Cavit Financial Model(1)
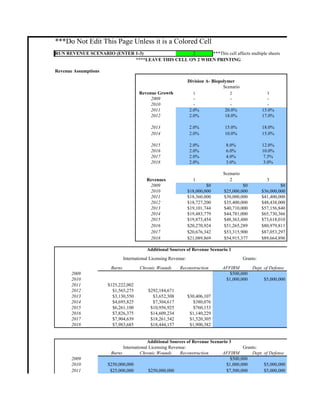
- 1. ***Do Not Edit This Page Unless it is a Colored Cell RUN REVENUE SCENARIO (ENTER 1-3) 2 ***This cell affects multiple sheets ****LEAVE THIS CELL ON 2 WHEN PRINTING Revenue Assumptions Division A- Biopolymer Scenario Revenue Growth 1 2 3 2009 - - - 2010 - - - 2011 2.0% 20.0% 15.0% 2012 2.0% 18.0% 17.0% 2013 2.0% 15.0% 18.0% 2014 2.0% 10.0% 15.0% 2015 2.0% 8.0% 12.0% 2016 2.0% 6.0% 10.0% 2017 2.0% 4.0% 7.5% 2018 2.0% 3.0% 3.0% Scenario Revenues 1 2 3 2009 $0 $0 $0 2010 $18,000,000 $25,000,000 $36,000,000 2011 $18,360,000 $30,000,000 $41,400,000 2012 $18,727,200 $35,400,000 $48,438,000 2013 $19,101,744 $40,710,000 $57,156,840 2014 $19,483,779 $44,781,000 $65,730,366 2015 $19,873,454 $48,363,480 $73,618,010 2016 $20,270,924 $51,265,289 $80,979,811 2017 $20,676,342 $53,315,900 $87,053,297 2018 $21,089,869 $54,915,377 $89,664,896 Additional Sources of Revenue Scenario 1 International Licensing Revenue: Grants: Burns Chronic Wounds Reconstruction AFFIRM Dept. of Defense 2009 $500,000 2010 $1,000,000 $5,000,000 2011 $125,222,002 2012 $1,565,275 $292,184,671 2013 $3,130,550 $3,652,308 $30,406,107 2014 $4,695,825 $7,304,617 $380,076 2015 $6,261,100 $10,956,925 $760,153 2016 $7,826,375 $14,609,234 $1,140,229 2017 $7,904,639 $18,261,542 $1,520,305 2018 $7,983,685 $18,444,157 $1,900,382 Additional Sources of Revenue Scenario 3 International Licensing Revenue: Grants: Burns Chronic Wounds Reconstruction AFFIRM Dept. of Defense 2009 $500,000 2010 $250,000,000 $1,000,000 $5,000,000 2011 $25,000,000 $250,000,000 $7,500,000 $5,000,000
- 2. 2012 $25,000,000 $25,000,000 $250,000,000 $5,000,000 $5,000,000 2013 $25,000,000 $25,000,000 $25,000,000 $5,000,000 $5,000,000 2014 $25,000,000 $25,000,000 $25,000,000 2015 $25,000,000 $25,000,000 $25,000,000 2016 $25,000,000 $25,000,000 $25,000,000 2017 $25,000,000 $25,000,000 $25,000,000 2018 $25,000,000 $25,000,000 $25,000,000 M 2009 2010 2011 Manufacturing (% of Revenues) 50% 50% 50% Lonza Sales Fee (% of Revenues) 15% 15% 15% Total Production Cost (% of Revenues) 65% 65% 65% Lonzas Share of Profit 50% 50% 50% McCoy's Profit (% of Revenues) 17.5% 17.5% 17.5% In HUD Phase 2009 2010 2011 Manufacturing (% of Revenues) 85% 85% 85% Price Paid (per cm³) Administrative (% of Revenues) 15% 15% 15% Selling Price (per cm³) McCoy's Profit (% of Revenues) 0% 0% 0% Manufacturing (% of Revenues) McCoy's Profit (% of Revenues) Expense A 2009 2010 2011 Operating Expenses Manufacturing Expenses- Division A $0 $12,500,000 $15,000,000 Manufacturing Expenses- Division B $0 $23,375,000 $46,750,000 Total Manufacturing Expense $0 $35,875,000 $61,750,000 Employee Expense HDE Application Clinical $30,000 $0 $0 HDE Application Technical $30,000 $0 $0 PMA Application Clinical $0 $100,000 $100,000 PMA Application Technical $0 $100,000 $100,000 Total Employee Expense $60,000 $200,000 $200,000 Laboratory: Trial Tech Transfer Verification Media Prep 1 $200,000 $400,000 $600,000 Media Release 1 $100,000 $200,000 $300,000 Lonza Skin Culture 1 $60,000 $120,000 $180,000 Cincinnati Skin Culture 1 $60,000 $60,000 $60,000 Lonza Mouse Graft Study 1 $50,000 $100,000 $150,000 Total Lab Expenses $470,000 $880,000 $1,290,000 Cost of Goods Sold $530,000 $36,955,000 $63,240,000 Sales, General & Administrative Lonza's "Sales and Marketing" Payments $0 $8,125,000 $9,750,000 CEO/Regulatory $100,000 $200,000 $200,000 COO/Corporate Liason $100,000 $200,000 $200,000 Senior Research Scientist Boyce $100,000 $100,000 $100,000
- 3. Adult Clinical Program Manager $50,000 $100,000 $225,000 National Securities 7% $525,000 $525,000 $0 Accounting Expenses Corporate Counsel $120,000 $200,000 $200,000 Total Personnel Expense $995,000 $9,450,000 $10,675,000 Patents $24,000 $24,000 $175,000 Office Supplies $1,200 $2,400 $4,800 Telephone, Internet Service $5,400 $10,800 $21,600 General Liability (Fire theft) $1,500 $3,000 $6,000 Product Liability $12,000 $24,000 $48,000 Key Man Insurance 3 Employees $5,400 $10,800 $21,600 Director & Officer Insurance $16,000 $32,000 $64,000 Employee Health Insurance $36,000 $72,000 $144,000 Marketing/Tradeshow/Conference $50,000 $100,000 $200,000 Employment Benefits $100,000 $200,000 $400,000 Travel $72,000 $144,000 $288,000 Increase in Patents & Legal due to Licensing Agreements 0 $25,000 $50,000 Increase in Travel due to Licensing Agreements 0 $25,000 $50,000 Additional Administrative Expense Related to HUD $0 $0 $0 Total Other Expenses $323,500 $673,000 $1,473,000 Total SG&A Expenses $1,318,500 $10,123,000 $12,148,000 R&D as % Gross Profit 10.0% R&D Expenses $0 $0 $0 2009 2010 2011 2012 35% 35% 35% 35% Am Years to Amortize Intangibles 15 Total Intangible Assets 2009 2010 2011 2009 $4,000,000 $266,667 $266,667 $266,667 2010 $4,000,000 $266,667 $266,667 2011 $- $0 2012 $- 2013 $- 2014 $- 2015 $- 2016 $- 2017 $- 2018 $- Total Amortization $266,667 $533,333 $533,333 Straight L Years to Depreciat Office Equipment 5 Years to Depreciate CAPEX 15 Office Equipment Purchase Capital Expenditures 2009 2010 2009 $30,000 $6,000 $6,000 2010 $30,000 $6,000
- 4. 2011 $30,000 $- 2012 $30,000 $- 2013 $30,000 $- 2014 $- $- 2015 $- $- 2016 $- $- 2017 $- $- 2018 $- $- Total Depreciation $6,000 $12,000 CASH FLOW 2009 2010 2011 Equity Issuance in Year 1 $7,500,000 $0 $0 Intangible Assets Licensing payment Lonza Marketing & Distribution $2,000,000 $0 $0 Cutanogen Final Payment Lonza $0 $2,000,000 $0 Milestone Payment Boyce HDE app $1,000,000 $0 $0 Milestone Payment University Cincinati HDE APP $1,000,000 $0 $0 Milestone Payment Boyce PMA application $0 $1,000,000 $0 Milestone Payment University Cinncinati PMA APP $0 $1,000,000 $0 Total Intangible Asset Purchased $4,000,000 $4,000,000 $0 Amortization $266,667 $533,333 $533,333 Net Intangible Value $3,733,333 $7,200,000 $6,666,667 Capital Expenditures Capital Expenditures $0 $0 $0 Asset Purchases Office Equipment $30,000 $30,000 $30,000 Total Capital Expenditures $30,000 $30,000 $30,000 Net Asset Value $24,000 $42,000 $54,000 Balance She 2009 2010 2011 Assets Accounts Receivable (% Revenues) 15% 15% 15% Restricted Cash $0 $0 $0 Other Current Assets $0 $0 $0 Other Current Asssets $0 $0 $0 Existing Goodwill and Intangibles $0 $0 $0 Transaction Goodwill $0 $0 $0 Other Long-Term Assets $0 $0 $0 Liabilities Accounts Payable (% Revenues) 10% 10% 10% Other Current Liabilities $0 $0 $0 Existing Long-Term Debt $0 $0 $0 Exisiting Other Debt Affirm Grant Payment $500,000 $0 Other Long-Term Liabilities $0 $0 $0 Minority Interest $0 $0 $0 % of Dividend to be Paid out of CF After Operations and 80% Investing Activities Post 2009
- 5. % of Dividend to be Paid out of CF After Operations and 80% Investing Activities Post 2009 If debt is incurred, make sure to accou Valuation Assumpt Cost of Equity- CAPM 2009 2010 2011 2012 Risk Free Rate (20-Year Treasury) 4.47% 4.47% 4.47% 4.47% Beta 1.0 1.0 1.0 1.0 Risk Premium 25% 25% 20% 20% Cost of Equity- CAPM 29.5% 29.5% 24.5% 24.5% Cost of Debt Milestone Payments Interest 0% 0% 0% 0% Working Capital Working Capital as % of Revenues 2% Terminal Value Calculation Terminal Growth Rate 3.0% Discount Rate 10.0% 1 2 3 4 2 3 4 5 6 7 8 9 10 11
- 6. Division B- HUD/PMA Scenario Revenue Growth 1 2009 - 2010 - 2011 - 2012 - 2013 - 2014 - 2015 0.0% 2016 0.0% 2017 0.0% 2018 0.0% Scenario Revenues 1 2009 $0 2010 $0 2011 $0 2012 $0 2013 $0 2014 $0 2015 $0 2016 $0 2017 $0 2018 $0 Additional Sources of Revenue Scenario 2 International Licensing Revenue: Total Additional Revenue Burns Chronic Wounds $500,000 2009 $6,000,000 2010 $125,222,002 2011 $125,222,002 $293,749,946 2012 $1,565,275 $292,184,671 $37,188,966 2013 $3,130,550 $3,652,308 $12,380,518 2014 $4,695,825 $7,304,617 $17,978,178 2015 $6,261,100 $10,956,925 $23,575,838 2016 $7,826,375 $14,609,234 $27,686,486 2017 $7,904,639 $18,261,542 $28,328,224 2018 $7,983,685 $18,444,157 Total Additional Total Revenues Revenue Scenario 1 $500,000 2009 $500,000 $256,000,000 2010 $24,000,000 $287,500,000 2011 $143,582,002
- 7. $310,000,000 2012 $312,477,146 $85,000,000 2013 $56,290,710 $75,000,000 2014 $31,864,297 $75,000,000 2015 $37,851,632 $75,000,000 2016 $43,846,761 $75,000,000 2017 $48,362,828 $75,000,000 2018 $49,418,093 Margin Assumptions Division A 2012 2013 2014 2015 2016 50% 50% 50% 50% 50% 15% 15% 15% 15% 15% 65% 65% 65% 65% 65% 50% 50% 50% 50% 50% 17.5% 17.5% 17.5% 17.5% 17.5% Division B In PMA Phase 2012 2013 2014 2015 e Paid (per cm³) $25.00 $25.00 $20.00 $20.00 ing Price (per cm³) $55.00 $55.00 $55.00 $55.00 ufacturing (% of Revenues) 45% 45% 36% 36% Coy's Profit (% of Revenues) 55% 55% 64% 64% Expense Assumptions 2012 2013 2014 2015 2016 $17,700,000 $20,355,000 $22,390,500 $24,181,740 $25,632,644 $70,125,000 $93,500,000 $116,875,000 $119,796,875 $122,791,797 $87,825,000 $113,855,000 $139,265,500 $143,978,615 $148,424,441 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $100,000 $100,000 $0 $0 $0 $100,000 $100,000 $0 $0 $0 $200,000 $200,000 $0 $0 $0 $400,000 $200,000 $0 $0 $0 $200,000 $100,000 $0 $0 $0 $120,000 $60,000 $0 $0 $0 $0 $0 $0 $0 $0 $100,000 $50,000 $0 $0 $0 $820,000 $410,000 $0 $0 $0 $88,845,000 $114,465,000 $139,265,500 $143,978,615 $148,424,441 $11,505,000 $13,230,750 $14,553,825 $15,718,131 $16,661,219 $200,000 $1,000,000 $1,000,000 $1,000,000 $1,000,000 $200,000 $1,000,000 $1,000,000 $1,000,000 $1,000,000 $100,000 $100,000 $100,000 $100,000 $100,000
- 8. $375,000 $375,000 $375,000 $375,000 $375,000 $0 $0 $0 $0 $0 $200,000 $200,000 $200,000 $200,000 $200,000 $12,580,000 $15,905,750 $17,228,825 $18,393,131 $19,336,219 $200,000 $200,000 $200,000 $200,000 $100,000 $9,600 $19,200 $19,200 $19,200 $19,200 $43,200 $86,400 $86,400 $86,400 $86,400 $12,000 $24,000 $24,000 $24,000 $24,000 $96,000 $192,000 $192,000 $192,000 $192,000 $43,200 $86,400 $86,400 $86,400 $86,400 $128,000 $256,000 $256,000 $256,000 $256,000 $288,000 $576,000 $576,000 $576,000 $576,000 $400,000 $800,000 $800,000 $800,000 $800,000 $800,000 $1,600,000 $1,600,000 $1,600,000 $1,600,000 $576,000 $1,152,000 $1,152,000 $1,152,000 $1,152,000 $50,000 $50,000 $50,000 $50,000 $50,000 $50,000 $50,000 $50,000 $50,000 $50,000 $0 $0 $0 $0 $0 $2,696,000 $5,092,000 $5,092,000 $5,092,000 $4,992,000 $15,276,000 $20,997,750 $22,320,825 $23,485,131 $24,328,219 $32,280,495 $7,343,397 $5,539,602 $6,330,054 $7,087,762 Tax Rate Assumption 2013 2014 2015 2016 2017 35% 35% 35% 35% 35% Amortizaiton Schedule 2012 2013 2014 2015 2016 $266,667 $266,667 $266,667 $266,667 $266,667 $266,667 $266,667 $266,667 $266,667 $266,667 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $533,333 $533,333 $533,333 $533,333 $533,333 Straight Line Depreciation Schedule 2011 2012 2013 2014 2015 $6,000 $6,000 $6,000 $0 $0 $6,000 $6,000 $6,000 $6,000 $0
- 9. $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $6,000 $0 $0 $0 $18,000 $24,000 $30,000 $24,000 $18,000 CASH FLOW ASSUMPTIONS 2012 2013 2014 2015 2016 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $533,333 $533,333 $533,333 $533,333 $533,333 $6,133,333 $5,600,000 $5,066,667 $4,533,333 $4,000,000 $0 $0 $0 $0 $0 $30,000 $30,000 $0 $0 $0 $30,000 $30,000 $0 $0 $0 $60,000 $60,000 $36,000 $18,000 $6,000 Balance Sheet Assumption 2012 2013 2014 2015 2016 15% 15% 15% 15% 15% $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 10% 10% 10% 10% 10% $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0
- 10. ebt is incurred, make sure to account for interest Valuation Assumptions 2013 2014 2015 2016 2017 4.47% 4.47% 4.47% 4.47% 4.47% 1.0 1.0 1.0 1.0 1.0 20% 15% 15% 10% 10% 24.5% 19.5% 19.5% 14.5% 14.5% 0% 0% 0% 0% 0% 5 6 7 8 9
- 11. Division B- HUD/PMA Scenario 2 3 - - - - - - - - - - - - 2.5% 5.0% 2.5% 5.0% 2.5% 5.0% 2.5% 5.0% Scenario 2 3 $0 $0 $27,500,000 $90,000,000 $55,000,000 $125,000,000 $82,500,000 $200,000,000 $110,000,000 $600,000,000 $137,500,000 $750,000,000 $140,937,500 $787,500,000 $144,460,938 $826,875,000 $148,072,461 $868,218,750 $151,774,272 $911,629,688 Additional Sources of Revenue Scenario 2 l Licensing Revenue: Grants: Total Additional Reconstruction AFFIRM Dept. of Defense Revenue $500,000 $500,000 $1,000,000 $5,000,000 $6,000,000 $7,500,000 $5,000,000 $137,722,002 $293,749,946 $30,406,107 $37,188,966 $380,076 $12,380,518 $760,153 $17,978,178 $1,140,229 $23,575,838 $1,520,305 $27,686,486 $1,900,382 $28,328,224 Total Revenues 2 3 $500,000 $500,000 $58,500,000 $382,000,000 $222,722,002 $453,900,000
- 12. $411,649,946 $558,438,000 $187,898,966 $742,156,840 $194,661,518 $890,730,366 $207,279,158 $936,118,010 $219,302,064 $982,854,811 $229,074,847 $1,030,272,047 $235,017,874 $1,076,294,583 2017 2018 50% 50% 15% 15% 65% 65% 50% 50% 17.5% 17.5% 2016 2017 2018 $20.00 $20.00 $20.00 $55.00 $55.00 $55.00 36% 36% 36% 64% 64% 64% 2017 2018 $26,657,950 $27,457,689 $125,861,592 $129,008,132 $152,519,542 $156,465,820 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $152,519,542 $156,465,820 $17,327,668 $17,847,498 $1,000,000 $1,000,000 $1,000,000 $1,000,000 $100,000 $100,000
- 13. $375,000 $375,000 $0 $0 $200,000 $200,000 $20,002,668 $20,522,498 $100,000 $100,000 $19,200 $19,200 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $86,400 $86,400 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $24,000 $24,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $192,000 $192,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $86,400 $86,400 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $256,000 $256,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $576,000 $576,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $800,000 $800,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $1,600,000 $1,600,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $1,152,000 $1,152,000 Assumed Growth Rate in Item Line Expense (Year 1 - 5): $50,000 $50,000 $50,000 $50,000 $0 $0 $4,992,000 $4,992,000 $24,994,668 $25,514,498 $7,655,531 $7,855,205 2018 35% 2017 2018 $266,667 $266,667 $266,667 $266,667 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $533,333 $533,333 2016 2017 2018 $0 $0 $0 $0 $0 $0
- 14. $0 $0 $0 $6,000 $0 $0 $6,000 $6,000 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $12,000 $6,000 $0 2017 2018 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $533,333 $533,333 $3,466,667 $2,933,333 $0 $0 $0 $0 $0 $0 $0 $0 2017 2018 15% 15% $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 $0 10% 10% $0 $0 $0 $0 $0 $0 $0 $0 $0 $0
- 15. 2018 4.47% 1.0 10% 14.5% 0% 10 11
- 16. USA STATISTICS USED FOR FORECASTING Total expenditure on health as % GDP (2005) GDP (2008, official exchange rate) S. Korea Total Expenditure on Health as % of GDP (2005] 05.9%² GDP (2008, official exchange rate) $857,500,000,000 Total Health Expenditure $50,592,500,000 Burns Market $69,681,520 Wounds $162,590,214 Reconstructive $12,310,402 Population and GDP figures from CIA world Health Data from WHO 2 Data reported from WHO on Republic of Kor Assumed Licensing Agreement as % of Mkt Value S. Korea Total Health Expenditure $50,592,500,000 Implied Burns Market $69,681,520 Implied Wounds $162,590,214 Implied Reconstructive $12,310,402 Licensing Agreement Payment S. Korea Burns Market $2,787,261 Wounds $6,503,609 Reconstructive $492,416 Burn 1 2009 $- 2010 $- 2011 $125,222,002 2012 $1,565,275 2013 $3,130,550 2014 $4,695,825 2015 $6,261,100 2016 $7,826,375 2017 $7,904,639 2018 $7,983,685
- 17. R&D as % of Gross Profit Under 0% Scenario 5.0%
- 21. Forecasting Licensing Agreements and Royalty P Health Care Expenditure 15.2% Burns Market $14,330,000,000,000 Wounds Reconstructive China Japan India Denmark Finland 4.7% 8.2% 5.0% 9.1% 7.5% $4,222,000,000,000 $4,844,000,000,000 $1,237,000,000,000 $369,600,000,000 $281,200,000,000 $198,434,000,000 $397,208,000,000 $61,850,000,000 $33,633,600,000 $21,090,000,000 $273,304,991 $547,078,268 $85,186,579 $46,323,870 $29,047,453 $637,711,647 $1,276,515,958 $198,768,685 $108,089,029 $67,777,390 $35,347,446 $149,899,445 $5,395,150 $12,986,125 $7,532,973 n and GDP figures from CIA world factbook orted from WHO on Republic of Korea 4% China Japan India "Europe" Denmark $198,434,000,000 $397,208,000,000 $61,850,000,000 $1,564,861,800,000 $33,633,600,000 $273,304,991 $547,078,268 $85,186,579 $2,155,298,692 $46,323,870 $637,711,647 $1,276,515,958 $198,768,685 $5,029,030,282 $108,089,029 $35,347,446 $149,899,445 $5,395,150 $557,200,235 $12,986,125 China Japan India "Europe" Total $10,932,200 $21,883,131 $3,407,463 $86,211,948 $125,222,002 $25,508,466 $51,060,638 $7,950,747 $201,161,211 $292,184,671 $1,413,898 $5,995,978 $215,806 $22,288,009 $30,406,107 Additional Sources of Revenue Hlookup T International Licensing Burn Chronic Wounds 2 3 1 2 3 $- $- $- $- $- $- $250,000,000 $- $- $- $125,222,002 $25,000,000 $- $- $250,000,000 $1,565,275 $25,000,000 $292,184,671 $292,184,671 $25,000,000 $3,130,550 $25,000,000 $3,652,308 $3,652,308 $25,000,000 $4,695,825 $25,000,000 $7,304,617 $7,304,617 $25,000,000 $6,261,100 $25,000,000 $10,956,925 $10,956,925 $25,000,000 $7,826,375 $25,000,000 $14,609,234 $14,609,234 $25,000,000 $7,904,639 $25,000,000 $18,261,542 $18,261,542 $25,000,000 $7,983,685 $25,000,000 $18,444,157 $18,444,157 $25,000,000 Forecasting Figures Scenario 2 Revs as % of Skin Graft Market ($11,000,000,000)
- 22. 0.250% Year 1 0.500% Year 2 0.750% Year 3 1.000% Year 4 1.250% Year 5 Assumed Royalty Payment Assumed Growth in Markets after first 5 years NI Under Revenue Scenario 1 for Valuation S 2009 2010 2011 2012 Revenues $0 $25,000,000 $30,000,000 $35,400,000 COGS $530,000 $10,080,000 $10,670,000 $10,383,600 SG&A $1,318,500 $7,798,000 $8,265,000 $9,757,340 R&D $0 $0 $0 $0 Depreciation $6,000 $12,000 $18,000 $24,000 Amortization $266,667 $533,333 $533,333 $533,333 Pretax Profit ($2,121,167) $6,576,667 $10,513,667 $14,701,727 Tax $0 $2,301,833 $3,679,783 $5,145,604 Corresponding NI ($2,121,167) $4,274,833 $6,833,883 $9,556,122 NI Under Revenue Scenario 2 for Valuation S 2009 2010 2011 2012 Revenues $500,000 $58,500,000 $222,722,002 $411,649,946 COGS $530,000 $36,955,000 $63,240,000 $88,845,000 SG&A $1,318,500 $10,073,000 $12,048,000 $15,176,000 R&D $0 $0 $0 $32,280,495 Depreciation $6,000 $12,000 $18,000 $24,000 Amortization $266,667 $533,333 $533,333 $533,333 Pretax Profit ($1,621,167) $10,926,667 $146,882,669 $274,791,119 Tax $0 $3,824,333 $51,408,934 $96,176,891 Corresponding NI ($1,621,167) $7,102,333 $95,473,735 $178,614,227 Matrix Calculations Revenue Matrix US Company Value with a 0% Chance of the Skin Gra 2009 2010 2011 2012 US 0% Skin Graft $500,000 $31,000,000 $42,500,000 $35,400,000 COGS $530,000 $12,500,000 $15,000,000 $17,700,000 SG&A $1,318,500 $10,073,000 $12,048,000 $15,176,000 R&D $0 $0 $0 $885,000 Depr. & Amort $272,667 $545,333 $551,333 $557,333 EBIT ($1,621,167) $7,881,667 $14,900,667 $1,081,667 Taxes $0 $2,758,583 $5,215,233 $378,583 NI ($1,621,167) $5,123,083 $9,685,433 $703,083 Non-Cash Charges $272,667 $545,333 $551,333 $557,333 Less Net Working Capital ($10,000) ($610,000) ($230,000) $142,000 FCFF ($1,358,500) $5,058,417 $10,006,767 $1,402,417 PV of FCFF ($1,068,339) $3,072,521 $4,883,255 $549,829 Terminal Value $0
- 23. Sum of PV of FCFF $6,698,474 Value of Firm $6,698,474 US 100% Skin Graft Revenues $500,000 $58,500,000 $97,500,000 $117,900,000 COGS $530,000 $36,955,000 $63,240,000 $88,845,000 SG&A $1,318,500 $10,073,000 $12,048,000 $15,176,000 R&D $0 $0 $0 $32,280,495 Depr. & Amort $272,667 $545,333 $551,333 $557,333 EBIT ($1,621,167) $10,926,667 $21,660,667 ($18,958,828) Taxes $0 $3,824,333 $7,581,233 ($6,635,590) NI ($1,621,167) $7,102,333 $14,079,433 ($12,323,238) Non-Cash Charges $272,667 $545,333 $551,333 $557,333 Less Net Working Capital ($10,000) ($1,160,000) ($780,000) ($408,000) FCFF ($1,358,500) $6,487,667 $13,850,767 ($12,173,905) PV of FCFF ($1,068,339) $3,940,659 $6,759,108 ($4,772,883) Terminal Value Sum of PV of FCFF $16,549,853 Value of Firm $16,549,853 International Revenues South Korea Burns $2,787,261 $34,841 Wounds $6,503,609 Reconstructive South Korea Total $2,787,261 $6,538,449 China Burns $10,932,200 $136,652 Wounds $25,508,466 Reconstructive China Total $10,932,200 $25,645,118 Japan Burns $21,883,131 $273,539 Wounds $51,060,638 Reconstructive Japan Total $21,883,131 $51,334,177 India Burns $3,407,463 $42,593 Wounds $7,950,747 Reconstructive India Total $3,407,463 $7,993,341 Europe Burns $86,211,948 $1,077,649 Wounds $201,161,211 Reconstructive Europe Total $86,211,948 $202,238,861 Incremental Change in SG&A Expenses as a result of Licens 2009 2010 2011 2012 Patents & Legal $0 $5,000 $10,000 $10,000 Travel $0 $5,000 $10,000 $10,000 Total $0 $10,000 $20,000 $20,000 Value of South Korea Licensing Agr 2009 2010 2011 2012
- 24. Increase in Revenue $0 $0 $2,787,261 $6,538,449 Increase in Expenses $0 $10,000 $20,000 $20,000 Increase (Decrease) in Taxes $0 ($3,500) $968,541 $2,281,457 Increase in NI $0 ($6,500) $1,798,720 $4,236,992 Change in NWC $0 0 ($55,745) ($75,024) Change in FCFF $0 ($6,500) $1,742,974 $4,161,968 PV of FCFF $0 ($3,948) $850,563 $1,631,735 Value of South Korea Licensing Agreement $2,924,935 Value of China Licensing Agreem 2009 2010 2011 2012 Increase in Revenue $0 $0 $10,932,200 $25,645,118 Increase in Expenses $0 $10,000 $20,000 $20,000 Increase (Decrease) in Taxes $0 ($3,500) $3,819,270 $8,968,791 Increase in NI $0 ($6,500) $7,092,930 $16,656,327 Change in NWC $0 0 ($218,644) ($294,258) Change in FCFF $0 ($6,500) $6,874,286 $16,362,069 PV of FCFF $0 ($3,948) $3,354,619 $6,414,888 Value of China Licensing Agreement $11,450,226 Value of Japan Licensing Agreem 2009 2010 2011 2012 Increase in Revenue $0 $0 $21,883,131 $51,334,177 Increase in Expenses $0 $10,000 $20,000 $20,000 Increase (Decrease) in Taxes $0 ($3,500) $7,652,096 $17,959,962 Increase in NI $0 ($6,500) $14,211,035 $33,354,215 Change in NWC $0 0 ($437,663) ($589,021) Change in FCFF $0 ($6,500) $13,773,372 $32,765,194 PV of FCFF $0 ($3,948) $6,721,340 $12,845,874 Value of Japan Licensing Agreement $23,665,893 Value of India Licensing Agreem 2009 2010 2011 2012 Increase in Revenue $0 $0 $3,407,463 $7,993,341 Increase in Expenses $0 $10,000 $20,000 $20,000 Increase (Decrease) in Taxes $0 ($3,500) $1,185,612 $2,790,669 Increase in NI $0 ($6,500) $2,201,851 $5,182,671 Change in NWC $0 0 ($68,149) ($91,718) Change in FCFF $0 ($6,500) $2,133,702 $5,090,954 PV of FCFF $0 ($3,948) $1,041,236 $1,995,952 Value of India Licensing Agreement $3,495,956 Value of Europe Licensing Agreem 2009 2010 2011 2012 Increase in Revenue $0 $0 $86,211,948 $202,238,861 Increase in Expenses $0 $10,000 $20,000 $20,000 Increase (Decrease) in Taxes $0 ($3,500) $30,167,182 $70,776,601 Increase in NI $0 ($6,500) $56,024,766 $131,442,259 Change in NWC $0 0 ($1,724,239) ($2,320,538) Change in FCFF $0 ($6,500) $54,300,527 $129,121,721 PV of FCFF $0 ($3,948) $26,498,399 $50,623,271 Value of Europe Licensing Agreement $93,030,003 NI ($1,621,167) $7,069,833 $95,408,735 $178,549,227 PV of FCFF ($1,068,339) $3,920,918 $45,225,265 $68,738,837
- 25. NI difference $0 $0 $0 Revenues $58,500,000 $222,722,002 $411,649,946 COGS $36,955,000 $63,240,000 $88,845,000 SG&A $10,123,000 $12,148,000 $15,276,000 R&D $0 $0 $32,280,495 Taxes $0 $3,806,833 $51,373,934 $96,141,891 NWC ($10,000) ($1,160,000) ($3,284,440) ($3,778,559) FCFF ($1,358,500) $6,455,167 $92,675,628 $175,328,001 ($1,068,339) $3,920,918 $45,225,265 $68,738,837 $0 $0 $0 $0 ($1,068,339) $3,920,918 $45,225,265 $68,738,837
- 26. greements and Royalty Payments Calculations Market as % of Health $2,178,160,000,000 Expenditures $3,000,000,000 0.138% $7,000,000,000 0.321% $1,000,000,000 0.046% France Germany Italy Netherlands Spain 11.2% 10.7% 8.9% 9.2% 8.2% $2,987,000,000,000 $3,818,000,000,000 $2,399,000,000,000 $909,500,000,000 $1,683,000,000,000 $334,544,000,000 $408,526,000,000 $213,511,000,000 $83,674,000,000 $138,006,000,000 $460,770,559 $562,666,654 $294,070,684 $115,244,977 $190,076,946 $1,075,131,303 $1,312,888,860 $686,164,928 $268,904,947 $443,512,873 $122,718,559 $144,230,219 $75,086,048 $24,931,330 $45,238,313 Finland France Germany Italy Netherlands $21,090,000,000 $334,544,000,000 $408,526,000,000 $213,511,000,000 $83,674,000,000 $29,047,453 $460,770,559 $562,666,654 $294,070,684 $115,244,977 $67,777,390 $1,075,131,303 $1,312,888,860 $686,164,928 $268,904,947 $7,532,973 $122,718,559 $144,230,219 $75,086,048 $24,931,330 nal Sources of Revenue Hlookup Table Grants Reconstruction AFIRM 1 2 3 1 2 $- $- $- $500,000 $500,000 $- $- $- $1,000,000 $1,000,000 $- $- $- $- $7,500,000 $- $- $250,000,000 $- $- $30,406,107 $30,406,107 $25,000,000 $- $- $380,076 $380,076 $25,000,000 $- $- $760,153 $760,153 $25,000,000 $- $- $1,140,229 $1,140,229 $25,000,000 $- $- $1,520,305 $1,520,305 $25,000,000 $- $- $1,900,382 $1,900,382 $25,000,000 $- $- ting Figures Graft Market ($11,000,000,000)
- 27. 20% 1% venue Scenario 1 for Valuation Sheet in Print Doc 2013 2014 2015 2016 2017 $40,710,000 $44,781,000 $48,363,480 $51,265,289 $53,315,900 $10,160,872 $9,741,889 $9,936,727 $10,135,462 $10,338,171 $13,875,067 $13,999,228 $14,125,873 $14,155,050 $14,286,811 $0 $0 $0 $0 $0 $30,000 $24,000 $18,000 $12,000 $6,000 $533,333 $533,333 $533,333 $533,333 $533,333 $16,110,728 $20,482,549 $23,749,547 $26,429,444 $28,151,585 $5,638,755 $7,168,892 $8,312,341 $9,250,305 $9,853,055 $10,471,973 $13,313,657 $15,437,205 $17,179,138 $18,298,530 venue Scenario 2 for Valuation Sheet in Print Doc 2013 2014 2015 2016 2017 $187,898,966 $194,661,518 $207,279,158 $219,302,064 $229,074,847 $114,465,000 $139,265,500 $143,978,615 $148,424,441 $152,519,542 $20,897,750 $22,220,825 $23,385,131 $24,228,219 $24,894,668 $7,343,397 $5,539,602 $6,330,054 $7,087,762 $7,655,531 $30,000 $24,000 $18,000 $12,000 $6,000 $533,333 $533,333 $533,333 $533,333 $533,333 $44,629,486 $27,078,258 $33,034,024 $39,016,308 $43,465,774 $15,620,320 $9,477,390 $11,561,909 $13,655,708 $15,213,021 $29,009,166 $17,600,868 $21,472,116 $25,360,600 $28,252,753 th a 0% Chance of the Skin Graft Getting FDA Approval 2013 2014 2015 2016 2017 $40,710,000 $44,781,000 $48,363,480 $51,265,289 $53,315,900 $20,355,000 $22,390,500 $24,181,740 $25,632,644 $26,657,950 $20,897,750 $22,220,825 $23,385,131 $24,228,219 $24,894,668 $1,017,750 $1,119,525 $1,209,087 $1,281,632 $1,332,898 $563,333 $557,333 $551,333 $545,333 $539,333 ($2,123,833) ($1,507,183) ($963,811) ($422,540) ($108,948) $0 $0 $0 $0 $0 ($2,123,833) ($1,507,183) ($963,811) ($422,540) ($108,948) $563,333 $557,333 $551,333 $545,333 $539,333 ($106,200) ($81,420) ($71,650) ($58,036) ($41,012) ($1,666,700) ($1,031,270) ($484,128) $64,757 $389,373 ($524,981) ($271,894) ($106,839) $12,484 $65,577
- 28. US 100% Skin Graft $150,710,000 $182,281,000 $189,300,980 $195,726,226 $201,388,361 $114,465,000 $139,265,500 $143,978,615 $148,424,441 $152,519,542 $20,897,750 $22,220,825 $23,385,131 $24,228,219 $24,894,668 $7,343,397 $5,539,602 $6,330,054 $7,087,762 $7,655,531 $563,333 $557,333 $551,333 $545,333 $539,333 $7,440,520 $14,697,740 $15,055,846 $15,440,471 $15,779,288 $2,604,182 $5,144,209 $5,269,546 $5,404,165 $5,522,751 $4,836,338 $9,553,531 $9,786,300 $10,036,306 $10,256,537 $563,333 $557,333 $551,333 $545,333 $539,333 ($656,200) ($631,420) ($140,400) ($128,505) ($113,243) $4,743,471 $9,479,444 $10,197,234 $10,453,134 $10,682,628 $1,494,110 $2,499,253 $2,250,354 $2,015,224 $1,799,133 International Revenues $69,682 $104,522 $139,363 $174,204 $175,946 $81,295 $162,590 $243,885 $325,180 $406,476 $492,416 $6,155 $12,310 $18,466 $24,621 $643,393 $273,268 $395,559 $517,850 $607,042 $273,305 $409,957 $546,610 $683,262 $690,095 $318,856 $637,712 $956,567 $1,275,423 $1,594,279 $1,413,898 $17,674 $35,347 $53,021 $70,695 $2,006,059 $1,065,343 $1,538,525 $2,011,707 $2,355,069 $547,078 $820,617 $1,094,157 $1,367,696 $1,381,373 $638,258 $1,276,516 $1,914,774 $2,553,032 $3,191,290 $5,995,978 $74,950 $149,899 $224,849 $299,799 $7,181,314 $2,172,083 $3,158,830 $4,145,577 $4,872,461 $85,187 $127,780 $170,373 $212,966 $215,096 $99,384 $198,769 $298,153 $397,537 $496,922 $215,806 $2,698 $5,395 $8,093 $10,790 $400,377 $329,246 $473,921 $618,597 $722,808 $2,155,299 $3,232,948 $4,310,597 $5,388,247 $5,442,129 $2,514,515 $5,029,030 $7,543,545 $10,058,061 $12,572,576 $22,288,009 $278,600 $557,200 $835,800 $1,114,400 $26,957,823 $8,540,578 $12,411,343 $16,282,108 $19,129,105 &A Expenses as a result of Licensing Agreements (per Country) 2013 2014 2015 2016 2017 $10,000 $10,000 $10,000 $10,000 $10,000 $10,000 $10,000 $10,000 $10,000 $10,000 $20,000 $20,000 $20,000 $20,000 $20,000 ue of South Korea Licensing Agreement 2013 2014 2015 2016 2017
- 29. $643,393 $273,268 $395,559 $517,850 $607,042 $20,000 $20,000 $20,000 $20,000 $20,000 $218,187 $88,644 $131,446 $174,247 $205,465 $405,205 $164,624 $244,113 $323,602 $381,577 $117,901 $7,403 ($2,446) ($2,446) ($1,784) $523,106 $172,027 $241,667 $321,157 $379,794 $164,769 $45,355 $53,332 $61,915 $63,964 Value of China Licensing Agreement 2013 2014 2015 2016 2017 $2,006,059 $1,065,343 $1,538,525 $2,011,707 $2,355,069 $20,000 $20,000 $20,000 $20,000 $20,000 $695,121 $365,870 $531,484 $697,097 $817,274 $1,290,938 $679,473 $987,041 $1,294,610 $1,517,795 $472,781 $18,814 ($9,464) ($9,464) ($6,867) $1,763,719 $698,287 $977,578 $1,285,146 $1,510,928 $555,540 $184,103 $215,735 $247,759 $254,465 Value of Japan Licensing Agreement 2013 2014 2015 2016 2017 $7,181,314 $2,172,083 $3,158,830 $4,145,577 $4,872,461 $20,000 $20,000 $20,000 $20,000 $20,000 $2,506,460 $753,229 $1,098,590 $1,443,952 $1,698,361 $4,654,854 $1,398,854 $2,040,239 $2,681,625 $3,154,100 $883,057 $100,185 ($19,735) ($19,735) ($14,538) $5,537,911 $1,499,039 $2,020,505 $2,661,890 $3,139,562 $1,744,344 $395,221 $445,891 $513,177 $528,755 Value of India Licensing Agreement 2013 2014 2015 2016 2017 $400,377 $329,246 $473,921 $618,597 $722,808 $20,000 $20,000 $20,000 $20,000 $20,000 $133,132 $108,236 $158,872 $209,509 $245,983 $247,245 $201,010 $295,049 $389,088 $456,825 $151,859 $1,423 ($2,894) ($2,894) ($2,084) $399,104 $202,433 $292,155 $386,194 $454,741 $125,711 $53,371 $64,474 $74,453 $76,586 alue of Europe Licensing Agreement 2013 2014 2015 2016 2017 $26,957,823 $8,540,578 $12,411,343 $16,282,108 $19,129,105 $20,000 $20,000 $20,000 $20,000 $20,000 $9,428,238 $2,982,202 $4,336,970 $5,691,738 $6,688,187 $17,509,585 $5,538,376 $8,054,373 $10,570,370 $12,420,918 $3,505,621 $368,345 ($77,415) ($77,415) ($56,940) $21,015,206 $5,906,721 $7,976,958 $10,492,955 $12,363,979 $6,619,419 $1,557,305 $1,760,377 $2,022,901 $2,082,300 $28,944,166 $17,535,868 $21,407,116 $25,295,600 $28,187,753 $10,703,893 $4,734,609 $4,790,162 $4,935,428 $4,805,203
- 30. $0 $0 $0 $0 $0 $7,343,397 $15,585,320 $9,442,390 $11,526,909 $13,620,708 $15,178,021 $4,475,020 ($135,251) ($252,353) ($240,458) ($195,456) $33,982,519 $17,957,950 $21,706,096 $25,600,476 $28,531,631 $10,703,893 $4,734,609 $4,790,162 $4,935,428 $4,805,203 $0 $0 $0 $0 $0 $10,703,893 $4,734,609 $4,790,162 $4,935,428 $4,805,203
- 31. Sweden Switzerland UK 9.2% 11.4% 8.2% $512,900,000,000 $492,600,000,000 $2,787,000,000,000 $47,186,800,000 $56,156,400,000 $228,534,000,000 $64,990,818 $77,344,731 $314,762,001 $151,645,242 $180,471,040 $734,444,669 $17,699,166 $15,391,602 $91,385,901 Spain Sweden Switzerland UK $138,006,000,000 $47,186,800,000 $56,156,400,000 $228,534,000,000 $190,076,946 $64,990,818 $77,344,731 $314,762,001 $443,512,873 $151,645,242 $180,471,040 $734,444,669 $45,238,313 $17,699,166 $15,391,602 $91,385,901 Grants AFIRM Dept. of Defense 3 1 2 3 $500,000 $- $- $- $1,000,000 $5,000,000 $5,000,000 $5,000,000 $7,500,000 $- $5,000,000 $5,000,000 $5,000,000 $- $- $5,000,000 $5,000,000 $- $- $5,000,000 $- $- $- $- $- $- $- $- $- $- $- $- $- $- $- $- $- $- $- $-
- 32. 2018 $54,915,377 $10,544,934 $14,421,207 $0 $0 $533,333 $29,415,902 $10,295,566 $19,120,336 2018 $235,017,874 $156,465,820 $25,414,498 $7,855,205 $0 $533,333 $44,749,018 $15,662,156 $29,086,861 2018 $54,915,377 ***From 2010 Onward Assumes Lab Expenses and Employee Expenses for HUD are Done upon FDA FAILURE $27,457,689 $25,414,498 ***Dropped R&D To 5% if HUD NOT APPROVE $1,372,884 $533,333 $136,973 $47,941 $89,033 $533,333 ($31,990) $590,376 $86,861 Assumes Terminal Value is Zero, Update when you get SG&A numbers for this scenario and have a realistic 2018 FCFF
- 33. $206,689,650 $156,465,820 What is Difference Between SG&A for him if HUD Does NOT GET APPROVE??????? $25,414,498 $7,855,205 $533,333 $16,420,793 $5,747,278 $10,673,516 $533,333 ($106,026) $11,100,823 $1,633,235 $177,705 $410,540 $30,776 $619,022 $696,996 $1,610,222 $88,369 $2,395,587 $1,395,186 $3,223,203 $374,749 $4,993,138 $217,247 $501,891 $13,488 $732,626 $5,496,550 $12,698,301 $1,393,001 $19,587,853 2018 $10,000 $10,000 $20,000 2018
- 34. $619,022 $20,000 $209,658 $389,364 ($240) $389,124 $57,251 2018 $2,395,587 $20,000 $831,455 $1,544,131 ($810) $1,543,321 $227,065 2018 $4,993,138 $20,000 $1,740,598 $3,232,540 ($2,414) $3,230,126 $475,240 2018 $732,626 $20,000 $249,419 $463,207 ($196) $463,010 $68,122 2018 $19,587,853 $20,000 $6,848,748 $12,719,104 ($9,175) $12,709,929 $1,869,978 $29,021,861 $4,330,890
- 35. $0 $15,627,156 ($118,861) $29,436,334 $4,330,890 $0 $4,330,890
- 36. Other Potentially Useful Forecasting Statistics S. Korea China Japan India Denmark 0-14 Population 8,166,097 265,167,835 17,149,265 362,874,979 997,664 15-64 Population 35,093,893 964,906,976 81,721,301 741,248,288 3,616,764 65 + Population 5,248,982 108,538,157 28,208,113 61,955,950 886,082 Population Growth 0.266% 0.655% -0.191% 1.548% 0.280% Private Expenditure on Health as % of Total Expenditures on Health (2005) 47.0%² 61.2% 17.8% 81.0% 15.9% Public Expenditure ### ### ### ### ### Private Expenditure ### ### ### ### $5,347,742,400
- 37. one upon FDA FAILURE o and have a realistic 2018 FCFF
- 41. Finland France Germany Italy Netherlands Spain Sweden Switzerland 861,202 11,968,714 11,238,882 7,870,226 2,902,872 5,864,419 740,518 1,188,171 3,506,287 41,892,404 54,384,520 38,511,780 11,324,401 27,306,506 5,930,396 5,176,918 882,786 10,559,015 16,706,356 11,744,206 1,525,726 7,354,077 1,703,464 1,239,378 0.098% 0.549% -0.053% -0.047% 0.412% 0.072% 0.158% 0.276% 22.2% 20.1% 23.1% 23.4% 35.1% 28.6% 18.3% 40.3% ### ### ### ### ### ### ### ### $4,681,980,000 ### ### ### ### ### ### ###
- 46. UK 10,219,887 41,020,711 9,872,607 0.279% 12.9% $199,053,114,000 $29,480,886,000
- 51. Income Statement 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 Net Sales $500,000 $58,500,000 $222,722,002 $411,649,946 $187,898,966 $194,661,518 $207,279,158 $219,302,064 $229,074,847 $235,017,874 % Growth NA 11600.0% 280.7% 84.8% -54.4% 3.6% 6.5% 5.8% 4.5% 2.6% Cost of Goods Sold $530,000 $36,955,000 $63,240,000 $88,845,000 $114,465,000 $139,265,500 $143,978,615 $148,424,441 $152,519,542 $156,465,820 Gross Profit ($30,000) $21,545,000 $159,482,002 $322,804,946 $73,433,966 $55,396,018 $63,300,543 $70,877,623 $76,555,306 $78,552,054 Gross Profit Margin -6.0% 36.8% 71.6% 78.4% 39.1% 28.5% 30.5% 32.3% 33.4% 33.4% SG&A $1,318,500 $10,123,000 $12,148,000 $15,276,000 $20,997,750 $22,320,825 $23,485,131 $24,328,219 $24,994,668 $25,514,498 SG&A (% of Sales) 263.7% 17.3% 5.5% 3.7% 11.2% 11.5% 11.3% 11.1% 10.9% 10.9% R&D $0 $0 $0 $32,280,495 $7,343,397 $5,539,602 $6,330,054 $7,087,762 $7,655,531 $7,855,205 R&D (% of Sales) 0.0% 0.0% 0.0% 7.8% 3.9% 2.8% 3.1% 3.2% 3.3% 3.3% EBITDA ($1,348,500) $11,422,000 $147,334,002 $275,248,452 $45,092,819 $27,535,591 $33,485,358 $39,461,642 $43,905,107 $45,182,351 EBITDA Margin -269.7% 19.5% 66.2% 66.9% 24.0% 14.1% 16.2% 18.0% 19.2% 19.2% Depreciation $6,000 $12,000 $18,000 $24,000 $30,000 $24,000 $18,000 $12,000 $6,000 $0 Amortization $266,667 $533,333 $533,333 $533,333 $533,333 $533,333 $533,333 $533,333 $533,333 $533,333 Pretax Income ($1,621,167) $10,876,667 $146,782,669 $274,691,119 $44,529,486 $26,978,258 $32,934,024 $38,916,308 $43,365,774 $44,649,018 Pretax Margin -324.2% 18.6% 65.9% 66.7% 23.7% 13.9% 15.9% 17.7% 18.9% 19.0% Income Taxes $0 $3,806,833 $51,373,934 $96,141,891 $15,585,320 $9,442,390 $11,526,909 $13,620,708 $15,178,021 $15,627,156 Effective Tax Rate 35.0% 35.0% 35.0% 35.0% 35.0% 35.0% 35.0% 35.0% 35.0% 35.0% Net Income to Common ($1,621,167) $7,069,833 $95,408,735 $178,549,227 $28,944,166 $17,535,868 $21,407,116 $25,295,600 $28,187,753 $29,021,861 Net Income Margin -324.2% 12.1% 42.8% 43.4% 15.4% 9.0% 10.3% 11.5% 12.3% 12.3%
- 52. Cash F Cash Flow from Operating Activities: Net Income Plus: Depreciation Amortization Funds Flow from Operations (Increase) / Decrease in Accounts Receivable (Increase) / Decrease in Other Current Assets (Increase) / Decrease in Current Operating Assets Increase / (Decrease) in Accounts Payable Increase / (Decrease) in Other Current Liabilities Increase / (Decrease) in Current Operating Liabilities (Increase) Decrease in Operating Working Capital (Increase) / Decrease in Other Long-Term Assets Increase / (Decrease) in Other Long-Term Liabilities Cash Flow from Operations Cash Flow from Investing Activities: Less: Capital Expenditures Cash Flow from Investing OPERATING CASH FLOW AFTER INVESTING ACTIVITIES Cash Flow from Financing Activities: Cash Flow from Anticipated Debt and Equity Issuance Anticipated Issuance of New Common Equity Issuance Total Anticipated Debt and Equity Issuance Total Change in Cash Beginning Cash Balance Total Change in Cash Ending Cash Balance