Conceptual Framework for a Clinical Supply Chain solution stack
•
2 recomendaciones•120 vistas
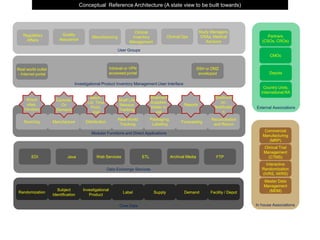
This document outlines the key components and functions of a clinical trial management system, including modules for data exchange, core data management, investigational product management, randomization, supply management, and tracking subjects and products throughout the clinical trial process from manufacturing to distribution to patient use. It presents a conceptual reference architecture showing how different sites, vendors, and user groups would interact with the system's various modules and applications.
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Recomendados
Recomendados
Más contenido relacionado
Similar a Conceptual Framework for a Clinical Supply Chain solution stack
Similar a Conceptual Framework for a Clinical Supply Chain solution stack (20)
PharmaLedger: A Digital Trust Ecosystem for Healthcare

PharmaLedger: A Digital Trust Ecosystem for Healthcare
Evaluación de riesgos asociados al puesto de trabajo: empleados, externos, vi...

Evaluación de riesgos asociados al puesto de trabajo: empleados, externos, vi...
Approach to enable your IT systems for FHIR (HL7 standards) compliance

Approach to enable your IT systems for FHIR (HL7 standards) compliance
Bridging Health Care and Clinical Trial Data through Technology

Bridging Health Care and Clinical Trial Data through Technology
Machine Learning with Apache Kafka in Pharma and Life Sciences

Machine Learning with Apache Kafka in Pharma and Life Sciences
Más de Sandeep Bhat
Más de Sandeep Bhat (12)
Observations, Issues, Benefits: Mobility in Life Sciences

Observations, Issues, Benefits: Mobility in Life Sciences
Strategy and Approach for Mobility in Life Sciences

Strategy and Approach for Mobility in Life Sciences
A Vision for Personalized Medicine: Collaboration, Enablement, Technology

A Vision for Personalized Medicine: Collaboration, Enablement, Technology
An industry view - integrated clinical analytics and reporting environment [2...

An industry view - integrated clinical analytics and reporting environment [2...
Engage Patients with Innovative Global Digital Patient Platform

Engage Patients with Innovative Global Digital Patient Platform
Nine Dimensions Of Commercial Excellence (Sandeep Bhat)

Nine Dimensions Of Commercial Excellence (Sandeep Bhat)
Social Media Considerations In Pharmacovigilance Visiongain 20110317 (Sande...

Social Media Considerations In Pharmacovigilance Visiongain 20110317 (Sande...
Último
Último (20)
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...

Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...
Six Myths about Ontologies: The Basics of Formal Ontology

Six Myths about Ontologies: The Basics of Formal Ontology
Navigating the Deluge_ Dubai Floods and the Resilience of Dubai International...

Navigating the Deluge_ Dubai Floods and the Resilience of Dubai International...
Introduction to Multilingual Retrieval Augmented Generation (RAG)

Introduction to Multilingual Retrieval Augmented Generation (RAG)
WSO2's API Vision: Unifying Control, Empowering Developers

WSO2's API Vision: Unifying Control, Empowering Developers
Apidays New York 2024 - APIs in 2030: The Risk of Technological Sleepwalk by ...

Apidays New York 2024 - APIs in 2030: The Risk of Technological Sleepwalk by ...
DEV meet-up UiPath Document Understanding May 7 2024 Amsterdam

DEV meet-up UiPath Document Understanding May 7 2024 Amsterdam
ICT role in 21st century education and its challenges

ICT role in 21st century education and its challenges
CNIC Information System with Pakdata Cf In Pakistan

CNIC Information System with Pakdata Cf In Pakistan
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Apidays New York 2024 - Scaling API-first by Ian Reasor and Radu Cotescu, Adobe

Apidays New York 2024 - Scaling API-first by Ian Reasor and Radu Cotescu, Adobe
ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke

ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke
Elevate Developer Efficiency & build GenAI Application with Amazon Q

Elevate Developer Efficiency & build GenAI Application with Amazon Q
Why Teams call analytics are critical to your entire business

Why Teams call analytics are critical to your entire business
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
Conceptual Framework for a Clinical Supply Chain solution stack
- 1. External Associations In house Associations Data Exchange Services Core Data EDI Java Web Services ETL Archival Media FTP Randomization Supply Subject Identification Master Data Management (MDM)Investigational Product DemandLabel Modular Functions and Direct Applications Sourcing Manufacture Distribution Real World Tracking Packaging, Labeling Forecasting Reconciliation and Return Conceptual Reference Architecture (A state view to be built towards) Which sites, Vendors Controls, On Demand Batch, Lot, Time Point, Visit Shelf Life Reissue Tracking Shipment Supplies, blister or vial Reports Destructi on, Notificatio n Interactive Randomization (IVRS, IWRS) Clinical Trial Management (CTMS) Commercial Manufacturing (MRP) Facility / Depot Investigational Product Inventory Management User Interface Real world outlet - Internet portal SSH or DMZ enveloped Intranet or VPN accessed portal User Groups Regulatory Affairs Quality Assurance Clinical Inventory Management Manufacturing Clinical Ops Study Managers, CRAs, Medical Advisors Partners (CSOs, CROs) CMOs Depots Country Units, International RA