_________&&&&____________
Cara Menggugurkan Kandungan Usia Janin 1 | 7 | 8 Bulan Dengan Cepat Dalam Hitungan Jam Secara Alami, Kami Siap Meneriman Pesanan Ke Seluruh Indonesia, Melputi: Ambon, Banda Aceh, Bandung, Banjarbaru, Batam, Bau-Bau, Bengkulu, Binjai, Blitar, Bontang, Cilegon, Cirebon, Depok, Gorontalo, Jakarta, Jayapura, Kendari, Kota Mobagu, Kupang, LhokseumaweCara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...Cara Menggugurkan Kandungan 087776558899
Último (20)
💚Call Girls In Amritsar 💯Anvi 📲🔝8725944379🔝Amritsar Call Girl No💰Advance Cash...

💚Call Girls In Amritsar 💯Anvi 📲🔝8725944379🔝Amritsar Call Girl No💰Advance Cash...
Goa Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Goa No💰Advanc...

Goa Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Goa No💰Advanc...
Cardiac Output, Venous Return, and Their Regulation

Cardiac Output, Venous Return, and Their Regulation
Pune Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Pune No💰Adva...

Pune Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Pune No💰Adva...
Call Girl in Chennai | Whatsapp No 📞 7427069034 📞 VIP Escorts Service Availab...

Call Girl in Chennai | Whatsapp No 📞 7427069034 📞 VIP Escorts Service Availab...
Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...

Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...
Kolkata Call Girls Shobhabazar 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Gir...

Kolkata Call Girls Shobhabazar 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Gir...
Exclusive Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bangal...

Exclusive Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bangal...
Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...

Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...
Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...

Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...
7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta

7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta
Call girls Service Phullen / 9332606886 Genuine Call girls with real Photos a...

Call girls Service Phullen / 9332606886 Genuine Call girls with real Photos a...
Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...

Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...
Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...

Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...
💰Call Girl In Bangalore☎️7304373326💰 Call Girl service in Bangalore☎️Bangalor...

💰Call Girl In Bangalore☎️7304373326💰 Call Girl service in Bangalore☎️Bangalor...
Premium Call Girls Nagpur {9xx000xx09} ❤️VVIP POOJA Call Girls in Nagpur Maha...

Premium Call Girls Nagpur {9xx000xx09} ❤️VVIP POOJA Call Girls in Nagpur Maha...
Call Girls in Lucknow Just Call 👉👉 8875999948 Top Class Call Girl Service Ava...

Call Girls in Lucknow Just Call 👉👉 8875999948 Top Class Call Girl Service Ava...
Call Girls Kathua Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kathua Just Call 8250077686 Top Class Call Girl Service Available
Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...
Prodrug as drug career
- 2. • Prodrug • Aim of prodrug design • Objectives of prodrug research • Classification • Carrier linked prodrugs • Bioprecursor prodrugs • Steps in prodrug design • Applications
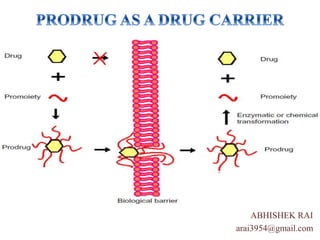
- 3. • pharmacologically inert chemical derivative that can be converted in vivo, enzymatically and/or a chemical transformation, to the active drug that exerts the intended therapeutic effect(s). • Bio-reversible derivatives of drug molecules. • Ideally, the prodrug should be converted to the parent drug as soon as it reach its goal, and then followed by the subsequent rapid metabolism and/or elimination of the released active group
- 4. • To mask undesirable drug properties, such as: – Physical Properties – Poor aqueous solubility – Low lipophilicity – low target selectivity, – chemical instability, – undesirable taste, – irritation or pain after local administration, – presystemic metabolism and – toxicity. – Pharmacokinetic Properties – Poor distribution across biological membranes – Good substrate for first-pass metabolism – Rapid absorption/excretion when long-term effect desired – Not site-specific
- 5. • To optimize: – absorption, – distribution, – metabolism, – excretion, and – unwanted toxicity of the parent drugs.
- 6. • According to Testa there are three basic, overlapping objectives in prodrug research: • 1. Pharmaceutical: to improve solubility, chemical stability, and organoleptic properties; to decrease irritation and/or pain after local administration, to reduce problems related with the pharmaceutical technology of the active agent. • 2. Pharmacokinetic: to improve absorption (oral and by non-oral routes), to decrease presystemic metabolism to improve time profile, to increase organ/ tissue-selective delivery of the active agent. • 3. Pharmacodynamic: to decrease toxicity and improve therapeutic index, to design single chemical entities combining two drugs.
- 7. • Classified into :- – Carrier Linked Prodrug: the active agent is attached through the metabolically labile linkage to a carrier or promoiety which is not necessary for the activity, but may impart some desirable properties of the drug. – Bioprecursor: contain no promoiety but is rather reliant on the metabolism by processes such as oxidation, reduction, phosphorylation, and sulfation activations to introduce the functionality necessary to create the desired active agent unlike the hydrolytic activation of the carrier-linked prodrugs.
- 8. • Bipartate Prodrug – This prodrug consists of the active drug covalently linked to an inert carrier or transport moiety, generally ester or amide. – Have greatly modified lipophilicity due to the attached carrier. – The active drug is released by hydrolytic cleavage either chemically or enzymatically.
- 9. • Tripartate Prodrug – Drug moiety is not directly attached to the carrier moiety. – First the drug moiety is attached with the linker and then this linker is attached with carrier.
- 10. • Mutual Prodrug – Carrier used is another biologically active drug instead of some inert molecule. – A mutual prodrug consists of two pharmacologically active agents which are coupled together in such a way to act as a promoiety for the other agent and vice versa. – The carrier selected may have the same biological action as that of the parent drug and thus might give synergistic action, or the carrier may have some additional biological action that is lacking in the parent drug, thus ensuring some additional benefit.
- 11. • Bioprecursor prodrugs do not include any carrier molecule. • An inactive drug undergoes chemical modification to convert into a compound which itself is active drug or further metabolized into active form which have a desired therapeutic efficacy. • The chemical reactions which can occur in this process include oxidation, reduction, phosphorylation, or sulfation .
- 12. Following steps to be followed for designing a prodrug: – Identification of drug delivery problem – Identification of desired physicochemical properties – Selection of transport moiety which will give prodrug desired transport properties be readily cleaved in the desired biological compartment.
- 13. Brain Targeting Colon Targeting To enhance aqueous solubility Odour masking Reduction of gastrointestinal irritation Drug targeting Reduction of stability problem Increase in patient compliance
- 14. • BRAIN TARGETING – The hydrophilic drugs can not be administered as such in the brain and thus the therapeutic efficacy cannot be achieved. – The hydrophilic drugs can be converted into their lipophilic forms to achieve the goal but this leads to the systemic toxicity due to the penetration of drugs into the other tissues. – Eg, • Dihydropridine pyridinium type redox system was developed for brain specific sustained delivery of drug. • The drug containing amine group is made lipophilic by coupling to dihydropyridine promoiety that facilitate penetration of prodrug through the blood brain barrier. • In the CNS, dihydropridine group converted to polar pyridinium salt, thus becomes poorly permeable to blood brain barrier and thus remain at the site and cleavage provides sustained release for action.
- 15. • COLON TARGETING – For sulphasalazine formed by coupling of diazotized sulphanilamide pyridine with 5-amino salicylicacid (ASA). – On oral administration, intact sulphasalazine reaches the colon. The azo reductase associated with colonic microflora convert sulphasalazin to its constituents entities, the active species 5ASA available for absorption in colon, while precolonic absorption responsible for side effects is reduced. • TO ENHANCE AQUEOUS SOLUBILITY – Prednisolone is a poorly aqueous soluble drug which should be converted into water soluble form for their delivery into the body. – prodrug prednisolone phosphate is prepared which is activated in vivo by the phosphotase.
- 16. • Odour masking – The odour of a compound is due to its vapour pressure. Compounds with Low boiling point will have a strong odour. – Eg, • Ethyl mercapta have a Foul smell is a liquid of boiling point 35°C useful in the treatment of leprosy. • Its foul smell is removed by converting it into its phthalate ester, diethyl isophthalate, which has higher boiling point and odorless. • The prodrug is administered by rubbing on the skin, after absorption, the ester is metabolized to parent drug by thioesterase. • Reduction of gastrointestinal irritation – Many drugs react with the acid secretion in stomach and causes gastric irritation for e.g NSAIDS. – In case of NSAIDS, this problem can be overcome by preparation of prodrug salsalates.
- 17. • Reduction of stability problem – Most of the drugs may be rapidly metabolized and rendered inactive before it reaches the site of action. – The structure may be modified to block the metabolism until the drug is at the desired site. – Some prodrugs protect the drug from first-pass effect. e.g: Propranolol. • It is a widely used antihypertensive drug, but its oral bioavalability is very low because of first pass metabolism. • The hemisuccinate ester of propranolol was prepared to block glucuronide formation. Hence oral administration of propranolol hemisuccinate elevates plasma levels of propranolol for about 8 times.
- 18. • Drug targeting – The distribution of drug to non targeted tissues causes toxicity and reduction of drug concentration in the target tissues due to which optimum therapeutic action can not be achieved. – So for obtaining desired therapeutic action drug targeting is necessary and this can be easily achieved by prodrug approach. • Increase in patient compliance – Many drugs have unpleasant taste, odour and some may causes GIT irritation and when given in form of injection causes pain. These problems cause poor patient compliance.