Basics of Insulin: Its History, Chemistry, Mechanism of Action and Physiologic Functions
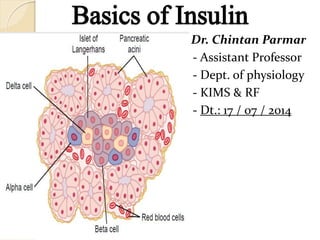
- 1. Basics of Insulin Dr. Chintan Parmar - Assistant Professor - Dept. of physiology - KIMS & RF - Dt.: 17 / 07 / 2014
- 2. Outline - Introduction - Brief history - Chemistry and Synthesis - Mechanism of action - Physiologic functions - Mechanisms of Secretion - Factors affecting Secretion
- 3. Intro. - The Pancreas Digestive + Endocrine functions Insulin and Glucagon - crucial for normal regulation of glucose, lipid and protein metabolism (1) the acini, which secrete digestive juices into the duodenum, (2) the islets of Langerhans, which secrete insulin and glucagon directly into the blood - three major types of cells; alpha, beta and delta cells + PP cells Beta cells – 60 % - insulin, Alpha – 25 % - glucagon, Delta – 10 % - somatostatin, PP - pancreatic polypeptide
- 5. Insulin Insulin is the first hormone to be isolated, purified, crystallized & synthesized. Insulin secretion is associated with energy abundance. When there is great abundance of energy- giving foods in the diet, especially excess amounts of carbohydrates, insulin is secreted in great quantity.
- 6. Insulin The insulin plays an important role in storing the excess energy. In the case of excess carbohydrates, it causes them to be stored as glycogen mainly in the liver and muscles. All the excess carbohydrates that cannot be stored as glycogen are converted under the stimulus of insulin into fats and stored in the adipose tissue.
- 7. Insulin In the case of proteins, insulin has a direct effect in promoting amino acid uptake by cells and conversion of these amino acids into protein. In addition, it inhibits the breakdown of the proteins that are already in the cells. Anabolic
- 8. HistoryInsulin was first isolated from the pancreas in 1922 by Banting and Best, and almost overnight the outlook for the severely diabetic patient changed from one of rapid decline and death to that of a nearly normal person. Children dying from diabetic ketoacidosis were kept in large wards, often with 50 or more patients in a ward, mostly comatose. Grieving family members were often in attendance, awaiting the (until then, inevitable) death. In one of medicine's more dramatic moments, Banting, Best and Collip went from bed to bed, injecting an entire ward with the new purified extract. Before they had reached the last dying child, the first few were awakening from their coma, to the joyous exclamations
- 9. Frederick Banting joined by Charles Best, 1924 The Nobel Prize committee in 1923 credited the practical extraction of insulin to a team at the University of Toronto and awarded the Nobel Prize to two men: Frederick Banting and J.J.R. Macleod. They were awarded the Nobel Prize in Physiology or Medicine in 1923 for the discovery of insulin. Banting, insulted that Best was not mentioned, shared his prize with him, and Macleod immediately shared his with
- 10. Insulin Chemistry and Synthesis Insulin is a small protein; - has a molecular weight of 5808. It is composed of two amino acid chains connected to each other by disulfide linkages Beta cells - beginning with translation of the insulin RNA by ribosomes attached to the ER to form an insulin preprohormone (11500) - cleaved in the ER to form a proinsulin (9000) - further cleaved in the Golgi apparatus to form insulin - secretory granules unbound form; it has a plasma half-life that averages only about 6 minutes - degraded by the enzyme insulinase mainly in the liver, to a lesser extent in the kidneys and muscles
- 11. C – peptide (Connecting Peptide) has no biological activity, but its estimation in plasma serves as an useful index for the endogenous production of insulin.
- 12. MOA Insulin first binds with and activates a membrane receptor protein (300,000) The insulin receptor is a combination of four subunits held together by disulfide linkages: Two alpha subunits that lie entirely outside the cell membrane Two beta subunits that penetrate through the membrane, protruding into the cell cytoplasm
- 13. MOA Insulin binds with alpha ↓ beta unit autophosphorylated ↓ tyrosine kinase ↓ phosphorylation of multiple other intracellular enzymes including a group called insulin-receptor substrates (IRS)
- 15. MOAWithin seconds, the membranes of about 80 per cent of the body’s cells markedly increase their uptake of glucose. This is especially true of muscle cells, adipose cells & liver cells but is not true of most neurons in the brain The cell membrane becomes more permeable to many of the amino acids, K ions and phosphate ions, causing increased transport of these substances into the cell
- 16. MOA Slower effects occur during the next 10 to 15 minutes to change the activity levels of many more intracellular metabolic enzymes Much slower effects continue to occur for hours and even several days. They result from changed rates of translation of messenger RNAs at the ribosomes to form new proteins and still slower effects from changed rates of transcription of DNA
- 18. Carbohydrate Metabolism - Muscle Immediately after a high-carbohydrate meal, the glucose that is absorbed into the blood causes rapid secretion of insulin The normal resting muscle membrane is only slightly permeable to glucose, except when the muscle fiber is stimulated by insulin – so During much of the day, muscle tissue depends not on glucose for its energy but on fatty acids Moderate or heavy exercise – exercising muscle fibers become more permeable to glucose even in the absence of insulin
- 19. Carbohydrate Metabolism - Liver Glucose absorbed after a meal to be stored almost immediately in the liver in the form of glycogen - Between meals – liver glycogen – glucose 1. Insulin inactivates liver phosphorylase - enzyme that causes liver glycogen to split into glucose. This prevents breakdown of the glycogen that has been stored in the liver cells. 2. It increases the activity of the enzyme glucokinase, which is one of the enzymes that causes the initial phosphorylation of glucose after it diffuses into the liver cells - phosphorylated glucose cannot diffuse back through the cell membrane
- 20. Carbohydrate Metabolism - Liver 3. Insulin also increases the activities of the enzymes that promote glycogen synthesis, including glycogen synthase - polymerization of the monosaccharide units to form the glycogen 4. Enzyme glucose phosphatase inhibited 5. Glycolysis (oxidation of glucose) is increased in muscle & liver by activating enzyme phosphofructokinase
- 21. Carbohydrate Metabolism - Liver Glucose Is Released from the Liver Between Meals 1. The decreasing blood glucose causes the pancreas to decrease its insulin secretion. 2. Stopping further synthesis of glycogen in the liver and preventing further uptake of glucose by the liver from the blood. 3. The lack of insulin along with increase of glucagon, activates the enzyme phosphorylase, which causes the splitting of glycogen into glucose phosphate. 4. The enzyme glucose phosphatase, becomes activated by the insulin lack and causes the phosphate radical to split away from the glucose
- 22. Carbohydrate Metabolism When the quantity of glucose entering the liver cells is more than can be stored as glycogen, insulin promotes the conversion of all this excess glucose into fatty acids – triglycerides in VLDL - adipose tissue and deposited as fat Insulin also inhibits gluconeogenesis & glycogenolysis. Thus inhibiting glucose production Insulin decreases the release of amino acids from muscle and other extrahepatic tissues and in turn the availability of these necessary precursors required for gluconeogenesis
- 23. Fat Metabolism - Liver Insulin increases the utilization of glucose by most of the body’s tissues – fat sparer. Promotes fatty acid synthesis in liver from excess glucose 1. Insulin increases the transport of glucose into the liver cells – extra glucose via glycolytic pathway – pyruvate – acetyl CoA – fatty acids 2. Energy from glucose via citric acid cycle - excess of citrate and isocitrate ions - activates acetyl CoA carboxylase – acetyl CoA to form malonyl CoA
- 24. Fat Metabolism – Adipose Tissue Fat storage in adipose tissue 1. Fatty acids (triglycerides) are then transported from the liver by way of the blood lipoproteins to the adipose cells. 2. Insulin activates lipoprotein lipase - splits the triglycerides again into fatty acids, a requirement for them to be absorbed into the adipose cells - again converted to triglycerides and stored
- 25. Fat Metabolism – Adipose Tissue - Insulin promotes glucose transport through the cell membrane into the fat cells - large quantities of alpha glycerol phosphate - supplies the glycerol that combines with fatty acids to form the triglycerides - Insulin inhibits the action of hormone- sensitive lipase – no hydrolysis of the triglycerides stored in the fat cells - release of fatty acids from the adipose tissue into the circulating blood is inhibited
- 26. Fat Metabolism Insulin deficiency - free fatty acid becomes the main energy substrate used by essentially all tissues of the body besides the brain – ketoacidosis – coma, death The excess of fatty acids in the plasma also promotes liver conversion of some of the fatty acids into phospholipids and cholesterol - atherosclerosis
- 28. Protein Metabolism and Growth 1. Insulin stimulates transport of many of the amino acids into the cells 2. Insulin increases the rate of transcription of selected DNA genetic sequences 3. Insulin increases the translation of mRNA 4. Insulin inhibits the catabolism of proteins 5. In the liver, insulin depresses the rate of gluconeogenesis - conserves the amino acids in the protein stores of the body Insulin deficiency – enhanced urea excretion in the urine - protein wasting – weakness Insulin and Growth Hormone Interact Synergistically to Promote Growth
- 30. Confused ??? Here is the summary…
- 31. Effects of insulin on various tissues Adipose issue Increased glucose entry Increased fatty acid synthesis Increased glycerol phosphate synthesis Increased triglyceride deposition Activation of lipoprotein lipase Inhibition of hormone-sensitive lipase Increased K+ uptake Muscle Increased glucose entry Increased glycogen synthesis Increased amino acid uptake Increased protein synthesis in ribosomes Decreased protein catabolism Decreased release of gluconeogenic amino acids Increased K+ uptake
- 32. Effects of insulin on various tissues Liver Decreased ketogenesis Increased protein synthesis Increased lipid synthesis Decreased gluconeogenesis Increased glycogen synthesis General Increased cell growth
- 33. Insulin also increase in the secretion of HCL by parietal cells in the stomach via vagus nerve Insulin test is done to check whether vagotomy is complete or not, as in case of treatment of peptic ulcer
- 34. Mechanisms of Insulin Secretion
- 36. Fasting level of blood glucose of 80 to 90 mg/100 ml, the rate of insulin secretion is minimal — 25 ng/kg of body weight per minute
- 37. Biphasic insulin response to Glucose, 1st rapid phase – preformed, 2nd slow rise phase - new
- 38. Glucagon Growth Hormone Insulin Vs Cortisol Epinephrine Thyroid hormone
- 39. Thank You…
