Denunciar
Compartir
Recomendados
Recomendados
final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...

final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...nasrollah najibi ilkhchy
Nanoparticles of the ZnO and TiO2 were synthesized and the physicochemical properties of the compounds were characterized by IR, X-ray diffraction (XRD), scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The XRD patterns of the ZnO and TiO2 nanoparticles could be indexed to hexagonal and rutile phase, respectively. Aggregated nanoparticles of ZnO and TiO2 with spherical-like shapes were observed with particle diameter in the range of 80-100 nm. These nanoparticles were used for photocatalytic degradation of various dyes, Rhodamine B (RhB), Methylene blue (MB) and Acridine orange (AO) under solar light irradiation at room temperature. Effect of the amount of catalyst on the rate of photodegradation was investigated. In general, because ZnO is unstable, due to incongruous dissolution to yield Zn(OH)2 on the ZnO particle surfaces and thus leading to catalyst inactivation,the catalytic activity of the system for photodegradation of dyes decreased dramatically when TiO2 was replaced by ZnO.Photocatalytic degradation of some organic dyes under solar light irradiation...

Photocatalytic degradation of some organic dyes under solar light irradiation...Iranian Chemical Society
Más contenido relacionado
La actualidad más candente
final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...

final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...nasrollah najibi ilkhchy
Nanoparticles of the ZnO and TiO2 were synthesized and the physicochemical properties of the compounds were characterized by IR, X-ray diffraction (XRD), scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The XRD patterns of the ZnO and TiO2 nanoparticles could be indexed to hexagonal and rutile phase, respectively. Aggregated nanoparticles of ZnO and TiO2 with spherical-like shapes were observed with particle diameter in the range of 80-100 nm. These nanoparticles were used for photocatalytic degradation of various dyes, Rhodamine B (RhB), Methylene blue (MB) and Acridine orange (AO) under solar light irradiation at room temperature. Effect of the amount of catalyst on the rate of photodegradation was investigated. In general, because ZnO is unstable, due to incongruous dissolution to yield Zn(OH)2 on the ZnO particle surfaces and thus leading to catalyst inactivation,the catalytic activity of the system for photodegradation of dyes decreased dramatically when TiO2 was replaced by ZnO.Photocatalytic degradation of some organic dyes under solar light irradiation...

Photocatalytic degradation of some organic dyes under solar light irradiation...Iranian Chemical Society
La actualidad más candente (20)
Bicrystalline Titania Photocatalyst for Reduction of CO2 to Solar Fuels

Bicrystalline Titania Photocatalyst for Reduction of CO2 to Solar Fuels
Electrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...

Electrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...
Synthesis of Spinel based Catalysts by Wet chemical methods for Colour Remova...

Synthesis of Spinel based Catalysts by Wet chemical methods for Colour Remova...
Solar photocatalytic process & solar photocatalytic reactors

Solar photocatalytic process & solar photocatalytic reactors
Applications of metal nanoparticles in photocatalysis

Applications of metal nanoparticles in photocatalysis
Roll of nanomaterials in water treatment as photocatalysts copy

Roll of nanomaterials in water treatment as photocatalysts copy
Mechanistic aspects of C-C cross coupling reaction

Mechanistic aspects of C-C cross coupling reaction
final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...

final accept-Optical and structural properties of TiO2 nanopowders with Co-Ce...
Photocatalytic degradation of some organic dyes under solar light irradiation...

Photocatalytic degradation of some organic dyes under solar light irradiation...
Renewable Fuels by Photocatalytic Reduction of carbondioxide (CO2); (Artifici...

Renewable Fuels by Photocatalytic Reduction of carbondioxide (CO2); (Artifici...
Similar a 高分子報告
Similar a 高分子報告 (20)
“Improving the sustainability of photovoltaic materials” – Dr Patrick Isherw...

“Improving the sustainability of photovoltaic materials” – Dr Patrick Isherw...
Integration of Nanomaterial with Semiconductor.pptx

Integration of Nanomaterial with Semiconductor.pptx
Sunlight-driven water-splitting using two-dimensional carbon based semiconduc...

Sunlight-driven water-splitting using two-dimensional carbon based semiconduc...
Sunlight-driven water-splitting using twodimensional carbon based semiconductors

Sunlight-driven water-splitting using twodimensional carbon based semiconductors
Más de submarine35
Más de submarine35 (6)
Último
💉💊+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHABI}}+971581248768
+971581248768 Mtp-Kit (500MG) Prices » Dubai [(+971581248768**)] Abortion Pills For Sale In Dubai, UAE, Mifepristone and Misoprostol Tablets Available In Dubai, UAE CONTACT DR.Maya Whatsapp +971581248768 We Have Abortion Pills / Cytotec Tablets /Mifegest Kit Available in Dubai, Sharjah, Abudhabi, Ajman, Alain, Fujairah, Ras Al Khaimah, Umm Al Quwain, UAE, Buy cytotec in Dubai +971581248768''''Abortion Pills near me DUBAI | ABU DHABI|UAE. Price of Misoprostol, Cytotec” +971581248768' Dr.DEEM ''BUY ABORTION PILLS MIFEGEST KIT, MISOPROTONE, CYTOTEC PILLS IN DUBAI, ABU DHABI,UAE'' Contact me now via What's App…… abortion Pills Cytotec also available Oman Qatar Doha Saudi Arabia Bahrain Above all, Cytotec Abortion Pills are Available In Dubai / UAE, you will be very happy to do abortion in Dubai we are providing cytotec 200mg abortion pill in Dubai, UAE. Medication abortion offers an alternative to Surgical Abortion for women in the early weeks of pregnancy. We only offer abortion pills from 1 week-6 Months. We then advise you to use surgery if its beyond 6 months. Our Abu Dhabi, Ajman, Al Ain, Dubai, Fujairah, Ras Al Khaimah (RAK), Sharjah, Umm Al Quwain (UAQ) United Arab Emirates Abortion Clinic provides the safest and most advanced techniques for providing non-surgical, medical and surgical abortion methods for early through late second trimester, including the Abortion By Pill Procedure (RU 486, Mifeprex, Mifepristone, early options French Abortion Pill), Tamoxifen, Methotrexate and Cytotec (Misoprostol). The Abu Dhabi, United Arab Emirates Abortion Clinic performs Same Day Abortion Procedure using medications that are taken on the first day of the office visit and will cause the abortion to occur generally within 4 to 6 hours (as early as 30 minutes) for patients who are 3 to 12 weeks pregnant. When Mifepristone and Misoprostol are used, 50% of patients complete in 4 to 6 hours; 75% to 80% in 12 hours; and 90% in 24 hours. We use a regimen that allows for completion without the need for surgery 99% of the time. All advanced second trimester and late term pregnancies at our Tampa clinic (17 to 24 weeks or greater) can be completed within 24 hours or less 99% of the time without the need surgery. The procedure is completed with minimal to no complications. Our Women's Health Center located in Abu Dhabi, United Arab Emirates, uses the latest medications for medical abortions (RU-486, Mifeprex, Mifegyne, Mifepristone, early options French abortion pill), Methotrexate and Cytotec (Misoprostol). The safety standards of our Abu Dhabi, United Arab Emirates Abortion Doctors remain unparalleled. They consistently maintain the lowest complication rates throughout the nation. Our Physicians and staff are always available to answer questions and care for women in one of the most difficult times in their lives. The decision to have an abortion at the Abortion Cl+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Último (20)
Apidays Singapore 2024 - Scalable LLM APIs for AI and Generative AI Applicati...

Apidays Singapore 2024 - Scalable LLM APIs for AI and Generative AI Applicati...
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
"I see eyes in my soup": How Delivery Hero implemented the safety system for ...

"I see eyes in my soup": How Delivery Hero implemented the safety system for ...
AWS Community Day CPH - Three problems of Terraform

AWS Community Day CPH - Three problems of Terraform
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER

EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Emergent Methods: Multi-lingual narrative tracking in the news - real-time ex...

Emergent Methods: Multi-lingual narrative tracking in the news - real-time ex...
ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke

ProductAnonymous-April2024-WinProductDiscovery-MelissaKlemke
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff

Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff
Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...

Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...
Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...

Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...
高分子報告
- 2. • make a new phase of carbon, called linear carbon • explosion in laboratory at Allied Chemical • injury and the end
- 3. Others persisted linear carbon is now coming closer to possible success
- 4. The story started from…. • Heeger, McDiaramid, and Shirakawa's discovery of conducting behavior in doped polyacetylene samples
- 5. Application nowadays • photovoltaic cells • field-effect transistors • chemical sensors
- 6. From Harvard University Digital Library for Physics and Astronomy • Improved photovoltaic cells and electrodes for use therein, particularly electrodes employing amorphous silicon or polyacetylene coating are produced by a process which includes …….. http://adsabs.harvard.edu/abs/1983doe..reptV....S
- 7. From Harvard University Digital Library for Physics and Astronomy • Planar and sandwich types of polyacetylene metal-semiconductor field- effect transistors (MESFETs) have been fabricated. http://nina.ecse.rpi.edu/2dfet/images/ntype2_big.gif http://adsabs.harvard.edu/abs/1991JaJAP..30.2101C
- 8. Chemical Sensor • Polyacetylene based nonlinear optical material prepared by ring-opening metathesis polymerization of cyclooctatetrene derivatives.
- 10. Unfortunately • However, Polyacetylene is thermally unstable and insoluble, making it unsuitable for general use.
- 12. Polyphenylenevinylene http://lupingyu.uchicago.edu/clip_image026.gif
- 13. Polyphenylenevinylene • Main material of Polymer Light Emitting Diode(PLED) http://www.euso.be/euso/home/images/test2.pdf
- 14. Polydiacetylenes • applications in optical limiters, waveguides, and thermometric sensors
- 15. Special properties • per-chain stiffness close to diamond • high coefficients for tripling the frequency of incoming light • negative thermal expansion coefficients tunable to near zero by introducing defects
- 16. Most Application • printed inks containing diacetylene microcrystals have been used as time- temperature indicators
- 17. Mechanism • Diacetylenes typically have an actuation energy for thermal polymerization of about 20 to 28 kcal/mol, which can be tuned to closely match the activation energy and degradation rate for important perishables
- 18. Good color indicator • Color changes of the diacetylene microcrystals during thermal polymerization provide a visual indication of whether a perishable product in the same thermal environment has degraded as a result of integrated time-temperature exposure
- 19. • Over a billion of these diacetylene polymerization-based indicators have been used on individual vaccine vials since 1996 to assist disease eradication in parts of the world that do not have a reliable cold chain
- 20. • The United States Army is using these diacetylene indicators on cartons of their MREs (Meals Ready to Eat)
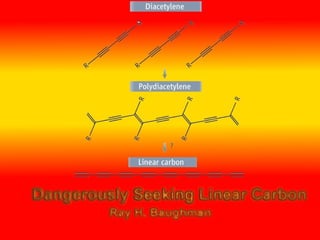
- 21. Explosive compound • Though the advantages mention before, there is a problem on synthesis Polydiacetylenes • With increasing n, crystals comprising only polyynes--that is, R − (C ≡ C) n − R molecules-- become increasingly unstable, unless R is a bulky substituent group
- 22. Dangerous….. • At room temperature, it can explode if subjected to physical shock, and it decomposes slowly under ambient light, forming an insoluble material with a graphite-like appearance
- 23. Solution? • Poly(diiododiacetylene), or PIDA represents a possible solution to the general problem of polydiacetylene synthesis
- 24. Why is it? • The abundance of transition metal–catalyzed reactions available for adding substituents in place of a carbon-halogen bond makes PIDA a potential precursor to a wide variety of polydiacetylenes. • Sn2 Sn1 − δ− Nu + C X C Nu + X− δ+ δ− + δ C X C + X− − C + Nu C Nu Organic Chemistry Bruice
- 25. A complex compound • C4I2 is complexed with a specially chosen agent, an oxalamide. In these crystals, 1,4-addition polymerization of diacetylene occurs spontaneously to produce single crystals of the complexed polydiacetylene
- 26. Getting Better • The complexing agent appears to stabilize the polymerized C4I2 against explosive decomposition and eliminates the type of disorder
- 27. Disorder comes from nature • Disordered along the direction of the molecular axis. In these crystals, the alignment between adjacent monomers is random, preventing ordered polymerization
- 28. Problem caused from disorder • Obtaining the desired optical and electronic properties of a given conjugated polymer requires a highly ordered molecular structure
- 29. Way to solve the problem • if the diynes are first aligned appropriately in the solid state at a distance commensurate with the repeat distance in the target polymer, then their arrangement in space will control their reactivity, leading to ordered topochemical polymerization
- 30. Best distance • Distance of 4.9A is highly preferable for the aligned dynes with target to form highly order structure.
- 31. Conclusion • With all the efforts to make the polydiethylene mentioned before, we shall have a blueprint on how could these material be utilized and how to make it real.
- 32. • Whether or not linear carbon synthesis is practical by the poly(C4I2) route, the idea with using poly C4I2 has provided both an exciting new electronic material and a route that could lead to novel related and derivative materials.
- 33. References • Dangerously Seeking Linear Carbon Ray H. Baughman Science mag Vol. 312. no. 5776, pp. 1009 - 1110 • Preparation of Poly(diiododiacetylene), an Ordered Conjugated Polymer of Carbon and Iodine Aiwu Sun, Joseph W. Lauher, Nancy S. Goroff Vol. 312. no. 5776, pp. 1030 - 1034 • Harvard University Digital Library for Physics and Astronomy • Organic Chemistry ver 4 Bruice Original Image Source: ( Images used in this PowerPoint are modified by Stan Yang) http://stc-mditr.org/research/oeoaomd/projects/2.2.1.cfm http://nina.ecse.rpi.edu/2dfet/images/ntype2_big.gif