EM Spectrum, de Broglie, PE Notes
•
1 recomendación•725 vistas
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
Limitations OF Classical Physics and Birth Of Quantum Mechanics

Limitations OF Classical Physics and Birth Of Quantum Mechanics
CHAPTER 5 Wave Properties of Matter and Quantum Mechanics I

CHAPTER 5 Wave Properties of Matter and Quantum Mechanics I
Destacado
Destacado (13)
B.sc (microbiology and biotechnology and biochemistry) ii inorganic chemistry...

B.sc (microbiology and biotechnology and biochemistry) ii inorganic chemistry...
Physics 2 notes: WAVES ONLY- Notes on the difference between longitudinal, t...

Physics 2 notes: WAVES ONLY- Notes on the difference between longitudinal, t...
14. física moderna. fundamentos de mecánica cuántica

14. física moderna. fundamentos de mecánica cuántica
Similar a EM Spectrum, de Broglie, PE Notes
Similar a EM Spectrum, de Broglie, PE Notes (20)
Beyond bohr de broglie and heisenberg for universe to atom module cfi

Beyond bohr de broglie and heisenberg for universe to atom module cfi
Diploma sem 2 applied science physics-unit 5-chap-2 photoelectric effect

Diploma sem 2 applied science physics-unit 5-chap-2 photoelectric effect
Más de dwsutherland
Más de dwsutherland (20)
Último
Último (20)
Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...

Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...
Boost PC performance: How more available memory can improve productivity

Boost PC performance: How more available memory can improve productivity
Automating Google Workspace (GWS) & more with Apps Script

Automating Google Workspace (GWS) & more with Apps Script
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
Handwritten Text Recognition for manuscripts and early printed texts

Handwritten Text Recognition for manuscripts and early printed texts
The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf

The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf
Presentation on how to chat with PDF using ChatGPT code interpreter

Presentation on how to chat with PDF using ChatGPT code interpreter
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
Unblocking The Main Thread Solving ANRs and Frozen Frames

Unblocking The Main Thread Solving ANRs and Frozen Frames
Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...

Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...
Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...

Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...
TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments

TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments
The 7 Things I Know About Cyber Security After 25 Years | April 2024

The 7 Things I Know About Cyber Security After 25 Years | April 2024
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
EM Spectrum, de Broglie, PE Notes
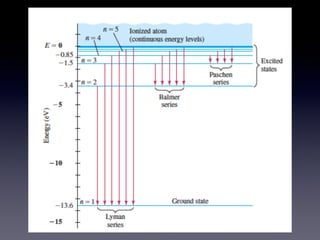
- 4. de Broglie • Pictured the electron in its circular orbit as a particle wave • Can produce "standing waves" under resonant conditions • Developed the idea that a particle with mass, m, and a velocity, v, has a wavelength associated with it => de Broglie Wavelength
- 5. de Broglie
- 6. de Broglie
- 7. de Broglie
- 8. Schrödinger • Used de Broglie wavelength to create a quantum theory based on waves • Did not keep the "orbits" • The wave/particle model cannot determine the location and momentum of an electron at the same time • The quantum model predicts the probability that an e- is at a specific location
- 9. Heisenberg Uncertainty Principle • Can only determine the location or the momentum (velocity) of the particle - not both at the same time!
- 11. Photons and Photoelectric Effect
- 12. Photoelectric Effect • Metal is illuminated by electromagnetic radiation • Energy that is absorbed near the surface can free electrons, causing e's to fly off • Released electrons are called photoelectrons
- 13. Wave theory predicts the following: • Significant time delay between the illumination and ejection - build up of KE to free e-'s • Increasing the intensity of light = cause electrons to leave with greater KE • Photoelectrons would be released regardless of frequency of light, as long as the intensity was great enough.... But these are FALSE!
- 14. Photoelectric Effect Findings • Photons were ejected immediately • Increasing the intensity did not change the KE although more e-'s were ejected, KE does not increase. • If the frequency fell below a threshold (specific for each metal), no photoelectrons would be ejected, regardless of intensity! • If the frequency increases above the threshold, KE increases linearly
- 16. PE Effect - Math • Threshold Frequency • Work Function - the minimum amount of energy required on a metal surface to eject an electron • How are these two related?
- 17. PE Effect - Math