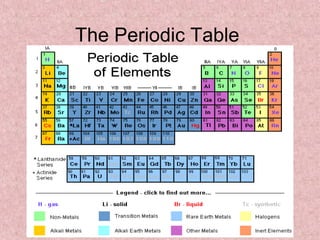
The periodic table
- 2. • Dmitri Mendeleev: Russian, who discovered a pattern to the elements by using a variety of properties of the elements. Predicted elements that had not been discovered yet.
- 3. • Periodic: describes something that occurs or repeats at regular intervals (Days of a week) • Periodic Law: the law that states that the repeating chemical and physical properties of elements change periodically with the atomic numbers of the elements (Henry Moseley)
- 4. Categories of Elements: 1. Metals: are shiny and they conduct heat energy and electric current. (Ductile, Malleable, Solid at Room Temp.) • Example: Copper, Lead, Tin
- 5. Categories of Elements: 2. Nonmetals: do not conduct heat or electric current, and are dull in appearance. (Gases at Room Temp., Opposite Metal) • Example: Neon, Iodine, Sulfur
- 6. Categories of Elements: 3. Metalloids: have properties of both metals and nonmetals (Zig zag Line) • Example: Boron, Silicon, Antimony
- 7. Parts of the Periodic Table: • Period: each horizontal row of elements on the periodic table (Reactivity and Conductivity) • Group: each vertical column of elements on the periodic table (Similar Physical and Chemical Properties)
- 8. Each element has its own: 1. Element Symbol 2. Element Name 3. Atomic Number 4.Atomic Mass
- 9. Isotopes: atoms with same number of protons but not neutrons • How do Atoms of Different Elements Differ? Neutrons • Radioactive atoms spontaneously fall apart after a certain amount of time. • How do you tell isotopes apart? Mass Number • Naming an Isotope: Name of element with hyphened mass number (Carbon-12)
- 10. Groups of Elements on the Periodic Table • Group 1: Alkali Metals Group contains: Metals Electrons in outer level: 1 Reactivity: Very Reactive Shared properties: Softness, Color of silver, shiny, low density
- 11. Groups of Elements on the Periodic Table Group 2: Alkaline-Earth Metals Group contains: Metals Electrons in outer level: 2 Reactivity: Very but less than group 1 Shared properties: Color of silver, higher density than group 1
- 12. Groups of Elements on the Periodic Table Group 3-12: Transition Metals Group contains: Metals Electrons in outer level: 1 or 2 Reactivity: Less reactive than group 2 Shared properties: shiny, good conductors, higher melting point and density over group 1 & 2
- 13. Groups of Elements on the Periodic Table Group 13: Boron Group Group contains: 1 metalloid and 4 metals Electrons in outer level: 3 Reactivity: Reactive Shared properties: Solid at room temp.
- 15. Groups of Elements on the Periodic Table Group 14: Carbon Group Group contains: 1 nonmetal, 2 metalloids, and 2 metals Electrons in outer level: 4 Reactivity: Varies Shared properties: Solid at room temp.
- 17. Groups of Elements on the Periodic Table Group 15: Nitrogen Group Group contains: 2 nonmetals, 2 metalloids, and 1 metal Electrons in outer level: 5 Reactivity: Varies Shared properties: Solid at room temp. except Nitrogen
- 18. Groups of Elements on the Periodic Table Group 16: Oxygen Group Group contains: 3 nonmetals, 1 metalloid, and 1 metal Electrons in outer level: 6 Reactivity: Reactive Shared properties: all except Oxygen solid at room temp.
- 20. Groups of Elements on the Periodic Table Group 17: Halogens Group contains: Nonmetals Electrons in outer level: 7 Reactivity: Reactive Shared properties: Poor Conductors, Violent reactions with alkali metals, never uncombined in nature
- 21. Groups of Elements on the Periodic Table Group 18: Noble Gases Group contains: Nonmetals Electrons in outer level: 8 Reactivity: Unreactive Shared properties: Colorless, Odorless, Gases
- 22. Hydrogen Electrons in outer level: 1 Reactivity: reactive Shared properties: Colorless, Odorless, Gases, low density, explosive with oxygen