Ethionamide: A 2nd Line Anti-TB Agent
•Descargar como PPT, PDF•
15 recomendaciones•7,351 vistas
This document discusses the anti-tuberculosis drug ethionamide. It is a second-line drug that is an analogue of isonicotinamide but contains sulfur instead of oxygen. It is less active in vitro but more active in vivo due to increased lipophilicity from its ethyl group. Its mechanism of action involves conversion to its active metabolite ethionamide sulfoxide via oxidation, which then inactivates the inhA enoyl reductase enzyme. Less than 1% is excreted unchanged in urine with the rest excreted as metabolites.
Denunciar
Compartir
Denunciar
Compartir
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...

Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...
MEDICINAL CHEMISTRY OF ANTI-TUBERCULAR AGENTS.pptx

MEDICINAL CHEMISTRY OF ANTI-TUBERCULAR AGENTS.pptx
Aminoglycosides(medicinal chemistry by p.ravisankar)

Aminoglycosides(medicinal chemistry by p.ravisankar)
antiviral drugs medicinal chemistry by padala varaprasad

antiviral drugs medicinal chemistry by padala varaprasad
Chloramphenicol: Unclassified Antibiotics-Shahare HV

Chloramphenicol: Unclassified Antibiotics-Shahare HV
Destacado
Destacado (20)
TB, historical perspective of anti TB drugs and medicinal chemistry of INH

TB, historical perspective of anti TB drugs and medicinal chemistry of INH
Emerging Paradigms in International Management Education

Emerging Paradigms in International Management Education
How would we recognise a truly sustainable enterprise if we saw one?

How would we recognise a truly sustainable enterprise if we saw one?
Digital Brand, corso di formazione sui social network

Digital Brand, corso di formazione sui social network
Similar a Ethionamide: A 2nd Line Anti-TB Agent
Similar a Ethionamide: A 2nd Line Anti-TB Agent (20)
General Reactions involved in amino acid metabolism

General Reactions involved in amino acid metabolism
Más de Khalid Hussain
Más de Khalid Hussain (20)
Último
Último (20)
Transcript: #StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024

Transcript: #StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024
Swan(sea) Song – personal research during my six years at Swansea ... and bey...

Swan(sea) Song – personal research during my six years at Swansea ... and bey...
From Event to Action: Accelerate Your Decision Making with Real-Time Automation

From Event to Action: Accelerate Your Decision Making with Real-Time Automation
Tech-Forward - Achieving Business Readiness For Copilot in Microsoft 365

Tech-Forward - Achieving Business Readiness For Copilot in Microsoft 365
IAC 2024 - IA Fast Track to Search Focused AI Solutions

IAC 2024 - IA Fast Track to Search Focused AI Solutions
Handwritten Text Recognition for manuscripts and early printed texts

Handwritten Text Recognition for manuscripts and early printed texts
Azure Monitor & Application Insight to monitor Infrastructure & Application

Azure Monitor & Application Insight to monitor Infrastructure & Application
Neo4j - How KGs are shaping the future of Generative AI at AWS Summit London ...

Neo4j - How KGs are shaping the future of Generative AI at AWS Summit London ...
#StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024

#StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024
Transforming Data Streams with Kafka Connect: An Introduction to Single Messa...

Transforming Data Streams with Kafka Connect: An Introduction to Single Messa...
Injustice - Developers Among Us (SciFiDevCon 2024)

Injustice - Developers Among Us (SciFiDevCon 2024)
[2024]Digital Global Overview Report 2024 Meltwater.pdf![[2024]Digital Global Overview Report 2024 Meltwater.pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![[2024]Digital Global Overview Report 2024 Meltwater.pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
[2024]Digital Global Overview Report 2024 Meltwater.pdf
Kotlin Multiplatform & Compose Multiplatform - Starter kit for pragmatics

Kotlin Multiplatform & Compose Multiplatform - Starter kit for pragmatics
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
Ethionamide: A 2nd Line Anti-TB Agent
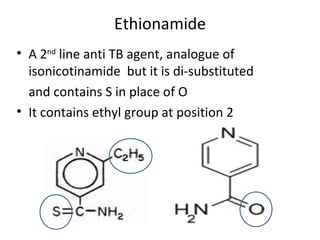
- 1. Ethionamide • A 2nd line anti TB agent, analogue of isonicotinamide but it is di-substituted and contains S in place of O • It contains ethyl group at position 2
- 2. • In vitro it is less active but in vivo more active because of increased lipocity due to C2H5 • Mechanism of action is similar to INH • Its active metabolite is ethionamide sulfoxide
- 3. Mechanism of action • Ethionamide upon oxidation with catalase- peroxidase is converted to an active acylating agent, ethionamide sulfoxide, which inturn inactivate inhA enoyal reductase. It acylates cystine No. 243 in inhA protein
- 4. Mechanism of action Ethionamide sulfoxide Ethionamide
- 5. Metabolism • Less than 1% of the drug is excreted unchanged in urine. Rest of the drug is excreted as one of the following metabolites, which are given as follows:
- 6. Cycloserine • Analogue of amino acid serine and it exists in cyclic form- a five member ring containing O and N at an adjacent positions, • Also called Isoxazolidine or oxazolidine • Obtained naturally as d-isomer • Contains Keto group at position 3 and NH2 at position 4, which is in front • d-isomer is more active
- 7. • It is 2nd line anti TB drug first isolated from Streptomyces orchidaceus, but now being synthesized in laboratory • It causes CNS toxicity • Bacteria become resistant after sometime • It acts on cell wall of bacteria and is not selective against MT because all bacteria contain peptidoglycan
- 8. • It acts on normal peptidoglycan portion of cell wall rather than acting on outer layer of mycolic acid • It inhibits alanine resemase and alanine ligase • Alanine resemase converts L-isomer of alanine to d-isomer. Because only d-form can be incorporated into cell wall. Alanine is present in levo form, hence need to be converted to d- form • Lygase is necessary for attachment of two alanine units
- 10. Synthesis
- 11. • Readily absorbed after oral administration and is widely distributed including CNS • It binds to neuronal N-methyl, D-aspaartate receptor and effects the synthesis and metabolism of aminobutyric acid leading to serious CNS effects
