Antiknock characteristics of vehicle fuel
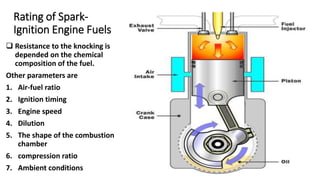
- 1. Rating of Spark- Ignition Engine Fuels Resistance to the knocking is depended on the chemical composition of the fuel. Other parameters are 1. Air-fuel ratio 2. Ignition timing 3. Engine speed 4. Dilution 5. The shape of the combustion chamber 6. compression ratio 7. Ambient conditions
- 2. Antiknock characteristics Two reference fuels for antiknock characteristics. High octane number, and less octane number. Example Iso-octane (C8H18) is rich in the octane. It is around 100 as the octane number Normal Heptane (C7H16) has 0 as the octane number
- 3. Octane number A= is the amount of Tetra- ethyl lead in ml/gal of fuel Is defined as the percentage by the volume of the iso-octane in the mixture of iso-Octane and normal heptane. Tetra ethyl lead to the iso-octane will result in the fuel with a greater antiknock property. Small change in the octane number brings grater antiknock property. Ex. ON increases from 92 to 93 result in the greater antiknock property than the ON increasing from the 30 to 31. Octane number above 100 and relatively performance of the engine computed by
- 4. CALORIFIC VALUE . "The total quantity of heat liberated, when a unit mass (or volume) of the fuel is burnt completely.“ Calorie' is the amount of heat required to raise the temperature of one gram of water through one degree centigrade (15-16°c). "Kilocalorie" is equal to 1,000 calories. It may be defined as 'the quantity of heat required to raise the temperature of one kilogram of water through one degree centigrade. 1 kcal = 1,000 calorie.
- 5. HCV and LCV • .. Gross or higher calorific value (HCV) is "the total amount of heat produced, when unit mass/volume of the fuel has been burnt completely and the products of combustion have been cooled to room temperature"(i.E., 15°C or 60°F ). Net or lower calorific value (LCV) is "the net heat produced, when unit mass /volume of the fuel is burnt completely and the products are permitted to escape". Net calorific value= gross calorific value - latent heat of condensation of water vapour produced = gcv - mass of hydrogen per unit weight of the fuel burnt x 9 x latent heat of condensation of water vapour.
- 6. Theoretical calculation of calorific value using Dulongs formula . HCV =1/100[8,080 C + 34,500 (H – O/8)+ 2,240 S] kcal/kg LCV = [ HCV - 9H/100 x 587] kcal/kg = [HCV - 0.09 H x 587] kcal/kg Flame Temperature The maximum temperature to which an object can be heated by the flame. Temperature of flame mainly depends upon calorific value and other properties. Theoretical flame temperature = (heat of combustion+ sensible heat in fuel and air) Total quantity of the combustion product)X (their mean specific heats)
- 7. Bomb Calorimeter of solid and liquid fuel . If we equate the heat given out by the fuel to the heat taken up by the calorimeter and the water, the calorific value of a fuel can be determined.
- 8. Construction and working principle of Bomb calorimeter . It consist of a stainless steel bomb in which combustion of fuel is made to take place. A known mass of the given fuel is taken inside the steel bomb which is connected with two electrodes. The bomb lid is tightly screwed and filled with O2 up to 25 atm. The water is stirred with the help of mechanical stirrer and the initial temp is recorded. The electrodes are then connected to 6 volt battery and the circuit is completed. Uniform stirring of water is continued and the maximum temp is recorded.
- 9. Calculation of Bomb Calorimeter . Heat liberated by the fuel = heat taken up by the calorimeter X * C = (W +w) (t2 - t1 ) C= (W +w) (t2 - t1 )/ X H.C. V = (W +w) (t2 - t1 )/ X X= mass in gm of the fuel sample W= mass of water in calorimeter w= water equivalent of calorimeter, stirrer, thermometer, bomb etc in gm t1= initial temp of water in oc t2= final temp of water in oc C= calorific value of the fuel
- 10. Boy’s Calorimeter for determination of calorific value of gases & volatile Liquid Fuels .
- 11. Description of Boy’s or Junkers . Description of the apparatus 1. Bunsen Burner: special type of Burner clamped at the bottom & pushed up in chamber during the carrying out combustion. 2. Gasometer: to measure the volume of gas burning per unit time and also pressure and temperature of the gas before burning can be read. 3. Pressure governor: It can control the supply of quantity of gas at give pressure. 4. Gas Calorimeter/ Combustion chamber: It is a vertical cylinder, which is surrounded by annular space for heating water and interchange coils. The entire is covered by an outer jacket in order to reduce the heat loss by radiation and convection.
- 12. Procedure of Determination • Gaseous fuel burnt (V) • At given T and p in period of time (t). • Quantity of water (w kg) passing • Rise in temperature (T2 -T1 ) • Steam condensed (in kg) Install the equipment on a flat rigid platform near an uninterrupted continuous water source of ½” size and a drain pipe. Connect the gas source to the pressure regulator, gas flow meter and the burner respectively in series. Insert the thermometer to measure water inlet and outlet temperatures. Start the water flow through the calorimeter at a study constant flow rate. Start the gas flow slowly and light the burner out side the calorimeter. Then the reading are taken simultaneously.
- 13. Calculation . • Volume of gas burn at STP in certain time (t)= V • Mass of the cooling water used in time t = W • Temperature of inlet water = T1, • Temperature of outlet water = T2 • Mass of steam condensed in time t in graduated cylinder = m • Higher calorific value of fuel = L , • Specific heat of water = S • Heat absorbed by circulating water = W(T2 -T1 )×specific heat of water (s) heat produced by combustion of fuel = VL
- 14. Ultimate analysis . Carbon and hydrogen About 1-2 g of accurately weighed coal sample is burnt in a current of oxygen in a combustion apparatus. C and H of the coal are converted into CO2 and H2O respectively. The gaseous products of combustion are absorbed respectively in KOH and CaCl2 tubes of known weights. The increase in weights of these are then determined.