Pendahuluan biologi-sel
- 1. BIOLOGI SEL: PENDAHULUAN BISEL07-SITH/ITB-MIT/IR 1
- 2. Sejarah perkembangan • Robert Hooke : sel mati : sel dari gabus • Anton van Leeuwenhoek : sel hidup • Matthias Schleiden : sel pada tumbuhan • Theodor Schwann (1839): Teori sel – Semua organisma terdiri dari satu atau lebih sel – Sel : unit struktural hidup • Schleiden & Schwann : sel dapat berasal dari materi-materi nonselular • Rudolf Virchow (1855) : sel berasal dari pembelahan sel yang sudah ada sebelumnya • Penggunaan sel dalam penelitian in vitro : HeLa (sel kanker manusia) – George Gey BISEL07-SITH/ITB-MIT/IR 2 (1951)
- 4. Karakteristik sel • Sel sangat kompleks – Molekul-molekul sederhana – kompleks organel sel misalnya C, H, O, N, S, P asam amino protein misalnya salah satu komponen dalam mitokondria yang merupakan organel dari sel BISEL07-SITH/ITB-MIT/IR 4
- 5. Karakteristik sel • Sel memiliki informasi genetik – Gen : blueprint untuk struktur sel, seluruh aktivitas dan fungsi sel • Sel dapat ber-reproduksi BISEL07-SITH/ITB-MIT/IR 5
- 6. Karakteristik sel • Sel memperoleh dan menggunakan energi • Sel melakukan metabolisme sel BISEL07-SITH/ITB-MIT/IR 6
- 7. Karakteristik sel • Terdapat suatu aktivitas mekanis dalam sel yang dinamis – Misalnya perubahan bentuk sel akibat aksi dari protein-protein dalam sitoplasma • Sel dapat memberi respons terhadap suatu stimulus – Reseptor hormon, reseptor faktor tumbuh, reseptor matriks ekstraselular, atau reseptor lainnya (G) – Respons : misalnya metabolisme sel, proliferasi sel atau gerakan sel Istirahat teraktivasi retraksi BISEL07-SITH/ITB-MIT/IR 7
- 8. Karakteristik sel • Sel mampu mengatur diri sendiri (self regulation) – Misalnya pengaturan siklus sel BISEL07-SITH/ITB-MIT/IR 8
- 9. Prokaryot -Eukaryot BISEL07-SITH/ITB-MIT/IR 9
- 10. Persamaan antara eukaryot dengan prokaryot: • konstruksi membran plasma sama BISEL07-SITH/ITB-MIT/IR 10
- 11. Persamaan antara eukaryot dengan prokaryot • informasi genetik dikode oleh DNA, dengan kode genetic yang identik • mekanisme transkripsi dan translasi Eukaryotes Prokaryotes BISEL07-SITH/ITB-MIT/IR 11
- 12. Persamaan antara eukaryot dengan prokaryot: • reaksi metabolisme • apparatus yang sama untuk konversi energi kimiawi – prokaryot membran plasma – eukaryot membran mitokondria BISEL07-SITH/ITB-MIT/IR 12
- 13. Persamaan antara eukaryot dengan prokaryot: • mekanisme fotosintesis yang sama (tumbuhan – sianobakteri) • mekanisme sintesa dan penyisipan protein membran • konstruksi proteosom yang sama (archaebacteria dengan eukaryot) BISEL07-SITH/ITB-MIT/IR 13
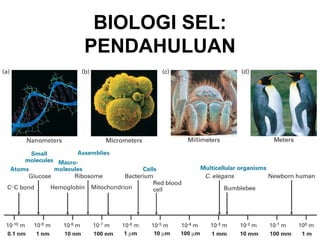
- 14. Perbedaan antara organisme prokaryot dengan eukaryot Prokaryot Eukaryot Organisme Bakteri, Protista, jamur, cyanobakteri tumbuhan dan hewan Ukuran sel Umumnya 1-10 Umumnya 5-100 μm μm Metabolisme Anaerobic atau Aerobik aerobik Organel Sedikit Mitokondria, kloroplas, retikulum endoplasma, dll Inti Tidak ada Ada DNA DNA sirkular DNA linier dan sangat dalam sitoplasma panjang, memiliki daerah yang dikode (ekson) dan tidak dikode /intron (sangat banyak); berada dalam BISEL07-SITH/ITB-MIT/IR inti 14
- 15. BISEL07-SITH/ITB-MIT/IR 15
- 16. Perbedaan antara organisme prokaryot dengan eukaryot Prokaryot Eukaryot RNA dan protein RNA dan protein disintesis pada RNA disintesis dan diproses di inti ruang yang sama Protein disintesis di sitoplasma Sitoplasma Tidak mengandung sitoskeleton, Dalam sitoplasma terdapat sitoskeleton tidak ada aliran sitoplasma dalam : filamen-filamen protein, ada aliran sel, tidak ada endositosis dan sitoplasma dalam sel, ada endositosis eksositosis dan eksositosis BISEL07-SITH/ITB-MIT/IR 16
- 17. Perbedaan antara organisme prokaryot dengan eukaryot Prokaryot Eukaryot Pembelahan sel Kromosom ditarik dengan cara Kromosom ditarik apparatus mitosis pelekatan pada membran plasma (komponen sitoskeleton) Organisasi sel Umumnya uniselular Umumnya multiselular, dan terjadi proses diferensiasi / spesialisasi sel BISEL07-SITH/ITB-MIT/IR 17
- 18. Virus – membawa informasi genetic berupa rantai tunggal atau ganda RNA atau DNA – Materi genetiknya mengkode : • Protein kapsul / kapsid – aktif jika berada pada sel hidup BISEL07-SITH/ITB-MIT/IR 18
- 19. BISEL07-SITH/ITB-MIT/IR 19
- 20. Bioenergetika BISEL07-SITH/ITB-MIT/IR 20
- 21. The Chemistry of Life: A network of metabolic pathways • Cell metabolism can be compared to an elaborate road map of the thousands of chemical reactions that occur in the cell It is an intricate network of metabolic pathways BISEL07-SITH/ITB-MIT/IR 21
- 22. • Catabolic pathways: They release energy by breaking down complex molecules to simpler compounds – A major catabolic pathway found in a cell is respiration which breaks down sugar glucose and other fuels into carbon dioxide and water with release of energy C6H12O6 + 6O2 6CO2 + 6H2O + Energy • Anabolic pathways: Build complex molecules from simpler ones by consuming energy e.g. Photosynthesis in plants 6CO2 + 6H2O + Light energy C6H12O6 + 6O2 + 6H2O BISEL07-SITH/ITB-MIT/IR 22
- 23. • Organisms Transform Energy: – Energy: The capacity to do work • Kinetic energy: The energy of motion possessed by all moving objects e.g. water gushing through a dam turns turbines • Potential energy: Energy that matter possesses because of its location or structure Chemical energy stored in Water behind dams has molecules as a result of the potential energy because arrangement of the atoms in of altitude these molecules • Bioenergetics – The study of how organisms manage their energy resources – to maintain its high level of activity, a cell must acquire & expend energy BISEL07-SITH/ITB-MIT/IR 23
- 24. Conversion of Energy from one form to the other: • Thermodynamics - study of the changes in energy that accompany events in the Universe • Two laws of Thermodynamics BISEL07-SITH/ITB-MIT/IR 24
- 25. The First Law of Thermodynamics • energy can be neither created nor destroyed (Law of Conservation of Energy); total energy in Universe remains constant (regardless of transduction process) – Energy can, however, be transduced - burning fuel, polysaccharide breakdown, photosynthesis • Several organism communities are independent of photosynthesis – communities residing in hydrothermal vents on ocean floor; depends on energy obtained by bacterial chemosynthesis • Some animals (fireflies, luminous fish) convert chemical energy back into light • ΔE = Q – W, where Q = heat energy & W = work energy Reactions that result in heat lost to the environment are called exothermic; those that result in heat gained from the environment are called endothermic BISEL07-SITH/ITB-MIT/IR 25
- 26. Couple of terms • System: Is used to denote the matter under study and refer to the rest of the universe- everything outside the systems the surroundings 1. Closed system: e.g. a liquid in a thermos bottle is isolated from its surroundings 2. Open system: Energy (&often matter) can be transferred between the system and its surroundings e.g. organisms • Entropy: A measure of disorder or randomness • Free energy: Is the portion of a system’s energy that can perform work when temperature is uniform through out the system BISEL07-SITH/ITB-MIT/IR 26
- 27. The Second Law of Thermodynamics • Every energy transfer or transformation increases the entropy of the universe (no machine is 100% efficient which would be necessary) • Some energy is inevitably lost as machine works (same is true of living organism) • car chemical energy (gasoline) converted to kinetic energy + the disorder of its surroundings will increase in the form of heat and small molecules that are the breakdown products of gasoline BISEL07-SITH/ITB-MIT/IR 27
- 28. • Together the 1st & 2nd laws of thermodynamics show that the energy of the universe is constant, but that entropy continues to increase toward a maximum • Gibbs combined concepts inherent in 1st & 2nd Laws to get equation: ΔH = ΔG + TΔS where: 1. ΔG is the change in free energy (the change during a process in energy available to do work) 2. ΔH - change in enthalpy (total energy content of system; equivalent to ΔE for our purposes) 3. T - absolute temperature (°K; °K = °C + 273) 4. ΔS - change in entropy of system BISEL07-SITH/ITB-MIT/IR 28
- 29. • Rearrange to ΔG = ΔH - TΔS - can predict direction in which process will proceed & the extent to which the process will occur 1. ΔG size shows the maximum amount of energy that can be passed on for use in another process 2. Spontaneous process has -ΔG (exergonic) & proceeds toward state of lower free energy; such a process is thermodynamically favored 3. Non-spontaneous process, +ΔG (endergonic); cannot occur spontaneously; it is thermodynamically unfavorable; make it go by coupling to high -ΔG (energy-releasing) reaction BISEL07-SITH/ITB-MIT/IR 29
- 30. ATP: Adenosine Triphosphate • An important renewable high energy compound that powers cellular work • ATP hydrolysis is used to drive most cellular endergonic processes A. ATP is used for diverse processes because its terminal phosphate group can be transferred to a variety of different types of molecules (amino acids, lipids, sugars, & proteins) B. In most coupled reactions, phosphate group is transferred in initial step from ATP to one of above acceptors & is subsequently removed in second step BISEL07-SITH/ITB-MIT/IR 30
- 31. Enzymes: Biocatalysts • A catalyst is a chemical agent that changes the rate of reaction without being consumed by the reaction • An enzyme is a catalytic protein – Enzymes are substrate-specific (key-lock relationship) – Enzymes are sensitive to temperature, pH and to some chemicals • Some Enzymes need co-factors/coenzymes to function BISEL07-SITH/ITB-MIT/IR 31
- 32. BISEL07-SITH/ITB-MIT/IR 32
- 33. Enzymes: Biocatalysts • Substrates can compete with other substrates to bind on the same position of the same enzyme interrupt the reaction • Enzymes can be inhibited by the addition of inhibitors BISEL07-SITH/ITB-MIT/IR 33
- 34. Enzymes: Biocatalysts • Feed back inhibition of enzymes: Feed inhibition is the switching off of a metabolic pathway by its end product which acts as an inhibitor of an enzyme within the pathway BISEL07-SITH/ITB-MIT/IR 34
- 35. • ATP formed 2 ways in cell: – oxidative phosphorylation inner membrane of mitochondria – substrate-level phosphorylation • Oxidative phosphorylation - dehydrogenases move 2 electrons & proton to NAD+ to make NADH 1. High energy NADH donates electrons to other molecules at electron transport (ET) chain 2. Because NADH transfers electrons so readily, it is said to have high electron transfer potential 3. As electron travels down ET system, it loses energy used to make ATP & is added to O2 to make H2O • Substrate-level phosphorylation - phosphate group moved from a substrate to ADP ATP 1. ATP formation is not that endergonic, formation of other molecules is more endergonic 2. Such molecules can donate their phosphates to ADP to make ATP BISEL07-SITH/ITB-MIT/IR 35