Lecture 12.3- Limiting Reagents and Percent Yield
•Descargar como PPT, PDF•
6 recomendaciones•4,008 vistas
Section 12.3 Lecture for Honors Chemistry Slides 1-7 are for Prep as well
Denunciar
Compartir
Denunciar
Compartir
Recomendados
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
SHS STEM General Chemistry 1: Atoms, Moles, Equations, Stoichiometry

SHS STEM General Chemistry 1: Atoms, Moles, Equations, Stoichiometry
Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions

Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions
Law of multiple proportions and law of definite proportions

Law of multiple proportions and law of definite proportions
Chapter 9.3 : Limiting Reactants and Percent Yield

Chapter 9.3 : Limiting Reactants and Percent Yield
Organic compound nomenclature (ALkanes, ALKYL GROUP, ALKENE, ALKYNES)

Organic compound nomenclature (ALkanes, ALKYL GROUP, ALKENE, ALKYNES)
Destacado
Destacado (7)
Chapter10 section02 Mole–Mass and Mole–Volume Relationships By Hamdy Karim

Chapter10 section02 Mole–Mass and Mole–Volume Relationships By Hamdy Karim
Chapter10 section03 Percent Composition and Chemical Formulas By Hamdy Karim.

Chapter10 section03 Percent Composition and Chemical Formulas By Hamdy Karim.
Similar a Lecture 12.3- Limiting Reagents and Percent Yield
Similar a Lecture 12.3- Limiting Reagents and Percent Yield (20)
526128650-Limiting-Reactants-and-the-Product-Formed.pdf

526128650-Limiting-Reactants-and-the-Product-Formed.pdf
526128650-Limiting-Reactants-and-the-Product-Formed.pdf

526128650-Limiting-Reactants-and-the-Product-Formed.pdf
2011 topic 01 lecture 3 - limiting reactant and percent yield

2011 topic 01 lecture 3 - limiting reactant and percent yield
Más de Mary Beth Smith
Más de Mary Beth Smith (20)
Chapter 3 and 5 lecture- Ecology & Population Growth

Chapter 3 and 5 lecture- Ecology & Population Growth
Biotechnology Chapter Five Lecture- Proteins (part b)

Biotechnology Chapter Five Lecture- Proteins (part b)
Biotechnology Chapter Five Lecture- Proteins (part a)

Biotechnology Chapter Five Lecture- Proteins (part a)
Biotechnology Chapter Two Lecture- Cells and Macromolecules

Biotechnology Chapter Two Lecture- Cells and Macromolecules
Último
HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...

HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...Nguyen Thanh Tu Collection
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...Postal Advocate Inc.
FINALS_OF_LEFT_ON_C'N_EL_DORADO_2024.pptx

FINALS_OF_LEFT_ON_C'N_EL_DORADO_2024.pptxConquiztadors- the Quiz Society of Sri Venkateswara College
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
YOUVE GOT EMAIL_FINALS_EL_DORADO_2024.pptx

YOUVE GOT EMAIL_FINALS_EL_DORADO_2024.pptxConquiztadors- the Quiz Society of Sri Venkateswara College
Último (20)
HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...

HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...
TataKelola dan KamSiber Kecerdasan Buatan v022.pdf

TataKelola dan KamSiber Kecerdasan Buatan v022.pdf
Inclusivity Essentials_ Creating Accessible Websites for Nonprofits .pdf

Inclusivity Essentials_ Creating Accessible Websites for Nonprofits .pdf
Visit to a blind student's school🧑🦯🧑🦯(community medicine)

Visit to a blind student's school🧑🦯🧑🦯(community medicine)
ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...

ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...
Procuring digital preservation CAN be quick and painless with our new dynamic...

Procuring digital preservation CAN be quick and painless with our new dynamic...
Difference Between Search & Browse Methods in Odoo 17

Difference Between Search & Browse Methods in Odoo 17
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx

MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx
Lecture 12.3- Limiting Reagents and Percent Yield
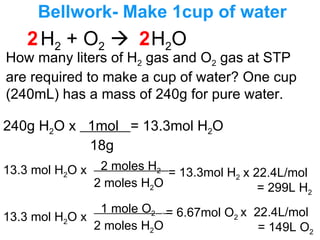
- 1. Bellwork- Make 1cup of water H 2 + O 2 H 2 O How many liters of H 2 gas and O 2 gas at STP are required to make a cup of water? One cup (240mL) has a mass of 240g for pure water. 240g H 2 O x 1mol = 13.3mol H 2 O 18g 13.3 mol H 2 O x 13.3 mol H 2 O x 2 2 2 moles H 2 2 moles H 2 O 1 mole O 2 . 2 moles H 2 O = 13.3mol H 2 = 6.67mol O 2 x 22.4L/mol = 299L H 2 x 22.4L/mol = 149L O 2
- 8. 12.7
- 9. 12.7
- 10. 12.7
- 11. 12.7
- 12. for Sample Problem 12.7
- 13. 12.8Q
- 14. 12.8
- 15. 12.8
- 16. 12.8
- 17. for Sample Problem 12.8
- 21. The actual yield is often lower than the theoretical yield because not all reactions go to completion. The actual yield should never be higher than your theoretical yield.
- 22. 12.9
- 23. 12.9
- 24. 12.9
- 25. 12.9
- 26. 12.10
- 27. 12.10
- 28. 12.10
- 29. 12.10
- 30. for Sample Problem 12.10
Notas del editor
- A batting average is actually a percent yield.