Mau va bach huyet (anh) 1
•Descargar como PPT, PDF•
0 recomendaciones•444 vistas
Denunciar
Compartir
Denunciar
Compartir
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
Jason Godo biology 120 chapter 2 presentation hematic system

Jason Godo biology 120 chapter 2 presentation hematic system
Destacado
Destacado (20)
How Semantic Web ideas connect to e-portfolio interoperability

How Semantic Web ideas connect to e-portfolio interoperability
Más de Pham Ngoc Quang
Más de Pham Ngoc Quang (20)
Último
Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service AvailableGENUINE ESCORT AGENCY
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...GENUINE ESCORT AGENCY
Último (20)
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Mumbai ] (Call Girls) in Mumbai 10k @ I'm VIP Independent Escorts Girls 98333...
Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available

Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available
Low Rate Call Girls Bangalore {7304373326} ❤️VVIP NISHA Call Girls in Bangalo...

Low Rate Call Girls Bangalore {7304373326} ❤️VVIP NISHA Call Girls in Bangalo...
Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available
Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available
Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426

Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426
Call Girls in Lucknow Just Call 👉👉7877925207 Top Class Call Girl Service Avai...

Call Girls in Lucknow Just Call 👉👉7877925207 Top Class Call Girl Service Avai...
Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time

Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time
Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...

Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...
Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available
Coimbatore Call Girls in Thudiyalur : 7427069034 High Profile Model Escorts |...

Coimbatore Call Girls in Thudiyalur : 7427069034 High Profile Model Escorts |...
💕SONAM KUMAR💕Premium Call Girls Jaipur ↘️9257276172 ↙️One Night Stand With Lo...

💕SONAM KUMAR💕Premium Call Girls Jaipur ↘️9257276172 ↙️One Night Stand With Lo...
Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available
8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad

8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad
Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...

Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...

Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...
Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...

Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...
Mau va bach huyet (anh) 1
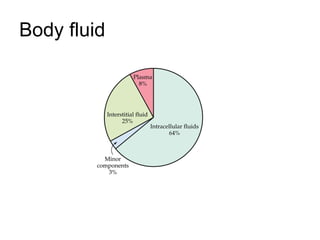
- 1. Body fluid
- 2. Hematocrit
- 3. Hematocrit
- 6. Hemoglobin
- 7. Life cycle of a RBC
- 8. Leukopoiesis
- 10. Step 2: Platelet plug
- 11. Extrinsic and Intrinsic coagulation pathways
- 16. Colorized SEM of Red Blood Cells (red), platelets (green) and White Blood Cells (purple)
- 18. TEM of Red Blood Cells (in cross section)
- 19. Sickle Cell Anemia (arrow = sickle cell)
- 20. Colorized SEM of Sickle Cell RBCs (green)
- 21. Normal RBC Sickled RBC
- 43. Platelet by TEM
Notas del editor
- Hemoglobin made up of globin that has 4 polypeptide chains 2 alpha and 2 beta, all of which are bound to the ring shaped heme Each hemoglobin molecule binds 4 O2 1 for each heme, 280 million hemoglobin molecules in a RBC—over a billion molecules of O2 can be bound by a single RBC Keeping hemoglobin in in erythrocytes rather than in plasma prevents it from breaking up and diffusing out of capillaries and it keeps hemoglobin from having effects on blood viscosity and osmotic pressure. Oxygen bound to hemoglobin forms the complex oxyhemoglobin, changes conformation and is a bright red color. When oxygen is released the hemoglobin becomes deoxyhemoglobin and changes shape again, becoming a darker red. Iron-2 reaction reverses as blood flows through capillaries CO2 binds with AAs in the globin part of hemoglobin—this is carbaminohemoglobin NO also binds to hemoglobin and hemoglobin can release NO to cause vasodilation
