Pharma Focus Asia Campbell
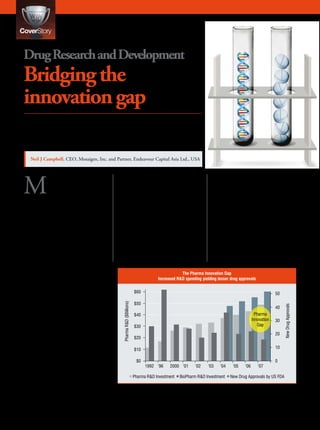
- 1. Drug Research and Development Bridging the innovation gap What needs to be done to improve or change the R&D productivity of the pharmaceutical industry? Is biotech consolidation the answer, and if not, what is? Neil J Campbell, CEO, Mosaigen, Inc. and Partner, Endeavour Capital Asia Ltd., USA M any think that 2007 may improve upon the natural biological risky development changes to the be regarded as the year properties and deliver the compounds parent compound. These new deriva- that Big Pharma went be- in a format that modern humans could tives of a proven drug or family of yond merely embracing biotechnol- take and comply with. drugs have become commonplace and ogy, but decided that consolidation The advent of small molecule have defined the scope of the large with its biologics breathren may well drugs came about because of their pharmaceuticals to this day. be the answer to the many problems availability for discovery, develop- According to analysts, small currently plagueing them. Over the ment and their ability to provide for molecules still dominate the overall past couple of decades, Big Pharma “incremental” development strategies. number of new drug candidates being has consolidated with other Big and These incremental strategies of creat- developed with 135 out of 254 mid-size Pharma companies in a so ing synthetic molecules, in what we (53.1%) entering the Phase I human called Pharma-Pharma consolidation. now call medicinal chemistry, allowed clinical trial process in 2007. These This Pharma-Pharma consolidation for smaller molecule drugs and less figures are based on the records of occurred several times and with it brought about ever-larger companies The Pharma Innovation Gap with even-greater drug pipelines. The Increased R&D spending yielding lesser drug approvals goal was to acquire pipelines from rival companies to boost revenues and, $60 50 in some cases, scientific capabilities $50 Pharma R&D ($Billions) in key growth disease areas. But this New Drug Approvals 40 approach eventually would not be Pharma $40 sustainable as the primary means for Innovation 30 new drug discovery and development. Gap $30 Times have changed. 20 $20 Perspectives on the drug industry 10 $10 The pharmaceutical industry, as we know it today, can be traced back to 0 $0 about 150 years when the source of 1992 ‘96 2000 ‘01 ‘02 ‘03 ‘04 ‘05 ‘06 ‘07 pharmaceuticals was from nature. Pharma R&D Investment BioPharm R&D Investment New Drug Approvals by US FDA Plants and organisms played a vital role in establishing the mainstay of Sources: Pharmaceutical Research and Manufacturers (PhRMA) Annual Report 2007; Burill & Company Figure 1 drug pipelines. The strategy was to Report 2003; PhRMA Annual Member Survey, 2007: US Food & Drug Administration Databases. 12 P h a r m a F o c u s a si a ISSUE - 6 2008
- 2. S t r at e g y Investigation of New Drug Applica- discoveries that were occurring in the expirations in 2006 and is expected to tions (INDs) submitted to the US FDA late 1970s in the biology and chemistry lose another US$ 12 billion in 2007, and announced by drug companies disciplines. These discoveries quickly according to IMS Health. But biotechs from around the world. Although this grew into a nascent industry that don’t have this problem, at least not may sound impressive for pre-clinical would change the way drugs would yet. They don’t have to compete with candidates entering Phase I, according be discovered, designed, developed “biogenerics” because the USFDA to the latest statistics from the Tufts and administered. With the advent of hasn’t created a system for regulating University Drug Development Study the discovery of DNA and the abil- them, which is a requirement for drug and the trade association of large phar- ity to manipulate it in the 1970s and companies to get their products onto maceuticals companies, Pharmaceu- 1980s, came a new science of genetic the market. Europe has proposals, but tical Research and Manufacturers of engineering that promised to fulfil the implementation may prove to be more America (PhRMA), the drug attrition shortcomings of the pharma industry. difficult than originally thought. rates are still one in ten of making it The commercial aspect of the Biotech Another issue that is plaguing Big to that market: a mere 25 drugs out of industry started out slowly in the early Pharma is the increasing cost to market 254 entered Phase I in 2007. 1980s, but has become one of the fast- drugs to the healthcare consumers. In The Big Pharma spent over US$ est growing and strategically important a January 2008 published study in 55.2 billion in 2006 (2007 estimates industries of the 21st century. the Public Library of Science (PLoS), are over US$ 63 billion) on drug Marc-Andre Gagnon and Joel Lexchin R&D a 100% increase over the US$ conducted a study that showed that Big Issues facing the pharmaceutical industry 26 billion they spent in the year 2000 Pharma spent almost twice as much on with a declining number of drug Big Pharma lost US$ 14 billion marketing and promoting their drugs approvals after that according to the worth of annual drug sales to patent than on R&D. The pair analysed data PhRMA report released in 2007. This from market research companies IMS so-called pharma innovation gap is and CAM and found that Big Pharma Issues facing the Big huge and getting larger each year. spent US$ 57.5 billion on promotional Pharma companies So why still focus on the small activities in 2004. By comparison, molecule approach? Although not spending on pharmaceutical R&D in Declining productivity with small revolutionary, it provides a limited the US was US$ 31.5 billion in the molecule programs source of continuing drug candidates same year, according to a report by the Increased costs and longer clinical along with new and extended forms National Science Foundation. development timelines of intellectual property protection. Additional regulatory requirements for Combining small molecule pipeline So, why biotech? safety and efficacy during trials and strategies with other drug product / Biotech drugs or biopharmaceuti- post-approval formulation strategies will give greater cals are made out of living biological Spiraling healthcare costs and pressures on drug pricing leverage to the research and develop- compounds and provide some attrac- ment efforts of these larger pharma- tive alternatives to synthetic small Expiration of drug patents of blockbuster drugs increasing over next ceutical companies. molecules that create traditional several years If it weren’t for a lack of innova- pharmaceuticals today. Increased generic drugs coming to tion on the part of Big Pharma compa- Although there is strong regula- market replacing many off-patent nies, the Biotechnology industry as we tory and political support in Europe blockbusters know it today would probably have not for Biosimiliars (biological generics) Acquisition of biopharmaceutical existed as an industry, but would have and strong resistance to date in the US companies to bolster science been another chapter in the evolu- (where there is still predominance with capabilities and drug pipelines tion of Big Pharma. We all know that the most drugs being developed in the Costs to market drugs to consumers large organisations struggle with crea- US) with the FDA, the complexities are exceeding the costs of drug R&D; this puts companies at risk of not tivity, entrepreneurship and original of redesign, manufacturing and gener- directing enough resources to develop- thinking. But large organisations can ating economies of scale in produc- ing drugs. The current Direct-to-Con- thrive on execution of ideas (sales and tion costs could be some obstacles for sumer marketing strategy is creating marketing), and the later-stage devel- certain biosimiliars than others. a larger problem as less drugs are opment of products (manufacturing). Big Biotechs can provide new developed and more are coming off patent. Spending should be in R&D, A creative and original thinking scientific capabilities, knowledge in new not promotion of existing drugs. movement began out of the seminal drug classes and revenue generation w w w . p h a r m a f o c u s a s i a . c o m 13
- 3. realising the importance of having Worldwide Biopharmaceutical Development both generic and ethical / prescrip- 3000 tion drugs in their marketing efforts to Number of Compounds in Development improve growth and revenues. United States 104% Increase in US 2500 Because taking on biopharma- (Clincal - Pre-registered) ceuticals means learning and hiring a 2000 large amount of people with a differ- ent set of skills and knowledge can 1500 be daunting for single in-licensing or Rest of World acquisition scenarios, it may be better EU 1000 to take on a complete company with larger capabilities that could be inte- Japan 500 grated well with a Big Pharma. Some 1997 1999 2001 2002 2005 examples from 2007 are the US biotech, MedImmune’s, acquisi- Sources: Pharma Annual Report 2007 Figure 2 tion for US$ 1.6 billion by Astra- Note: Comparisions were completed in June of each year. Some compounds will be at different phases for different indication: Adis R&D Insight Database, customized run in December 2005 Zeneca Plc to integrate with its prior acquisition of Cambridge Antibody Biopharmaceuticals can be attractive for the Technologies (CAT) of the UK to form a very large and capable founda- following reasons tion for the creation of a biotechnol- ogy division. AstraZeneca also added The Biotech industry is research intensive and can provide for better knowledge of the UK firm, Arrow Therapeutics to disease pathways and drug mechanisms thereby possibly reducing risks with safety expand their original science in infec- and efficacy tious diseases along with drug candi- The biological drug candidates can be specifically engineered to target particular dates for development. sources in the body in a more directed and personalised approach, hopefully providing Others are taking on more original better odds of achieving approval with better responding populations thinking and entrepreneurship with a The biological drugs can provide for very complex and expansionary strategies for long-term view of drug development patent protection by setting up incubators with an idea Strategies for developing generic biotech drugs will be harder to do from both a of turning them into scientific centres regulatory and technical standpoint. of excellence. Pfizer announced in late 2007 its intention of setting up a potential for the Big Pharmas. This first-mover tactics which could become biotech research centre in California, latest trend shows no signs of slow- the trends for the next few years. Are similar in concept to Xerox’s PARC ing down in the next couple of years. generics really that bad? Generics, if centre that launched dozens of prod- Although it should be noted that proactively incorporated into a drug ucts and developed cutting-edge tech- the number of viable candidates will portfolio, can provide for additional nologies, by investing huge amounts to diminish over time and force a move- growth in a broader drug portfo- setup and finance the existing technol- ment towards more creative options. lio management strategy. In many ogies. Aventis moved quickly to acquire Many of these creative options have pharma companies, life cycle manage- Sanofi and is aggressively pursuing started with some Big Pharmas taking ment could be adjusted to incorporate more sizable biotech buyouts in the the proactive approach and imple- their own generics instead of planning not too distant future. Most of the Big menting some potentially very accre- on how to “defend” against them once Pharmas will continue to transform tive plans. Could these be just short- the patent protection expires. their drug pipelines with consolida- term trends or longer-term models of In fact, Novartis has made it a part tions of biopharmaceuticals for several commercialisation? of its aggressive strategic plan. It has years to come. seen generics as part of its product line and market penetration strategy by New models for consideration? Big Pharma looks more like big biotech A few companies have started deploy- using M&A to gain new groups. Many ing more original strategies out of of the pharmaceutical companies in Some drug makers like Novartis have necessity and 2007 had seen some the Indian sub-continent are now diversified so much that they have 14 P h a r m a F o c u s a si a ISSUE - 6 2008
- 4. S t r at e g y effectively become pharma-biotech Accretive Strategies hybrids. The biotech industry is Pharmaceutical Products Organisational Strategies expanding more rapidly than the pharma industry now. According to New drug formulations/applications M&A of larger Biopharmaceuticals the data released by IMS Health in Nutriceuticals Incubate and accelerate directed startups 2007, the sales of US biotech grew at various stages of development Cosmeceuticals 20% to US$ 40.3 billion in 2006, Develop targeted therepies Therapeutic Devices (personalised) while pharma sales grew by only 8% Biopharmaceuticals Expand geography with therapies to US$ 275 billion. Small Molecule Generics Grow expansion plans into Asia Another consideration is the size Biogenerics/Biosimiliars or economies of scale. The sheer size of Table 1 Big Pharma, if they could become more creative with these alternative strategies, Value Drivers: alternative growth strategies? could bring advantages in several criti- cal areas for biotech companies, espe- Nutraceuticals are fast becoming a strong growth category with the global market valued cially through complete acquisitions at the retail level, was approximately US$ 110 billion in 2005 according to the American by the Big Pharma. The large size of a Chemistry Report. Nutraceuticals are pharmaceutical-grade, cGMP manufactured Big Pharma company provides an edge products that can provide drug-like benefits and stop short of the extensive clinical in launching and promoting new drugs trials and efficacy claims that approved drugs go through. or additional indications or formula- Cosmeceuticals are pharmaceutical grade and, in some cases, regulatory-approved tions. A large company can increase products for cosmetics, dermatology indications, etc. One of the most recognised the number of bets that it can place on cosmeceutical is Botox®, the derma filler for wrinkles sold by Allergan of the US. Botox® is an engineered attenuated version of botulism and is approved by regulatory new technologies, novel therapies or authorities. additional disease indicators. If a big Another approach is to combine pharmaceutical compounds with medical devices to pharma company manages its clinical create a therapeutic device. These therapeutic devices can deliver, be formulated into programmes well, it can do the clinical a drug and/or be used as a device itself with therapeutic benefits. The drug-eluting trials both in-house as well as outsource cardiovascular stents used in angioplasty to prop open an artery are a good example to Contract Research Organisations of therapeutic devices. Many devices are being developed to work in the eye and other (CROs) to help it complete clinical organs to provide a more targeted, localised approach to therapeutic intervention. trials more quickly and broadly. The growth of Asia as both a technology and manufacturing region has become more drug formats. The accretive strategies Big Pharma reaching a point of critical mature in terms of product manufactur- table shows some of the more accretive mass where they will have to find ways ing and clinical trial programmes. Also, new strategies currently being employed to deconsolidate and yet, control the growth in original research represents by pharmaceutical companies to expand development and expansion of its estate a vast new population of people who product portfolios and growing more of drugs and intellectual property. would serve a growing pharmaceutical protected revenues. The biotechnology industry has market. In the end, larger Pharma compa- provided and will continue to provide nies will have to take a broader look at cutting-edge science, innovated tech- how they serve the overall healthcare nologies and products that Big Pharma Looking ahead Just as generics provide for a greater marketplace and provide, where needed, will be interested in. Biotech provides opportunity with expanding drug port- focussed solutions to unmet medical some, but not all, of the answers to the folios and life cycle management chal- and healthcare problems. An expand growth problems Big Pharma faces in lenges, a new and emerging group of and contract scenario is most likely with the years to come. product categories and technologies could provide new proprietary market Neil J Campbell is currently Chairman and CEo for Mosaigen™, Author growth to augment and complement Inc., a global Life Science development corporation, located the addition of biotech products and in rockville, Maryland and Partner with Endeavour Capital in Asia. During his career, he has successfully developed and technologies. introduced over 200 products in healthcare, life sciences Many companies are expanding and information technology. he earned his MBA and MA in their product offerings and are diver- Management Systems from Webster university in Saint Louis, Missouri and his BS-BA from Norwich university in New England. sifying outside traditional prescription w w w . p h a r m a f o c u s a s i a . c o m 15