2013 Medicines in Development: Older Americans
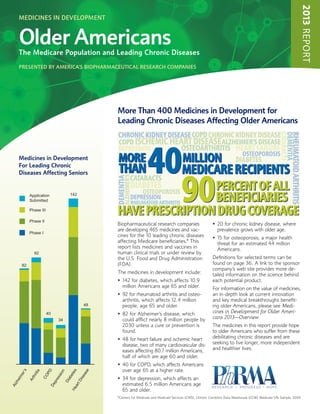
- 1. 2013 REPORT Medicines in Development Older Americans The Medicare Population and Leading Chronic Diseases presented by america’s biopharmaceutical research companies More Than 400 Medicines in Development for Leading Chronic Diseases Affecting Older Americans CHRONIC KIDNEY DISEASE COPD CHRONIC KIDNEY DISEASE CATARACTS DEMENTIA RHEUMATOID ARTHRITIS COPD ISCHEMIC HEART DISEASE ALZHEIMER'S DISEASE 40 DEPRESSION OSTEOARTHRITIS HEART FAILURE OSTEOPOROSIS Medicines in Development MORE MILLION DIABETES For Leading Chronic THAN MEDICARE RECIPIENTS 90 Diseases Affecting Seniors CATARACTS ALZHEIMER'S DEMENTIA DIABETES OSTEOPOROSIS PERCENT OF ALL Application Submitted 142 DEPRESSION RHEUMATOID ARTHRITIS BENEFICIARIES Phase III HAVE PRESCRIPTION DRUG COVERAGE Phase II Biopharmaceutical research companies • 0 for chronic kidney disease, where 2 are developing 465 medicines and vac- prevalence grows with older age. Phase I cines for the 10 leading chronic diseases • 5 for osteoporosis, a major health 1 affecting Medicare beneficiaries.* This threat for an estimated 44 million report lists medicines and vaccines in Americans. 92 human clinical trials or under review by the U.S. Food and Drug Administration Definitions for selected terms can be 82 (FDA). found on page 36. A link to the sponsor company’s web site provides more de- The medicines in development include: tailed information on the science behind • 42 for diabetes, which affects 10.9 1 each potential product. million Americans age 65 and older. For information on the value of medicines, 9 • 2 for rheumatoid arthritis and osteo- an in-depth look at current innovation arthritis, which affects 12.4 million and key medical breakthroughs benefit- 48 people, age 65 and older. ing older Americans, please see Medi- 40 8 • 2 for Alzheimer’s disease, which cines in Development for Older Ameri- 34 could afflict nearly 8 million people by cans 2013—Overview. 2030 unless a cure or prevention is The medicines in this report provide hope found. to older Americans who suffer from these 4 • 8 for heart failure and ischemic heart debilitating chronic diseases and are disease, two of many cardiovascular dis- seeking to live longer, more independent eases affecting 80.7 million Americans, and healthier lives. half of which are age 60 and older. • 0 for COPD, which affects Americans 4 over age 65 at a higher rate. ’s PD s e s ion ete as riti er CO s eim ise • 4 for depression, which affects an 3 th ab es Ar tD Di pr zh estimated 6.5 million Americans age De ar Al 65 and older. He *Centers for Medicare and Medicaid Services (CMS), Chronic Condition Data Warehouse (CCW) Medicare 5% Sample, 2009
- 2. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase* 18F-florbetaben Piramal Healthcare Alzheimer’s disease (diagnosis) Phase III (BAY 94-9172) Mumbai, India piramalenterprises.com AAB-003/PF-05236812 Janssen Alzheimer Immunotherapy Alzheimer’s disease Phase I (amyloid beta protein inhibitor) South San Francisco, CA janimm.com Pfizer pfizer.com New York, NY ABT-126 AbbVie mild to moderate Alzheimer’s Phase II (α7-NNR antagonist) North Chicago, IL disease abbvie.com ABT-288 AbbVie Alzheimer’s disease Phase II completed (neurotransmitter receptor North Chicago, IL abbvie.com modulator) ABT-384 AbbVie Alzheimer’s disease Phase II completed North Chicago, IL abbvie.com ABT-957 AbbVie Alzheimer’s disease Phase I (calpain inhibitor) North Chicago, IL abbvie.com AC-1204 Accera Alzheimer’s disease Phase II/III Broomfield, CO accerapharma.com ACC-001/PF-05236806 Janssen Alzheimer Immunotherapy Alzheimer’s disease Phase II South San Francisco, CA janimm.com Pfizer pfizer.com New York, NY AD02 vaccine Affiris treatment of Alzheimer’s disease Phase II Vienna, Austria gsk.com GlaxoSmithKline Rsch. Triangle Park, NC AD03 vaccine Affiris treatment of Alzheimer’s disease Phase I Vienna, Austria gsk.com GlaxoSmithKline Rsch. Triangle Park, NC AD4833/TOMM40 Takeda Pharmaceuticals International Alzheimer’s disease prevention Phase I Deerfield, IL takeda.com APH-0703 Aphios Alzheimer’s disease Phase I/II (protein kinase C activator) Woburn, MA aphios.com *For more information about a specific medicine or company in the report, please click on the provided link. 2 Medicines in Development Older Americans 2013
- 3. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase ARC029 Archer Pharmaceuticals Alzheimer’s disease Phase I (nilvadipine) Sarasota, FL archerpharma.com ARC031 Archer Pharmaceuticals Alzheimer’s disease Phase I (soluble amyloid reducing/clearing Sarasota, FL archerpharma.com agent) ARC031 SR Archer Pharmaceuticals Alzheimer’s disease Phase I (soluble amyloid reducing/clearing Sarasota, FL archerpharma.com agent) AVN 101 Avineuro Pharmaceuticals Alzheimer’s disease Phase II (serotonin 5-HT6 receptor San Diego, CA avineuro.com antagonist) AVN 322 Avineuro Pharmaceuticals Alzheimer’s disease Phase I (serotonin 5-HT6 receptor San Diego, CA avineuro.com antagonist) AVN 397 Avineuro Pharmaceuticals Alzheimer’s disease Phase II San Diego, CA avineuro.com AZD1446 AstraZeneca Alzheimer’s disease Phase I (alpha4/beta2 neuronal nicotinic Wilmington, DE astrazeneca.com receptor agonist) Targacept Winston-Salem, NC AZD2184 Navidea Biopharmaceuticals Alzheimer’s disease (diagnosis) Phase I (PET imaging) Dublin, OH navidea.com AZD2995 Navidea Biopharmaceuticals Alzheimer’s disease (diagnosis) Phase I (PET imaging) Dublin, OH navidea.com AZD3293 AstraZeneca Alzheimer’s disease Phase I (beta secretase) Wilmington, DE astrazeneca.com Astex Pharmaceuticals Dublin, CA AZD5213 AstraZeneca Alzheimer’s disease Phase II (histamine-3 receptor antagonist) Wilmington, DE astrazeneca.com ß-secretase inhibitor Eli Lilly Alzheimer’s disease Phase II (LY2886721) Indianapolis, IN (slow disease progression) lilly.com BAN2401 Eisai early-stage Alzheimer’s disease Phase II Woodcliff Lake, NJ eisai.com BAY 85-8101 Piramal Healthcare Alzheimer’s disease (diagnosis) Phase I completed (18F-labeled radiopharmaceutical) Mumbai, India piramalenterprises.com Medicines in Development Older Americans 2013 3
- 4. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase BIIB037 Biogen Idec Alzheimer’s disease Phase I (anti-ß-amyloid antibody) Weston, MA biogenidec.com bisnorcymserine QR Pharma Alzheimer’s disease Phase I (BNC) Berwyn, PA qrpharma.com BMS-241027 Bristol-Myers Squibb Alzheimer’s disease, Phase I (microtubule stabilizer) Princeton, NJ senile dementia bms.com CAD106 Novartis Pharmaceuticals Alzheimer’s disease Phase II East Hanover, NJ novartis.com CERE-110 Ceregene Alzheimer’s disease Phase II (AAV-NGF gene therapy) San Diego, CA ceregene.com crenezumab Genentech Alzheimer’s disease Phase II (anti-Abeta) South San Francisco, CA gene.com davunetide intranasal Allon Therapeutics Alzheimer’s disease Phase II Vancouver, Canada allontherapeutics.com donepezil/memantine Adamas Pharmaceuticals moderate to severe Alzheimer’s Phase II extended-release fixed-dose Emeryville, CA disease adamaspharma.com combination Forest Laboratories frx.com New York, NY DSP-8658 Sunovion Pharmaceuticals Alzheimer’s disease Phase I (PPAR alpha/gamma agonist) Marlborough, MA (see also diabetes) sunovion.com E2609 Eisai Alzheimer’s disease Phase I completed Woodcliff Lake, NJ eisai.com ELND005 Elan Alzheimer’s disease Phase II (amyloid beta-protein inhibitor) Dublin, Ireland elan.com EVP-0962 EnVivo Pharmaceuticals Alzheimer’s disease Phase II (gamma secretase modulation) Watertown, MA envivopharma.com EVP-6124 EnVivo Pharmaceuticals Alzheimer’s disease Phase II (nicotine A7 agonist) Watertown, MA envivopharma.com Exebryl-1® ProteoTech Alzheimer’s disease Phase I Alzheimer’s disease therapeutic Kirkland, WA proteotech.com F-18 T808 Siemens Molecular Imaging Alzheimer’s disease (diagnosis) Phase 0 (PET imaging agent) Malvern, PA medical.siemens.com F18-flutemetamol GE Healthcare Alzheimer’s disease (diagnosis) application submitted (PET imaging agent) Waukesha, WI gehealthcare.com 4 Medicines in Development Older Americans 2013
- 5. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase Gammagard® Baxter International early-stage Alzheimer’s disease, Phase III immune globulin Deerfield, IL mid-stage Alzheimer’s disease baxter.com gamma secretase modulator, Bristol-Myers Squibb Alzheimer’s disease, in clinical trials A-beta modulator Princeton, NJ senile dementia bms.com gantenerumab Roche early-stage Alzheimer’s disease Phase II/III (amyloid beta-protein inhibitor) Nutley, NJ roche.com GSK742457 GlaxoSmithKline dementia Phase II (5-HT6 receptor antagonist) Rsch. Triangle Park, NC gsk.com GSK2647544 GlaxoSmithKline Alzheimer’s disease Phase I (Lp-PLA2 inhibitor) Rsch. Triangle Park, NC gsk.com HPP 854 High Point Pharmaceuticals Alzheimer’s disease Phase I (BACE1 protein inhibitor) High Point, NC highpointpharma.com human immunoglobulin Grifols Alzheimer’s disease Phase III (intravenous) Los Angeles, CA grifols.com KU-046 Kareus Therapeutics Alzheimer’s disease Phase I (amyloid-ß protein modulator) La Chaux-de-Fonds, Switzerland kareustherapeutics.com Lu AE58054 Lundbeck USA Alzheimer’s disease Phase II Deerfield, IL lundbeck.com masitinib AB Science USA Alzheimer’s disease Phase III completed (AB-1010) Short Hills, NJ ab-science.com MCD-386 Mithridion Alzheimer’s disease Phase I Madison, WI mithridion.com MK-3328 Merck Alzheimer’s disease (diagnosis) Phase I completed (PET imaging) Whitehouse Station, NJ merck.com MK-8931 Merck Alzheimer’s disease Phase II/III (BACE1 protein inhibitor) Whitehouse Station, NJ merck.com MSDC-0160 Metabolic Solutions Development Alzheimer’s disease Phase II Kalamazoo, MI (see also diabetes) msdrx.com NAV4694 Navidea Biopharmaceuticals Alzheimer’s disease (diagnosis) Phase III (fluorine-18 labeled PET Dublin, OH navidea.com imaging agent) NIC5-15 Humanetics Alzheimer’s disease Phase II Minneapolis, MN humaneticscorp.com Medicines in Development Older Americans 2013 5
- 6. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase PF-05212377 Pfizer Alzheimer’s disease Phase II (SAM-760) New York, NY pfizer.com Posiphen® QR Pharma Alzheimer’s disease, Phase II R-phenserine Berwyn, PA mild cognitive impairment qrpharma.com PRX-3140 Nanotherapeutics Alzheimer’s disease Phase II (serotonin 4 receptor agonist) Alachua, FL nanotherapeutics.com RG1577 Roche Alzheimer’s disease Phase II (MAO-B inhibitor) Nutley, NJ roche.com RG7129 Roche Alzheimer’s disease Phase I (BACE-1 inhibitor) Nutley, NJ roche.com rilapladib GlaxoSmithKline Alzheimer’s disease Phase II (Lp-PLA2 inhibitor) Rsch. Triangle Park, NC gsk.com RVX-208 Resverlogix Alzheimer’s disease Phase I Calgary, Canada resverlogix.com SAR110894 Sanofi US Alzheimer’s disease Phase II (H3 antagonist) Bridgewater, NJ sanofi.com SAR228810 Sanofi US Alzheimer’s disease Phase I (anti-protofibrillar AB mAb) Bridgewater, NJ sanofi.com sGC-1061 sGC Pharma Alzheimer’s disease Phase I Wellesley, MA sgcpharma.com solanezumab Eli Lilly Alzheimer’s disease Phase III (LY2062430) Indianapolis, IN lilly.com ST101 Sonexa Therapeutics Alzheimer’s disease Phase II San Diego, CA sonexa.com T-817MA Toyota Chemical mild to moderate Alzheimer’s Phase II completed (amyloid beta-protein inhibitor) Tokyo, Japan disease toyama-chemical.co.jp TAK-357 Takeda Pharmaceuticals International Alzheimer’s disease Phase I (oral cognitive enhancer) Deerfield, IL takeda.com TC-1734 Targacept Alzheimer’s disease Phase II (alpha4/beta2 neuronal nicotinic Winston-Salem, NC targacept.com receptor agonist) TC-5619 Targacept Alzheimer’s disease Phase I (alpha7 nAChR) Winston-Salem, NC targacept.com 6 Medicines in Development Older Americans 2013
- 7. Medicines in Development for Older Americans Alzheimer’s Disease and Dementia Product Name Sponsor Indication Development Phase TRx-0237 TauRx Therapeutics Alzheimer’s disease, dementia Phase III (LMTX) Singapore, Republic of Singapore taurx.com TTP4000 TransTech Pharma Alzheimer’s disease Phase I High Point, NC ttpharma.com TTP488 TransTech Pharma Alzheimer’s disease Phase II High Point, NC ttpharma.com UB-311 United Biomedical mild to moderate Alzheimer’s Phase I (amyloid beta protein inhibitor Hauppauge, NY disease unitedbiomedical.com vaccine) V950 Merck Alzheimer’s disease Phase I Whitehouse Station, NJ merck.com velusetrag Theravance Alzheimer’s disease Phase I completed (TD-5108) South San Francisco, CA theravance.com VI-1121 VIVUS Alzheimer’s disease Phase II Mountain View, CA vivus.com XEL-001HP Xel Pharmaceuticals Alzheimer’s disease Phase I (transdermal patch) Draper, UT xelpharmaceuticals.com Arthritis Product Name Sponsor Indication Development Phase 2ME2 EntreMed rheumatoid arthritis Phase I Rockville, MD entremed.com ABT-122 AbbVie rheumatoid arthritis Phase I (anti-TNF/anti-IL-17 mAb) North Chicago, IL abbvie.com ABT-413 AbbVie rheumatoid arthritis Phase I (sphingosine 1 phosphate receptor North Chicago, IL abbvie.com modulator) ABT-494 AbbVie rheumatoid arthritis Phase I (JAK-1, 2, 3 inhibitor) North Chicago, IL abbvie.com Medicines in Development Older Americans 2013 7
- 8. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase ABT-652 AbbVie osteoarthritis of the knee Phase II (histamine H3 receptor modulator) North Chicago, IL abbvie.com ABT-981 AbbVie osteoarthritis Phase I (anti-IL-1 α and β mAb) North Chicago, IL abbvie.com Actemra® Genentech rheumatoid arthritis application submitted tocilizumab South San Francisco, CA (first-line therapy) gene.com Roche roche.com Nutley, NJ -------------------------------------- ----------------------------------------- rheumatoid arthritis Phase III (early-stage disease) gene.com roche.com adalimumab biosimilar Boehringer Ingelheim Pharmaceuticals rheumatoid arthritis Phase I completed Ridgefield, CT boehringer-ingelheim.com AF-219 Afferent Pharmaceuticals osteoarthritis Phase II (purinoceptor P2X3 antagonist) San Mateo, CA afferentpharma.com AIN457 Novartis Pharmaceuticals rheumatoid arthritis Phase III (secukinumab) East Hanover, NJ novartis.com ARG301 arGentis Pharmaceuticals rheumatoid arthritis Phase I Memphis, TN argentisrx.com ARRY-797 Array BioPharma moderate to severe pain associated Phase II (p38 MAPK inhibitor) Boulder, CO with osteoarthritis of the knee arraybiopharma.com ASP015K Astellas Pharma US rheumatoid arthritis Phase II (JAK-3 inhibitor) Northbrook, IL astellas.com ASP2408 Perseid Therapeutics rheumatoid arthritis Phase I (CTLA4-Ig fusion protein) Redwood City, CA perseidtherapeutics.com baricitinib Eli Lilly rheumatoid arthritis Phase III (LY3009104) Indianapolis, IN (see also chronic kidney) lilly.com BI-695500 Boehringer Ingelheim Pharmaceuticals rheumatoid arthritis Phase III (rituximab biosimilar) Ridgefield, CT boehringer-ingelheim.com BMS-817399 Bristol-Myers Squibb osteoarthritis, Phase II (CCR1 antagonist) Princeton, NJ rheumatoid arthritis bms.com BT-061 AbbVie rheumatoid arthritis Phase II North Chicago, IL abbvie.com Biotest biotest.de Dreieich, Germany 8 Medicines in Development Older Americans 2013
- 9. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase buprenorphine transmucosal BioDelivery Sciences International chronic pain associated with Phase III Raleigh, NC osteoarthritis bdsi.com Endo Pharmaceuticals endo.com Malvern, PA canakinumab Novartis Pharmaceuticals gouty arthritis application submitted (ACZ885) East Hanover, NJ (see also diabetes) novartis.com -------------------------------------- ----------------------------------------- osteoarthritis Phase II novartis.com cebranopadol Forest Laboratories pain due to osteoarthritis of the Phase II New York, NY knee frx.com CEP-33237 Cephalon osteoarthritis pain Phase III (hydrocodone extended-release) Frazer, PA cephalon.com CF101 Can-Fite BioPharma rheumatoid arthritis Phase II Waltham, MA canfite.com Chondrogen™ Osiris Therapeutics osteoarthritis (prevention) Phase II mesenchymal stem cell therapy Columbia, MD osiris.com Cimzia® UCB early rheumatoid arthritis Phase III certolizumab pegol Smyrna, GA ucb.com clazakizumab Bristol-Myers Squibb osteoarthritis, Phase II (anti-IL6-mAb) Princeton, NJ rheumatoid arthritis bms.com CNTO-1959 Janssen Biotech rheumatoid arthritis Phase II Horsham, PA janssenbiotech.com COV-155 Mallinckrodt osteoarthritis Phase III Hazelwood, MO mallinckrodt.com COV-795 Mallinckrodt osteoarthritis Phase III Hazelwood, MO mallinckrodt.com CT-1578 Cell Therapeutics rheumatoid arthritis Phase I (JAK2/FLT-3/c-Fms inhibitor) Seattle, WA celltherapeutics.com Dekavil Pfizer rheumatoid arthritis Phase I (F8-IL10) New York, NY pfizer.com Philogen philogen.com Sovicille, Italy DMI-9523 Ampio Pharmaceuticals osteoarthritis Phase I Greenwood Village, CO ampiopharma.com Medicines in Development Older Americans 2013 9
- 10. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase etoricoxib Merck rheumatoid arthritis Phase III Whitehouse Station, NJ merck.com fostamatinib AstraZeneca rheumatoid arthritis Phase III (SYK inhibitor) Wilmington, DE astrazeneca.com Rigel Pharmaceuticals rigel.com South San Francisco, CA FX005 Flexion Therapeutics osteoarthritis of the knee Phase I/II (p38 MAPK inhibitor) Woburn, MA flexiontherapeutics.com FX006 Flexion Therapeutics osteoarthritis of the knee Phase II (triamcinolone acetonide Woburn, MA flexiontherapeutics.com intra-articular sustained-release) gevokizumab XOMA osteoarthritis of the hand Phase II (XOMA 052) Berkeley, CA xoma.com GLPG-0555 Galapagos osteoarthritis Phase I Mechelen, Belgium gsk.com GlaxoSmithKline Rsch. Triangle Park, NC GLPG-0634 AbbVie rheumatoid arthritis Phase II North Chicago, IL abbvie.com Galapagos Mechelen, Belgium GSK2941266 GlaxoSmithKline rheumatoid arthritis Phase II (CCR1 chemokine receptor Rsch. Triangle Park, NC gsk.com antagonist) HE3286 Harbor Biosciences rheumatoid arthritis Phase II San Diego, CA (see also diabetes) harbortx.com INCB39110 Incyte active rheumatoid arthritis Phase II (JAK1 inhibitor) Wilmington, DE incyte.com IP045 Iroko Pharmaceuticals pain associated with osteoarthritis Phase I (meloxicam) Philadelphia, PA of the knee iroko.com IP-880 Iroko Pharmaceuticals osteoarthritis pain Phase III (low dose diclofenac) Philadelphia, PA iroko.com 10 Medicines in Development Older Americans 2013
- 11. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase ISIS-CRPRx Isis Pharmaceuticals rheumatoid arthritis Phase II (C-reactive protein inhibitor) Carlsbad, CA isispharm.com JNJ-38518168 Janssen Research Development rheumatoid arthritis Phase II (histamine H4 receptor Raritan, NJ janssenrnd.com antagonist) JNJ-39439335 Janssen Research Development chronic pain associated with Phase I (mavatrep) Raritan, NJ osteoarthritis of the knee janssenrnd.com JNJ-40346527 Janssen Research Development rheumatoid arthritis Phase II (M-CSF receptor antagonist) Raritan, NJ janssenrnd.com LT-NS001 Logical Therapeutics osteoarthritis Phase II/III (naproxen etemesil) Foxboro, MA logicaltx.com LX2931 Lexicon Pharmaceuticals rheumatoid arthritis Phase II (S1P lyase inhibitor) The Woodlands, TX lexicon-genetics.com mavrilimumab AstraZeneca rheumatoid arthritis Phase II (anti-GM-CSFR mAb) Wilmington, DE astrazeneca.com MedImmune medimmune.com Gaithersburg, MD MEDI-5117 AstraZeneca rheumatoid arthritis Phase I (anti-IL-6 mAb) Wilmington, DE astrazeneca.com MedImmune medimmune.com Gaithersburg, MD mesenchymal stem cell therapy for Medipost osteoarthritis Phase I/II cartilage repair Seoul, South Korea medi-post.com MK-8457 Merck rheumatoid arthritis Phase II Whitehouse Station, NJ merck.com MK-8808 Merck rheumatoid arthritis Phase I Whitehouse Station, NJ merck.com MORAb-022 Eisai rheumatoid arthritis Phase I (IgG1 mAb) Woodcliff Lake, NJ eisai.com namilumab Takeda Pharmaceuticals International rheumatoid arthritis Phase I Deerfield, IL takeda.com naproxcinod NicOx osteoarthritis application submitted Sophia Antipolis, France nicox.com Medicines in Development Older Americans 2013 11
- 12. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase NN8209 Novo Nordisk rheumatoid arthritis Phase II (anti-C5aR-151) Princeton, NJ novonordisk.com NN8210 Novo Nordisk rheumatoid arthritis Phase I (anti-C5aR-215) Princeton, NJ novonordisk.com NN8226 Novo Nordisk rheumatoid arthritis Phase II (anti-IL20 mAb) Princeton, NJ novonordisk.com NN8765 Novo Nordisk rheumatoid arthritis Phase I (anti-NKG2A) Princeton, NJ novonordisk.com NN8828 Novo Nordisk rheumatoid arthritis Phase II (anti-IL21 mAb) Princeton, NJ novonordisk.com olokizumab UCB rheumatoid arthritis Phase II Smyrna, GA ucb.com OLT-1177 Olatec Industries pain associated with osteoarthritis Phase II New York, NY of the knee osteoarthritis medicine Eli Lilly osteoarthritis pain Phase I (NCE) Indianapolis, IN lilly.com otelixizumab GlaxoSmithKline rheumatoid arthritis Phase I (CD3 mAb) Rsch. Triangle Park, NC gsk.com Otrexup™ Antares Pharma rheumatoid arthritis application submitted methotrexate subcutaneous Ewing, NJ antarespharma.com auto-injection ozoralizumab Ablynx rheumatoid arthritis Phase II Ghent, Belgium ablynx.com PDA-001 Celgene Cellular Therapeutics rheumatoid arthritis Phase I/II (stem cell therapy) Summit, NJ celgene.com Pennsaid® 2% Mallinckrodt osteoarthritis pain and stiffness application submitted diclofenac transdermal Hazelwood, MO mallinckrodt.com PF-04171327 Pfizer rheumatoid arthritis Phase II (SGRM) New York, NY pfizer.com PF-04236921 Pfizer rheumatoid arthritis Phase I (mAb) New York, NY pfizer.com PF-05280586 Pfizer rheumatoid arthritis Phase I (rituximab biosimilar) New York, NY pfizer.com 12 Medicines in Development Older Americans 2013
- 13. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase PLX5622 Plexxikon rheumatoid arthritis Phase I (FGF-1 antagonist) Berkeley, CA plexxikon.com Prolia® Amgen rheumatoid arthritis Phase II denosumab Thousand Oaks, CA amgen.com PRTX-100 Protalex rheumatoid arthritis Phase I (staphylococcal protein A) Summit, NJ protalex.com Ravax™ Immune Response BioPharma rheumatoid arthritis Phase III rheumatoid arthritis vaccine New York, NY immuneresponsebiopharma.com RX-10001 Resolvyx Pharmaceuticals rheumatoid arthritis Phase I (synthetic resolvin) Cambridge, MA resolvyx.com SAN-300 Santarus rhematoid arthritis Phase I (anti-VLA-1 antibody) San Diego, CA santarus.com SAR113945 Sanofi US osteoarthritis Phase II (IKK-ß inhibitor) Bridgewater, NJ sanofi.com SAR292833 Sanofi US osteoarthritis pain Phase II (TRVP3 antagonist) Bridgewater, NJ sanofi.com sarilumab Sanofi US rheumatoid arthritis Phase III (anti-IL-6R mAb) Bridgewater, NJ sanofi.com Simponi® Janssen Biotech moderate to severe rheumatoid application submitted golimumab (intravenous) Horsham, PA arthritis janssenbiotech.com sirukumab GlaxoSmithKline rheumatoid arthritis Phase III (anti-IL-6 mAb) Rsch. Triangle Park, NC gsk.com Janssen Biotech janssenbiotech.com Horsham, PA sprifermin EMD Serono osteoarthritis Phase I (FGF-18) Rockland, MA emdserono.com TG-C TissueGene osteoarthritis Phase II (allogeneic cell therapy) Rockville, MD tissuegene.com Stelara® Janssen Research Development rheumatoid arthritis Phase II ustekinumab Raritan, NJ janssenrnd.com Medicines in Development Older Americans 2013 13
- 14. Medicines in Development for Older Americans Arthritis Product Name Sponsor Indication Development Phase V116517 Purdue Pharma pain associated with osteoarthritis Phase II Stamford, CT of the knee purduepharma.com veltuzumab Immunomedics rheumatoid arthritis Phase II subcutaneous Morris Plains, NJ immunomedics.com Takeda Pharmaceuticals International takeda.com Deerfield, IL VX30 Vaccinex rheumatoid arthritis Phase I Rochester, NY vaccinex.com VX-509 Vertex Pharmaceuticals rheumatoid arthritis Phase II Cambridge, MA vrtx.com Cataracts Product Name Sponsor Indication Development Phase dexamethasone ophthalmic EyeGate Pharma cataracts Phase II (EGP-437) Waltham, MA eyegatepharma.com Chronic Kidney Disease Product Name Sponsor Indication Development Phase Acthar® Gel Questcor Pharmaceuticals diabetic nephropathy Phase II corticotropin gel Anaheim Hills, CA questcor.com atrasentan AbbVie diabetic nephropathy Phase II North Chicago, IL abbvie.com AZD1722 Ardelyx chronic kidney disease Phase I (NHE3 inhibitor) Fremont, CA ardelyx.com AstraZeneca astrazeneca.com Wilmington, DE baricitinib Eli Lilly diabetic nephropathy Phase II (LY3009104) Indianapolis, IN (see also arthritis) lilly.com 14 Medicines in Development Older Americans 2013
- 15. Medicines in Development for Older Americans Chronic Kidney Disease Product Name Sponsor Indication Development Phase BB-3 Angion chronic kidney fibrosis Phase I Uniondale, NY angion.com BMS-813160 Bristol-Myers Squibb chronic kidney disease Phase I (dual CCR2/CCR5 antagonist) Princeton, NJ bms.com CCX140 ChemoCentryx diabetic nephropathy Phase I (CCR2 receptor antagonist) Mountain View, CA chemocentryx.com chronic kidney disease therapeutic Eli Lilly chronic kidney disease Phase I Indianapolis, IN lilly.com CLP-1004 Sorbent Therapeutics chronic kidney disease Phase I (potassium channel modulator) Sunnyvale, CA sorbent.com CTP-499 Concert Pharmaceuticals diabetic nephropathy Phase I Lexington, MA concertpharma.com INV-144 InVasc Therapeutics chronic kidney disease Phase II (losartan/thioctic acid) Tucker, GA invasc.net linagliptin Boehringer Ingelheim Pharmaceuticals diabetic nephropathy Phase III Ridgefield, CT boehringer-ingelheim.com Eli Lilly lilly.com Indianapolis, IN LY3016859 Eli Lilly diabetic nephropathy Phase I Indianapolis, IN lilly.com MP-146 Mitsubishi Tanabe Pharma chronic kidney disease Phase III Osaka, Japan mt-pharma.co.jp MR antagonist Eli Lilly chronic kidney disease Phase II (LY2623091) Indianapolis, IN lilly.com PF-00489791 Pfizer diabetic nephropathy Phase II (PDE5 inhibitor) New York, NY pfizer.com PF-04634817 Pfizer diabetic nephropathy Phase II (CCR2/CCR5 receptor antagonist) New York, NY pfizer.com Pyridorin™ NephroGenex diabetic nephropathy Phase II pyridoxamine Rsch. Triangle Park, NC (Fast Track) nephrogenex.com TGF-ß mAb Eli Lilly diabetic nephropathy Phase II (LY2382770) Indianapolis, IN lilly.com VTP-27999 Vitae Pharmaceuticals chronic kidney disease Phase I (renin inhibitor) Fort Washington, PA vitaepharma.com Medicines in Development Older Americans 2013 15
- 16. Medicines in Development for Older Americans Chronic Obstructive Pulmonary Disease (COPD) Product Name Sponsor Indication Development Phase aclidinium bromide/formoterol Forest Laboratories chronic obstructive pulmonary Phase III combination New York, NY disease (COPD) frx.com AZD2115 AstraZeneca COPD Phase II (MABA) Wilmington, DE astrazeneca.com Pulmagen Therapeutics Slough, United Kingdom AZD5423 AstraZeneca COPD Phase II (inhaled SGRM) Wilmington, DE astrazeneca.com AZD7594 AstraZeneca COPD Phase I (inhaled SGRM) Wilmington, DE astrazeneca.com BCT197 Novartis Pharmaceuticals COPD Phase II East Hanover, NJ novartis.com bedoradrine MediciNova exacerbations of COPD Phase II (MN-221) San Diego, CA medicinova.com benralizumab AstraZeneca COPD Phase II (anti-IL-5R mAb) Wilmington, DE astrazeneca.com MedImmune medimmune.com Gaithersburg, MD BI-137882 Boehringer Ingelheim Pharmaceuticals COPD Phase I Ridgefield, CT boehringer-ingelheim.com BIO-11006 BioMarck Pharmaceuticals COPD Phase II Durham, NC biomarck.com Dulera® Merck COPD application submitted mometasone/formoterol Whitehouse Station, NJ merck.com fluticasone furoate/vilanterol/ GlaxoSmithKline COPD Phase I umeclidinium combination Rsch. Triangle Park, NC gsk.com formoterol/fluticasone propionate Mylan Specialty COPD Phase II completed fixed-dose combination Basking Ridge, NJ mylanspecialty.com glycopyrrolate Sunovion Respiratory Development COPD Phase II (SUN-101) Marlborough, MA sunovion.com GSK225629 GlaxoSmithKline COPD Phase I (soluble epoxide hydrolase Rsch. Triangle Park, NC gsk.com inhibitor) 16 Medicines in Development Older Americans 2013
- 17. Medicines in Development for Older Americans Chronic Obstructive Pulmonary Disease (COPD) Product Name Sponsor Indication Development Phase GSK1325756 GlaxoSmithKline COPD Phase I (CXCR2 chemokine receptor Rsch. Triangle Park, NC gsk.com antagonist) GSK2269557 GlaxoSmithKline COPD Phase I (phosphoinositide 3 kinase Rsch. Triangle Park, NC gsk.com inhibitor) GSK961081 GlaxoSmithKline COPD Phase II (muscarinic antagonist/beta2 Rsch. Triangle Park, NC gsk.com agonist) Theravance theravance.com South San Francisco, CA losmapimod GlaxoSmithKline COPD Phase II (oral p38 kinase inhibitor) Rsch. Triangle Park, NC gsk.com MEDI-7814 AstraZeneca COPD Phase I (anti-C5/C5a mAb) Wilmington, DE astrazeneca.com MedImmune medimmune.com Gaithersburg, MD MEDI-8968 AstraZeneca COPD Phase II (nati-IL-1R mAb) Wilmington, DE astrazeneca.com MedImmune medimmune.com Gaithersburg, MD NVA237 Novartis Pharmaceuticals COPD Phase III (glycopyrrolate inhalation) East Hanover, NJ novartis.com olodaterol/tiotropium bromide Boehringer Ingelheim Pharmaceuticals COPD Phase III fixed-dose combination Ridgefield, CT boehringer-ingelheim.com OPC-6535 Otsuka America Pharmaceutical COPD Phase II (tetomilast) Rockville, MD otsuka.com PF-03715455 Pfizer COPD Phase I (MAPK-14 inhibitor) New York, NY pfizer.com PH-797804 Pfizer COPD Phase II (p38 MAP kinase inhibitor) New York, NY pfizer.com Prochymal™ Osiris Therapeutics COPD Phase II remestemcel-L Columbia, MD (see also diabetes) osiris.com PT001 Pearl Therapeutics COPD Phase II (glycopyrrolate inhalation aerosol) Redwood City, CA pearltherapeutics.com PT003 Pearl Therapeutics COPD Phase II (glycopyrrolate/formoterol Redwood City, CA pearltherapeutics.com inhalation aerosol) Medicines in Development Older Americans 2013 17
- 18. Medicines in Development for Older Americans Chronic Obstructive Pulmonary Disease (COPD) Product Name Sponsor Indication Development Phase PT005 Pearl Therapeutics COPD Phase II (formoterol inhalation aerosol) Redwood City, CA pearltherapeutics.com PUR118 Pulmatrix COPD Phase I Lexington, MA pulmatrix.com QMF149 Novartis Pharmaceuticals COPD Phase II (indacaterol/mometasome East Hanover, NJ novartis.com fixed-dose combination) QVA149 Novartis Pharmaceuticals COPD Phase III (glycopyrrolate/indacaterol East Hanover, NJ novartis.com fixed-dose combination) salbutamol dry-powder Teva Pharmaceutical COPD Phase II inhalation North Wales, PA tevapharm.com Striverdi Respimat™ Boehringer Ingelheim Pharmaceuticals COPD application submitted olodaterol Ridgefield, CT boehringer-ingelheim.com suvorexant Merck COPD Phase I completed (dual orexin receptor antagonist) Whitehouse Station, NJ merck.com TD-4208 Theravance COPD Phase II (LAMA) South San Francisco, CA theravance.com umeclidinium GlaxoSmithKline COPD Phase III (muscarinic acetylcholine Rsch. Triangle Park, NC gsk.com antagonist) umeclidinium/vilanterol GlaxoSmithKline COPD application submitted fixed-dose combination Rsch. Triangle Park, NC gsk.com vilanterol GlaxoSmithKline COPD Phase III (long-acting beta2 agonist) Rsch. Triangle Park, NC gsk.com Theravance theravance.com South San Francisco, CA vilanterol/fluticasone furoate GlaxoSmithKline COPD application submitted fixed-dose combination Rsch. Triangle Park, NC gsk.com Theravance theravance.com South San Francisco, CA -------------------------------------- ----------------------------------------- COPD (mortality claim) Phase III gsk.com theravance.com 18 Medicines in Development Older Americans 2013
- 19. Medicines in Development for Older Americans Depression Product Name Sponsor Indication Development Phase ABT-436 AbbVie major depressive disorder Phase II (selective vasopressin receptor North Chicago, IL abbvie.com antagonist) ALKS-5461 Alkermes major depressive disorder Phase II (buprenorphine/samidorphan Waltham, MA alkermes.com fixed-dose combination) amitifadine Euthymics major depressive disorder Phase II/III Cambridge, MA euthymics.com ARA 290 Araim Pharmaceuticals depressive disorders Phase II Ossining, NY araim.org armodafinil Teva Pharmaceutical major depressive disorder Phase III Tikvah, Israel tevapharm.com AZD6765 AstraZeneca major depressive disorder Phase II (NMDA receptor antagonist) Wilmington, DE astrazeneca.com BCI-632 BrainCells major depressive disorder Phase I (mGluR2/3 antagonist) San Diego, CA braincellsinc.com BCI-838 BrainCells major depressive disorder Phase I (mGluR2/3 antagonist) San Diego, CA braincellsinc.com BMS-820836 Bristol-Myers Squibb major depressive disorder Phase II (triple reuptake inhibitor) Princeton, NJ bms.com brexpiprazole Otsuka Pharmaceutical adjunct therapy for major Phase III (OPC-34712) Rockville, MD depressive disorder otsuka.com Brintellix™ Lundbeck major depressive disorder application submitted vortioxetine Deerfield, IL lundbeck.com Takeda Pharmaceuticals International takeda.com Deerfield, IL cariprazine Forest Laboratories major depressive disorder Phase III New York, NY frx.com CX157 CeNeRx BioPharma major depressive disorder Phase II completed (monoamine oxidase A inhibitor) Cary, NC cenerx.com DSP-1053 Sunovion Pharmaceuticals depression Phase I (serotonin uptake inhibitor) Marlborough, MA sunovion.com Medicines in Development Older Americans 2013 19
- 20. Medicines in Development for Older Americans Depression Product Name Sponsor Indication Development Phase edivoxetine Eli Lilly major depressive disorder Phase III (LY2216684) Indianapolis, IN lilly.com GLYX-13 Naurex major depressive disorder Phase II Evanston, IL naurex.com HT-2157 Dart NeuroScience major depressive disorder Phase I/II (GALR3 antagonist) San Diego, CA dartneuroscience.com JNJ-40411813 Janssen Research Development major depressive disorder with Phase II (mGluR2 modulator) Raritan, NJ anxiety symptoms janssenrnd.com levomilnacipran Forest Laboratories major depressive disorder application submitted (F2695) New York, NY frx.com lisdexamfetamine Shire major depressive disorder Phase III Wayne, PA shire.com lurasidone Sunovion Pharmaceuticals bipolar depression application submitted Marlborough, MA sunovion.com LY2940094 Eli Lilly major depressive disorder Phase II Indianapolis, IN lilly.com mifepristone Corcept Therapeutics psychotic major depression Phase III Menlo Park, CA (Fast Track) corcept.com MK-6096 Merck major depressive disorder Phase II (orexin receptor 1/2 antagonist) Whitehouse Station, NJ merck.com nefiracetam Neuren Pharmaceuticals major depressive disorder Phase II Bethesda, MD post-stroke neurenpharma.com NSI-189 Neuralstem major depressive disorder Phase I (stimulating neurogenesis) Rockville, MD neuralstem.com PNB01 PharmaNeuroBoost severe major depressive disorder Phase III (citalopram/pipamperone Alken, Belgium pharmaneuroboost.com fixed-dose combination) RG1578 Roche major depression disorder Phase II (mGluR2 modulator) Nutley, NJ roche.com RG7090 Roche treatment-resistent depression Phase II (mGluR5 antagonist) Nutley, NJ roche.com RO4995819 Roche major depressive disorder Phase II Nutley, NJ roche.com 20 Medicines in Development Older Americans 2013
- 21. Medicines in Development for Older Americans Depression Product Name Sponsor Indication Development Phase SEP-228432 Sunovion Pharmaceuticals major depressive disorder Phase I Marlborough, MA sunovion.com SKL-DEP SK Biopharmaceuticals major depressive disorder Phase I Seoul, South Korea skbp.com tedatioxetine Lundbeck major depressive disorder Phase II Deerfield, IL lundbeck.com Takeda Pharmaceuticals International takeda.com Deerfield, IL TGBA01AD Fabre-Kramer Pharmaceuticals major depressive disorder Phase II Houston, TX fabrekramer.com Diabetes Product Name Sponsor Indication Development Phase 11ß-HSD1 inhibitor Eli Lilly type 2 diabetes Phase II (LY2523199) Indianapolis, IN lilly.com AC-201 TWi Pharmaceuticals type 2 diabetes Phase II Taipei, Taiwan twipharma.com Afrezza® Mannkind type 1 diabetes, type 2 diabetes Phase III insulin inhalation Valencia, CA mannkindcorp.com albiglutide GlaxoSmithKline type 2 diabetes application submitted Rsch. Triangle Park, NC (see also heart) gsk.com aleglitazar Genentech type 2 diabetes Phase III South San Francisco, CA gene.com Roche roche.com Nutley, NJ alpha-1 antitrypsin (AAT) OmniBio Pharmaceuticals type 1 diabetes Phase I/II Greenwood Village, CO omnibiopharma.com AMG 151 Amgen type 2 diabetes Phase II Thousand Oaks, CA amgen.com anagliptin Kowa Research Institute type 2 diabetes Phase II (DPP-4 inhibitor) Morrisville, NC kowaus.com Medicines in Development Older Americans 2013 21
- 22. Medicines in Development for Older Americans Diabetes Product Name Sponsor Indication Development Phase analog insulin-PH20 Halozyme type 1 diabetes, type 2 diabetes Phase II San Diego, CA halozyme.com ARI-2243 Arisaph Pharmaceuticals type 2 diabetes Phase I (DPP-4 inhibitor) Boston, MA arisaph.com BGP-15 N-Gene Research Laboratories type 2 diabetes Phase II New York, NY ngene.us BI-135585 Boehringer Ingelheim Pharmaceuticals type 2 diabetes Phase I (11ß-HSD1 inhibitor) Ridgefield, CT boehringer-ingelheim.com BIOD-123 Biodel type 1 diabetes Phase II (ultra-rapid-acting recombinant Danbury, CT biodel.com human insulin) BIOD-125 Biodel type 1 diabetes Phase I completed (ultra-rapid-acting recombinant Danbury, CT biodel.com human insulin) BIOD-238 Biodel diabetes Phase I (ultra-rapid-acting recombinant Danbury, CT biodel.com human insulin) BIOD-250 Biodel diabetes Phase I (ultra-rapid-acting recombinant Danbury, CT biodel.com human insulin) BMS-770767 Bristol-Myers Squibb type 2 diabetes Phase II (11ßHSD inhibitor) Princeton, NJ bms.com canagliflozin Janssen Research Development type 2 diabetes application submitted Raritan, NJ janssenrnd.com canagliflozin/metformin Janssen Research Development type 2 diabetes Phase III extended-release formulation Raritan, NJ janssenrnd.com canagliflozin/metformin Janssen Research Development type 2 diabetes application submitted immediate-release formulation Raritan, NJ janssenrnd.com canakinumab Novartis Pharmaceuticals type 2 diabetes Phase II/III (ACZ885) East Hanover, NJ (see also arthritis) novartis.com CAT 1004 Catabasis Pharmaceuticals type 2 diabetes Phase I Cambridge, MA catabasis.com 22 Medicines in Development Older Americans 2013
- 23. Medicines in Development for Older Americans Diabetes Product Name Sponsor Indication Development Phase cetilistat Norgine type 2 diabetes in clinically obese Phase II Amsterdam, Netherlands patients norgine.com CJC-1134-PC ConjuChem type 2 diabetes Phase II Los Angeles, CA conjuchem.com COV 096 Pfizer type 2 diabetes Phase I (PF-04856883) New York, NY pfizer.com dapagliflozin AstraZeneca type 2 diabetes application submitted Wilmington, DE astrazeneca.com Bristol-Myers Squibb bms.com Princeton, NJ dapagliflozin/metformin AstraZeneca type 2 diabetes Phase III fixed-dose combination Wilmington, DE astrazeneca.com Bristol-Myers Squibb bms.com Princeton, NJ DC-9703 Obio Pharmaceutical type 2 diabetes Phase I Kowloon, Hong Kong obiopharma.com diabetes medicine Eli Lilly diabetes Phase I (biologic) Indianapolis, IN lilly.com diabetes medicine Eli Lilly diabetes Phase I (biologic) Indianapolis, IN lilly.com diabetes medicine Eli Lilly diabetes Phase I (biologic) Indianapolis, IN lilly.com diabetes medicine Eli Lilly diabetes Phase I (biologic) Indianapolis, IN lilly.com diabetes medicine Eli Lilly diabetes Phase I (NCE) Indianapolis, IN lilly.com diabetes medicine Eli Lilly diabetes Phase I (NCE) Indianapolis, IN lilly.com DiaPep277® Andromeda Biotech type 1 diabetes Phase III Yavne, Israel andromedabio.com DS-7250 Daiichi Sankyo diabetes Phase I (DGAT1 inhibitor) Parsippany, NJ dsi.com Medicines in Development Older Americans 2013 23
- 24. Medicines in Development for Older Americans Diabetes Product Name Sponsor Indication Development Phase DSP-8658 Sunovion Pharmaceuticals type 2 diabetes Phase I (PPAR alpha/gamma agonist) Marlborough, MA (see also Alzheimer’s) sunovion.com dulaglutide Eli Lilly type 2 diabetes Phase III (LY2189265) Indianapolis, IN lilly.com EGT-0001442 Theracos type 2 diabetes Phase II (SGLT2 inhibitor) Marlborough, MA theracos.com EGT-0001474 Theracos type 2 diabetes Phase I completed (SGLT2 inhibitor) Marlborough, MA theracos.com empagliflozin Boehringer Ingelheim Pharmaceuticals type 2 diabetes Phase III (BI-10773) Ridgefield, CT boehringer-ingelheim.com Eli Lilly lilly.com Indianapolis, IN empagliflozin/linagliptin Boehringer Ingelheim Pharmaceuticals type 2 diabetes Phase III fixed-dose combination Ridgefield, CT boehringer-ingelheim.com Eli Lilly lilly.com Indianapolis, IN empagliflozin/metformin Boehringer Ingelheim Pharmaceuticals type 2 diabetes Phase I fixed-dose combination Ridgefield, CT boehringer-ingelheim.com Eli Lilly lilly.com Indianapolis, IN emricasan Conatus Pharmaceuticals type 1 diabetes Phase I/II San Diego, CA conatuspharma.com enclomifene Repros Therapeutics type 2 diabetes in men with Phase II The Woodlands, TX secondary hypogonadism reprosrx.com fasiglifam Takeda Pharmaceuticals International type 2 diabetes Phase III (TAK-875) Deerfield, IL takeda.com GK1-399 Forest Laboratories type 2 diabetes Phase II (glucokinase activator) New York, NY frx.com glimepiride extended-release/ GlaxoSmithKline type 2 diabetes Phase I metformin extended-release Rsch. Triangle Park, NC gsk.com fixed-dose combination glucagon-R antagonist Eli Lilly type 2 diabetes Phase II (LY2409021) Indianapolis, IN lilly.com GPR40 Bristol-Myers Squibb type 2 diabetes in clinical trials Princeton, NJ bms.com 24 Medicines in Development Older Americans 2013
