South Korea medical device approval chart - Emergo
•
2 recomendaciones•4,941 vistas
Easy to understand chart describes the KFDA medical device registration process in South Korea.
Denunciar
Compartir
Denunciar
Compartir
Descargar para leer sin conexión
Recomendados
Recomendados
Más contenido relacionado
La actualidad más candente
La actualidad más candente (20)
Europe CE Marking for medical devices under new MDR

Europe CE Marking for medical devices under new MDR
Europe IVD medical registration and approval chart - EMERGO

Europe IVD medical registration and approval chart - EMERGO
Device registration and listing of medical devices on the US market

Device registration and listing of medical devices on the US market
TGA Presentation: Medical Devices - Manufacturer Evidence and applications fo...

TGA Presentation: Medical Devices - Manufacturer Evidence and applications fo...
Classification of In Vitro Diagnostic Devices per FDA and IVDR Rules

Classification of In Vitro Diagnostic Devices per FDA and IVDR Rules
Regulation of In Vitro Diagnostic Medical Devices - Transition to the New IVD...

Regulation of In Vitro Diagnostic Medical Devices - Transition to the New IVD...
Similar a South Korea medical device approval chart - Emergo
Similar a South Korea medical device approval chart - Emergo (20)
CE Marking , FDA Approval and Associated Regulations for Wellness

CE Marking , FDA Approval and Associated Regulations for Wellness
Strategies for Device Approval in China, India, South Korea and Australia

Strategies for Device Approval in China, India, South Korea and Australia
Scheme for Notifying Examiner of Electronic Evidence Under section 79A of the...

Scheme for Notifying Examiner of Electronic Evidence Under section 79A of the...
BIS REGISTRATION FOR WEBCAM (FINISHED PRODUCT) – IS 616 : 2017

BIS REGISTRATION FOR WEBCAM (FINISHED PRODUCT) – IS 616 : 2017
Key Differences between Medical Device User Fees and Establishment Registrati...

Key Differences between Medical Device User Fees and Establishment Registrati...
Último
Hello, Guys welcome to Manalifun Goa Escort service. Are you want Top call girls in Goa at just ₹10000 then no further anywhere because we have a large number of local beautiful girls. We are a genuine platform to provide unlimited classification escort ads service without any commission. 9316020077
Here many Goa Independent call girls and ladies, publish their ads. Our call girl in Goa is well-known for real sexual fun in Goa. We are not allow any prostitute to work here without checking the details, Firstly all ads check by our team then we publish them here. So don’t hesitate to book Low rate call girls in Goa. 9316020077
Goa call girls: A real wonder in Goa
Who are the best Goa Escort Service provider for Goa call girls
High-Class call girls in Goa escort service for 100% Satisfaction
Choose a trusted call girl service in Goa with Us +91-9316020077
Goa Escorts Provide 100% Client Satisfaction
How Our Goa Call Girls Are Perfect For Instant Satisfaction
100% Guaranteed Goa call girls will make you excited
How to Find Cheap Call Girls in Goa
Our Reliable Escort Service in Goa Local Areas
Goa Escorts (cheap escort service in Goa)
Rate Chart of Goa call girls, (call girl Rate in Goa)
5-star hotel For Goa call girls service
Call girls in Goa are the ideal sex partner for you
BOOK YOUR FAVORITE Goa CALL GIRLS SERVICE WITH US CALL! US NOW~ 9316020077
Best way to Hire call girls in Goa
What’s the cost of escort service in Goa
North Goa Call Girls
Location :-
Baga , Caclangute , Candolim , Anjuna , Panaji Arpora , Vagator , Morjim , Siolim , Mandrem , Arambol , etc.
Vasco , Bambolim , Madgaon, Colva , Etc9316020077📞Goa Call Girls Numbers, Call Girls Whatsapp Numbers Goa

9316020077📞Goa Call Girls Numbers, Call Girls Whatsapp Numbers Goarussian goa call girl and escorts service
Hello, Guys welcome to Manalifun Goa Escort service. Are you want Top call girls in Goa at just ₹10000 then no further anywhere because we have a large number of local beautiful girls. We are a genuine platform to provide unlimited classification escort ads service without any commission. 9316020077
Here many Goa Independent call girls and ladies, publish their ads. Our call girl in Goa is well-known for real sexual fun in Goa. We are not allow any prostitute to work here without checking the details, Firstly all ads check by our team then we publish them here. So don’t hesitate to book Low rate call girls in Goa. 9316020077
Goa call girls: A real wonder in Goa
Who are the best Goa Escort Service provider for Goa call girls
High-Class call girls in Goa escort service for 100% Satisfaction
Choose a trusted call girl service in Goa with Us +91-9316020077
Goa Escorts Provide 100% Client Satisfaction
How Our Goa Call Girls Are Perfect For Instant Satisfaction
100% Guaranteed Goa call girls will make you excited
How to Find Cheap Call Girls in Goa
Our Reliable Escort Service in Goa Local Areas
Goa Escorts (cheap escort service in Goa)
Rate Chart of Goa call girls, (call girl Rate in Goa)
5-star hotel For Goa call girls service
Call girls in Goa are the ideal sex partner for you
BOOK YOUR FAVORITE Goa CALL GIRLS SERVICE WITH US CALL! US NOW~ 9316020077
Best way to Hire call girls in Goa
What’s the cost of escort service in Goa
North Goa Call Girls
Location :-
Baga , Caclangute , Candolim , Anjuna , Panaji Arpora , Vagator , Morjim , Siolim , Mandrem , Arambol , etc.
Vasco , Bambolim , Madgaon, Colva , EtcCall Girls Service In Goa 💋 9316020077💋 Goa Call Girls By Russian Call Girl...

Call Girls Service In Goa 💋 9316020077💋 Goa Call Girls By Russian Call Girl...russian goa call girl and escorts service
❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jalandhar Call Girls Service ☎️
Call Girls In Jalandhar 8264406502 Jalandhar railway station Near Radisson Hotel Jalandhar, Majestic Grand Hotel, Ramada by Wyndham Jalandhar City Centre, Park Plaza Ludhiana, Windsor Fountain, G.T Road Jalandhar escort all Jalandhar service Russian available model female girls in Jalandhar VIP Lo price personal Jalandhar off class call girls payment high profile model and female escort 70% Off On Your First Booking Jalandhar Call Girls Service Cash Payment
Welcome to NehaChopra Jalandhar Call Girl Service, the Trusted call girl agency around. We Offer 70% Discount On Your First Booking For Jalandhar Call Girls Service Cash Payment is available.❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...

❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...chandigarhentertainm
Último (20)
VIP Call Girls Noida Jhanvi 9711199171 Best VIP Call Girls Near Me

VIP Call Girls Noida Jhanvi 9711199171 Best VIP Call Girls Near Me
9316020077📞Goa Call Girls Numbers, Call Girls Whatsapp Numbers Goa

9316020077📞Goa Call Girls Numbers, Call Girls Whatsapp Numbers Goa
Vip Call Girls Makarba 👙 6367187148 👙 Genuine WhatsApp Number for Real Meet

Vip Call Girls Makarba 👙 6367187148 👙 Genuine WhatsApp Number for Real Meet
Call Girls Service In Goa 💋 9316020077💋 Goa Call Girls By Russian Call Girl...

Call Girls Service In Goa 💋 9316020077💋 Goa Call Girls By Russian Call Girl...
ooty Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

ooty Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Call Girls Thane Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Thane Just Call 9907093804 Top Class Call Girl Service Available
Muzaffarpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Muzaffarpur Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Call Girls Service Anantapur 📲 6297143586 Book Now VIP Call Girls in Anantapur

Call Girls Service Anantapur 📲 6297143586 Book Now VIP Call Girls in Anantapur
VIP Call Girls Noida Sia 9711199171 High Class Call Girl Near Me

VIP Call Girls Noida Sia 9711199171 High Class Call Girl Near Me
💚 Punjabi Call Girls In Chandigarh 💯Lucky 🔝8868886958🔝Call Girl In Chandigarh

💚 Punjabi Call Girls In Chandigarh 💯Lucky 🔝8868886958🔝Call Girl In Chandigarh
❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...

❤️Call girls in Jalandhar ☎️9876848877☎️ Call Girl service in Jalandhar☎️ Jal...
Kolkata Call Girls Miss Inaaya ❤️ at @30% discount Everyday Call girl

Kolkata Call Girls Miss Inaaya ❤️ at @30% discount Everyday Call girl
Best Lahore Escorts 😮💨03250114445 || VIP escorts in Lahore

Best Lahore Escorts 😮💨03250114445 || VIP escorts in Lahore
Mangalore Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Mangalore Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Call Girls Hyderabad Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Hyderabad Just Call 9907093804 Top Class Call Girl Service Available
dehradun Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

dehradun Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
raisen Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

raisen Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Tirupati Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

Tirupati Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
Russian Call Girls in Noida Pallavi 9711199171 High Class Call Girl Near Me

Russian Call Girls in Noida Pallavi 9711199171 High Class Call Girl Near Me
neemuch Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet

neemuch Call Girls 👙 6297143586 👙 Genuine WhatsApp Number for Real Meet
South Korea medical device approval chart - Emergo
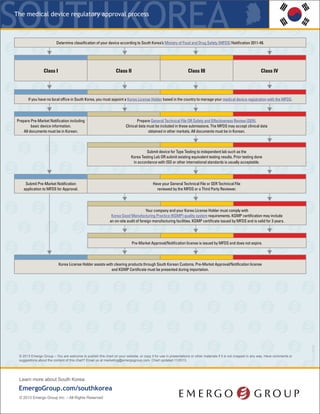
- 1. The regulatory process for medical devices KRSouth Korea * If your device (all classes) is new or does not have a predicate in the South Korean market, then the MFDS will require a Clinical Data Review, formerly Safety and Efficacy Review (SER). Manufacturers will be required to submit Clinical Data with their application and the registration approval timeline and fees will increase accordingly. ** There are approximately 350 Class II devices that MFDS considers “Special Equivalent,” which require a less robust application, i.e. no Technical File, as well as an expedited review by a Third Party Reviewer. *** Prior testing done in accordance with ISO or other international standards will satisfy some testing requirements; however, most manufacturers have to conduct additional testing, including performance testing, to Korea specific product standards. This is a simplified overview of the process. The MFDS may choose to audit your submission and request more documents, which will add time to your approval. © 2015 Emergo – You are welcome to publish this chart on your website, or copy it for use in presentations or other materials if it is not cropped in any way. Have comments or suggestions about the content of this chart? Email us at marketing@emergogroup.com. Chart updated 03/2015. 5040-0315 EmergoGroup.com/south-korea Determine classification of your device based on the device database, regulations provided by South Korea’s Ministry of Food and Drug Safety (MFDS), as well as predicate devices in the South Korean market. If no predicate exists, the device will follow the Clinical Data Review route.* Class I Class II** Class III Class IV Ifyou do not have a local office in South Korea,you must appoint a Korea License Holder based in the countryto manageyour medical device registration with the MFDS. Prepare Pre-Market Notification including basic device information. Prepare registration application in Korean, including technical file, test reports and KGMP certificate. Prepare registration application in Korean, including technical file, test reports and KGMP certificate. Class IV devices require an additional technical file in STED format. Send your device to Korea for testing OR prepare existing equivalent Korea testing results. *** Appoint a qualified importer/distributor to bring your device into South Korea. You are now authorized to begin marketing your device. KLH submits Pre-Market Notification application to the MFDS database. Pay application fees. KLHsubmitsKoreanregistrationdossierto acertifiedThirdPartyReviewer. Application includestechnicalfile,testreports,and KGMPCertificate. AftersuccessfulThird PartyReview,theMFDSwillissuethefinal approval.Payapplicationfees. KLH submits Korean registration dossier to the MFDS for review. Dossier includes the Technical File plus STED Technical File (Class IV), test reports, and KGMP certificate. Pay application fees. Obtain Korea Good Manufacturing Practices (KGMP) certificationthrough an onsite audit. Class II device manufacturing facilities are usuallyaudited byaThird PartyAuditor. KGMPcertification must be renewed every3years. Obtain Korea Good Manufacturing Practices (KGMP) certification through an onsite audit. Class III and IV device manufacturing facilities are audited by a Third Party Auditor and the MFDS. KGMPcertification must be renewed every3years. Class I devices do not require approval, only Notification to the MFDS. The MFDS will publish the device on their website. Notifications do not expire. Full application review conducted. Pre-Market Approval license is issued by the MFDS and published on their website. Registrations do not expire.
- 2. The regulatory process for medical devices EmergoGroup.com/south-korea This is a simplified overview of the process. South Korea’s Ministry of Food and Drug Safety (MFDS) may choose to audit your submission and request more documents, which will add time to your approval. NOTE 1: The time frames shown above are typical for the majority of medical device submissions but assume that your device does not contain animal tissue, medicinal substances or employ entirely novel technology. Your length of approval will depend on the quality and completeness of your technical documentation and how much time you take to address additional information requests from authorities after submission. YOUR SUBMISSION(S) MAY TAKE MORE TIME THAN WHAT IS SHOWN ABOVE. NOTE 2: The device license does not expire, but is subject to reevaluation. Periodically the MFDS will select devices codes to reevaluate as a whole. Typically this does not apply to products that were recently approved under that selected device code, as this process is intended to ensure older products meet the current regulatory requirements. Manufacturers will need to submit documents for reevaluation; documents include but are not limited to: updated vigilance, updated QMS documents, updated approvals from other markets. Announcements of device codes up for reevaluation typically happen in April or May, and manufacturers must submit before the end of the year. Failure to comply with the reevaluation will cause the device license to be revoked. NOTE 3: The device notification (Class I) and license (Class II-IV) does not expire, so no renewal is required. NOTE 4: Our rating of the complexity of the registration process is based on our experience and the opinion of nearly 1,000 QA/RA professionals worldwide who were asked to rate the difficulty of registering a device in each country. The European CE Marking process is considered the mid-point to which all other markets are compared. NOTE 5: Low = Less than US$5000; Midpoint = US$15000-$30000: High = More than US$50000. Overall cost includes registration application fees, product testing, in-country representation, submission preparation consulting and translation of registration documents but not IFU. Costs assume you already have approval for your device in the United States, Europe, Canada or Japan. Does not include cost of implementing or auditing a quality management system, if applicable. * If your device (all classes) is new or does not have a predicate in the South Korean market, then the MFDS will require a Clinical Data Review, formerly Safety and Efficacy Review (SER). Manufacturers will be required to submit Clinical Data with their application and the registration approval timeline and fees will increase accordingly. †† There are approximately 350 Class II devices that MFDS considers “Special Equivalent,” which require a less robust application, i.e. no Technical File, as well as an expedited review by a Third Party Reviewer. © 2015 Emergo – You are welcome to publish this chart on your website, or copy it for use in presentations or other materials if it is not cropped in any way. Have comments or suggestions about the content of this chart? Email us at marketing@emergogroup.com. Chart updated 03/2015. Device classification in South Korea How long you should expect to wait after submission until approval is granted. (See note 1) Validity period for device registrations. (See note 2) Registration renewal should be started this far in advance. (See note 3) Complexity of the registration process for this classification. (See note 4) Overall cost of gaining regulatory approval. (See note 5) CLASS I 1 month Does not expire Not applicable Simple Complex Low High CLASS II*† 4-6 months Does not expire Not applicable Simple Complex Low High CLASS III* 6-10 months Does not expire Not applicable Simple Complex Low High CLASS IV* 6-10 months Does not expire Not applicable Simple Complex Low High KRSouth Korea
