1 6 How Are Solutes Dissolved
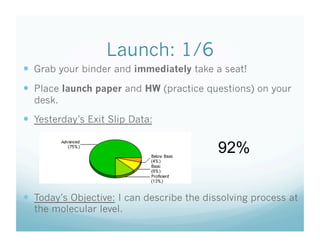
- 1. Launch: 1/6 Grab your binder and immediately take a seat! Place launch paper and HW (practice questions) on your desk. Yesterday’s Exit Slip Data: 92% Today’s Objective: I can describe the dissolving process at the molecular level.
- 2. Launch: 1/6 Grab your binder and immediately take a seat! Place launch paper and HW (practice questions) on your desk. Yesterday’s Exit Slip Data: 84% Today’s Objective: I can describe the dissolving process at the molecular level.
- 3. Launch: 1/6 Grab your binder and immediately take a seat! Place launch paper and HW (practice questions) on your desk. Yesterday’s Exit Slip Data: 88% Today’s Objective: I can describe the dissolving process at the molecular level.
- 4. Launch: 1/6 Grab your binder and immediately take a seat! Place launch paper and HW (practice questions) on your desk. Yesterday’s Exit Slip Data: - Today’s Objective: I can describe the dissolving process at the molecular level.
- 5. Launch 1/6 1. In 1-2 sentences, define a solute and a solvent.
- 6. Launch 1/6 2. Which of the following statements about solutions is correct? a. In a solution, the solute is dissolved into the solvent. b. In a solution, the solvent is dissolved into the solute. c. In a solution, the solvents are usually a solid. d. In a solution, the solutes and solvents are unequally mixed.
- 7. Launch 1/6 3. A teaspoon of dry coffee crystals dissolves when mixed in a cup of hot water. This process produces a coffee solution. The original crystals are classified as a a. solute. b. solvent. c. reactant. d. product.
- 8. New Changes for the New Year! Late homework policy Late homework in now due the day after you return Notes packets, handouts, and practice questions will can be found: at the back of the classroom
- 9. New Changes for the New Year! Class consequences 1. Name on board 2. Take a break 3. Call home 4. Sent to Mr. Wandera Rules: still the same 7
- 10. Monday January Calendar LPS Chem, Assessments, Unit Plan Tuesday Wednesday Thursday Jan 2010 (Pacific Time) Friday 28 29 30 31 1 4 5 6 7 8 Solutions 11 12 13 14 15 Solutions Acids and Bases Quiz 18 19 20 21 22 MLK No School Acids and Bases End of Semester Quiz 25 26 27 28 29 Acids and Bases Unit #6 Exam
- 11. How are solutes dissolved? Mr. Heffner 1/6/10
- 12. Review: What is a solution? A solution is… a homogeneous mixture made up of a solute and a solvent solution = solute + solvent • a solid • a liquid • is dissolved • does the dissolving • molecules don’t move • random movement
- 13. How are solutes dissolved? Solvent molecules randomly hit the solute and attack! Example: Solute = table salt (NaCl) Held together by ionic bonds Solvent = water Polar solvent: unequal sharing – +
- 14. How are solutes dissolved? Attack! http://www.mhhe.com/physsci/chemistry/ essentialchemistry/flash/molvie1.swf Movie questions: 1. What is the solute? 2. What is the solvent? 3. In your own words, describe how the salt was dissolved?
- 15. How are solutes dissolved? water molecules attack solute overcome the attractive forces of the solid dissolve the ion
- 16. Activity In 3-4 sentences, describe what we just acted out. Who were the solutes and what were the doing? Who were the solvents and what did they do?
- 17. Practice Questions Practice question worksheet
- 18. Exit Slip 1. In a solution, a. the solvent particles are attacked by the solute. b. the solvents are dissolved by the solutes. c. the two substances are not evenly mixed throughout. d. the solutes are dissolved by the solvents.
- 19. Exit Slip 2. Which of the following is correct statement. a. In a solution, the solute molecules randomly move around while the solvent molecules don’t move. b. In a solution, the solvent molecules randomly move around while the solute molecules don’t move. c. A solution is an example of a heterogeneous mixture. d. Solvents are usually solids
- 20. Exit Slip 3. Which of the following is a correct statement about the dissolving process? a. The solvent molecules randomly bump into the solutes and attack. b. In a solution, the solvent is dissolved into the solute. c. In a solution, the solvents are usually a solid. d. In a solution, the solutes and solvents are unequally mixed.
- 21. Exit Slip 4. An ionic salt (KBr) is dissolved into water. In this example, a. both KBr and water are solvents. b. KBr (the solvent) is attacked by water (the solute), splitting it into K+ and Br– ions. c. KBr (the solute) is attacked by water (the solvent), splitting it into K+ and Br– ions. d. the KBr attacks the water and dissolves it into solution.
- 22. Exit Slip 5. If the attractive forces among solid particles are less than the attractive forces between the solid and the liquid, the solid will a. probably form a new precipitate as its crystal lattice is broken and reformed. b. be unaffected because the attractive forces within the crystal lattice are too strong for dissolution to occur. c. begin the process of melting to form a liquid. d. dissolve as particles are pulled away from the crystal lattice by the water molecules
- 23. Homework Finish practice questions