Key Concepts Unit A Chemistry 30
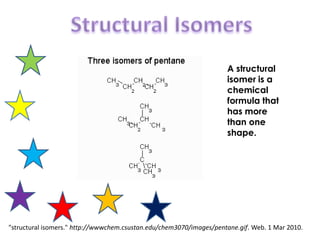
- 1. Structural Isomers A structural isomer is a chemical formula that has more than one shape. "structural isomers." http://wwwchem.csustan.edu/chem3070/images/pentane.gif. Web. 1 Mar 2010.
- 2. Saturated/Unsaturated Hydrocarbons Saturated hydrocarbons have single bonds and cannot bond with compounds anymore. Unsaturated hydrocarbons have double or triple bonds and can bond with compounds.
- 3. Halogenated Hydrocarbons Are produced When a Hydrocarbon has one or more hydrogen group replaced with a Halogen(group 17) Can help create a saturated hydrocarbon by breaking double or triple bonds. http://www.youtube.com/watch?v=1qGPWdm2MlI (watch to 1:50)
- 4. Carboxylic Acids The Basic Structure is R-COOH (R can be replaced by a hydrogen) They are used in an esterfication reaction with an alcohol When naming you change the ending to “oic” acid (methane would be methanoic acid) They have hydrogen bonds increasing their boiling points.
- 5. Structuralformulas The structural formula of a chemical compound is a graphical representation showing how the atoms are arranged. This is a structural formula where atoms are arranged forming a compound Structural formulas rock my world!!! http://en.wikipedia.org/wiki/Structural_formula http://www.1-formula.com/upl/Image/800px-Acetone-structural.png
- 6. Esterification http://en.wikipedia.org/wiki/Esterification This is an Esterification reaction I love Esterification reactions because they smell good Esterification is the general name for a chemical reaction in which two reactants (typically an alcohol and a carboxylic acid) form an ester as the reaction product.
- 7. Addition Reaction An addition reaction is the reaction between two or more unsaturated hydrocarbons to produce one large molecule.
- 8. Substitution Reaction When a carbon-hydrogen bond breaking occurs in an alkane or aromatic organic reaction An example is the reaction in which the chlorine atom in chloromethane is displaced by the hydroxide ion to form methanol CH3Cl + -OH CH3OH + Cl-
- 9. Addition and substitution reaction Video http://www.youtube.com/watch?v=nVDUB4mWJ8M
- 11. A Complete Combustion reaction is when the reactant will burn in oxygen producing a limited number of products.
- 12. An incomplete combustion reaction is when there isn’t enough oxygen to react with.C?H? + O2 CO2 + H2O C?H? + O2 CO + H2O "combustion reactions.". http://upload.wikimedia.org/wikipedia/commons/3/3c/Et_baal.jpg. Web. 1 Mar 2010
- 13. Monomers monomer - a simple compound whose molecules can join together to form polymers.
- 14. Polymers Polymers are created when two or more monomers are linked together in a chain like pattern
- 15. Polymerization Polymerization - The chemical process, normally needs the aid of a catalyst, to form a polymer by bonding together monomers. http://www.youtube.com/watch?v=LJ5hjUeZt7U&feature=related
- 16. Polymers are made from Monomers http://www.youtube.com/watch?v=Jpv2RUjNFjo