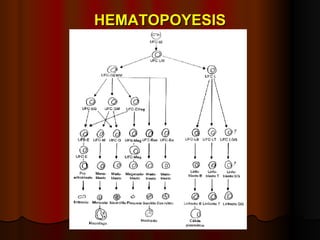
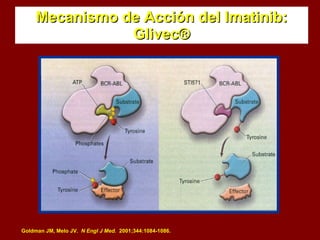
El documento describe los síndromes mieloproliferativos crónicos, incluyendo la leucemia mieloide crónica y la policitemia vera. La leucemia mieloide crónica se caracteriza por la presencia del cromosoma Filadelfia y la proteína de fusión Bcr-Abl. La policitemia vera es el resultado de la proliferación anómala de células pluripotenciales que son insensibles a la eritropoyetina. Ambas enfermedades presentan hipercelularidad medular, esplenome