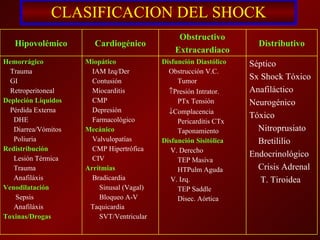
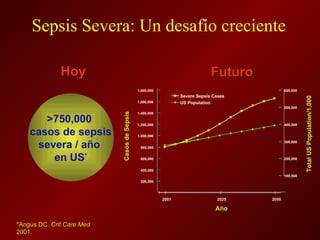
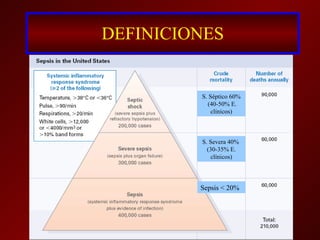
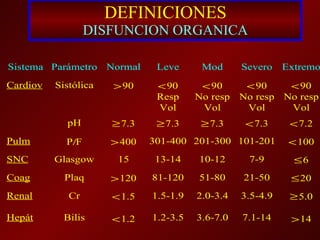
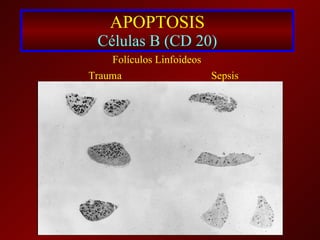
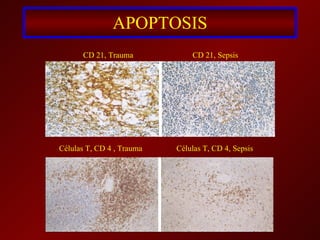
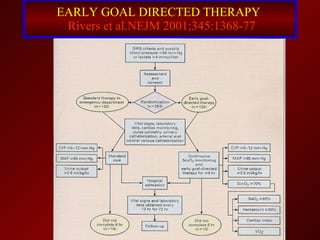
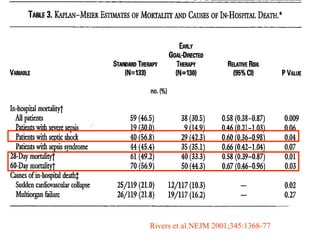
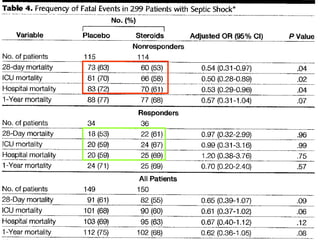
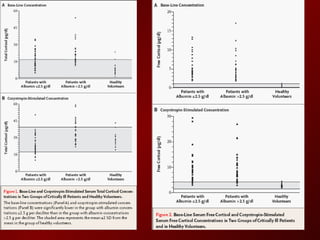
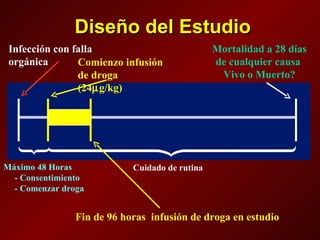
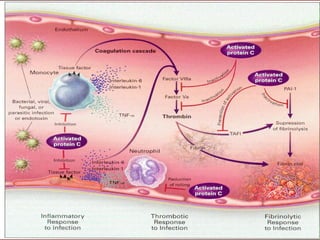
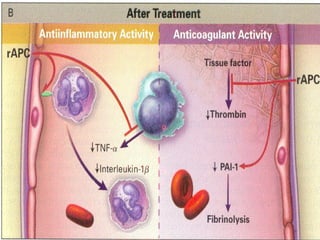
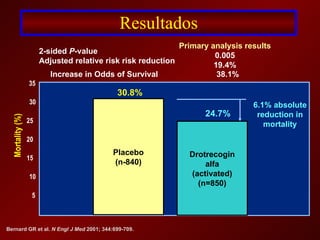
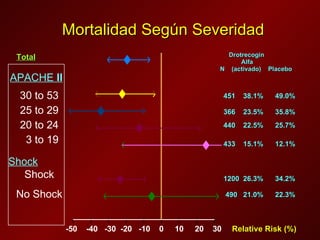
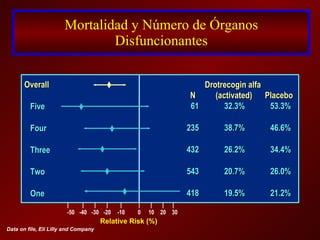
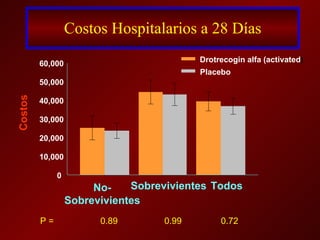
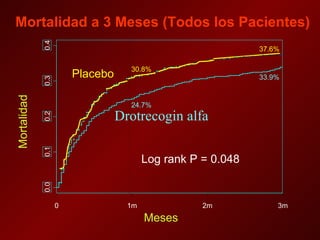
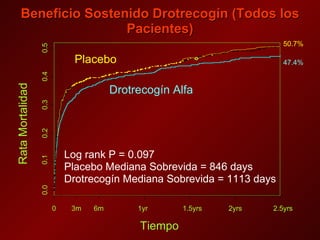
El documento trata sobre el shock séptico en embarazadas. Brevemente:
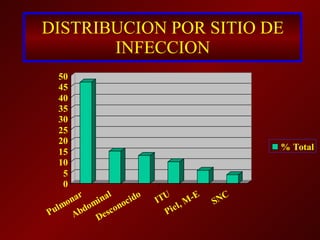
1) La prevalencia estimada de bacteremia en pacientes obstétricas en EEUU es de 7.5 por 1000 admisiones y la tasa de sepsis en esta población es de 8-10%.
2) Los organismos causales más comunes son Escherichia coli, bacteroides y streptococos.
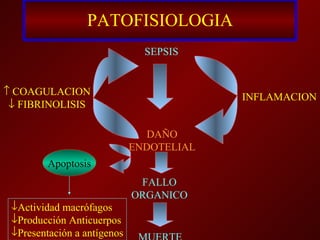
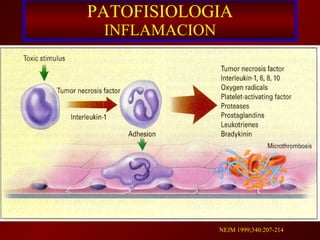
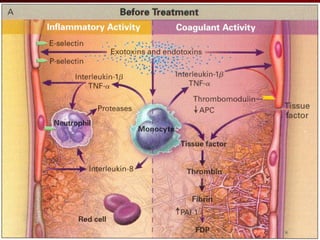
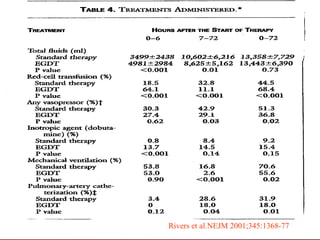
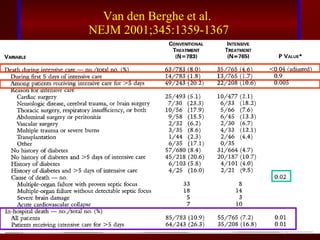
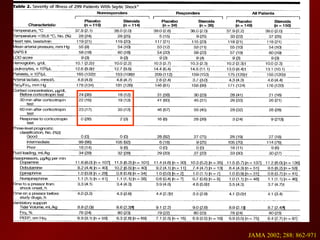
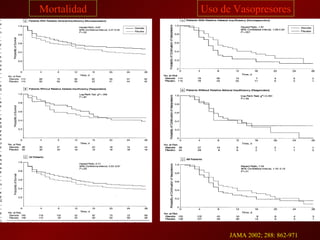
3) El tratamiento incluye identificar y controlar el foco infeccioso, iniciar antibióticos de amplio espectro tempranamente, resucitación